Abstract
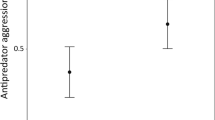
In antagonistic encounters individuals' displays reflect both the quality of the resource under dispute and their perception of the threat posed by their rival. All else being equal, as the value or threat to contested resources increases, so should an individual's level of aggression. Using a territorial species of Australian agamid lizard, the tawny dragon (Ctenophorus decresii), we tested three hypotheses about the relationship between territory quality and aggression. In three experiments we measured aggression whilst manipulating time in residence, perch height as a measure of territory quality and distance to an opponent's territory. Our measurement of aggression was a summary of behaviours used by tawny dragons in antagonistic displays (wrestling, hind-leg push-up display, chasing, raising of nuchal or vertebral crests, back arching, lateral compression, lowering dewlap, jerky walk and tail flick). Animals had significantly lower aggression scores when the opponent's territory was further away, but time in residence and perch height did not affect our measures of aggression. These experimental results provide good grounding for further tests of these hypotheses in field scenarios specifically manipulating distance between neighbours to determine what maintains the spatial distribution of tawny dragons in the wild.


Similar content being viewed by others
References
Arnott G, Elwood RW (2007) Fighting for shells: how private information about resource value changes hermit crab pre-fight displays and escalated fight behaviour. Proc Roy Soc B 274:3011–3017
Arnott G, Elwood RW (2008) Information gathering and decision making about resource value in animal contests. Anim Behav 76:529–542
Arnott G, Elwood RW (2009) Assessment of fighting ability in animal contests. Anim Behav 77:991–1004
Call J (1999) The effect of inter-opponent distance on the occurrence of reconciliation in stumptail (Macaca arctoides) and rhesus macaques (Macaca mulatta). Primates 40:515–523
Cooper WE, Avalos A (2010) Escape decisions by the syntopic congeners Sceloporus jarrovii and S. virgatus: comparative effects of perch height and of predator approach speed, persistence, and direction of turning. J Herpetol 44:425–430
DeCourcy K, Jenssen TA (1994) Structure and use of male territorial headbob signals by the lizard Anolis carolinensis. Anim Behav 47:251–262
Doake S, Elwood RW (2010) How resource quality differentially affects motivation and ability to fight in hermit crabs. Proc Roy Soc B 278:567–573
Dugan B (1982) A field study of the headbob displays of male green iguanas (Iguana iguana): variation in form and context. Anim Behav 30:327–338
Enquist M, Ghirlanda S, Hurd PL (1998) Discrete conventional signalling of a continuous variable. Anim Behav 56:749–754
Fayed SA, Jennions MD, Backwell PRY (2008) What factors contribute to an ownership advantage? Biol Lett 4:143–145
Fox SF, Baird TA (1992) The dear enemy phenomenon in the collared lizard, Crotaphytus collaris, with a cautionary note on experimental methodology. Anim Behav 44:780–782
Gibbons JRH (1977) Comparative ecology and behaviour of lizards of the Amphibolurus decresii species complex. PhD thesis, University of Adelaide
Gibbons JRH (1979) Hind leg pushup display of the Amphibolurus decresii species complex (Lacertilia: Agamidae). Copeia 1:29–40
Hammerstein P, Parker GA (1982) The asymmetric war of attrition. J Theor Biol 96:647682
Hardy ICW, Kemp DJ (2001) Skink skirmishes: why do owners win? Trends Ecol Evol 16:174
Hoefler CD (2002) Is contest experience a trump card? The interaction of residency status, experience, and body size on fighting success in Misumenoides formosipes Araneae: Thomisidae. J Insect Behav 15:779–790
Hof D, Hazlett N (2010) Low-amplitude song predicts attack in a North American wood warbler. Anim Behav 80:821–828
Husak JF (2004) Signal use by collared lizards, Crotaphytus collaris: the effects of familiarity and threat. Behav Ecol Sociobiol 55:602–607
Jenssen TA (1977) Evolution of anoline lizard display behaviour. Amer Zool 17:203–215
Knowlton N, Keller B (1982) Symmetric fights as a measure of escalation potential in a symbiotic, territorial snapping shrimp. Behav Ecol Sociobiol 10:289–292
Kokko H, Lopez-Sepulcre A, Morrell LJ (2006) From hawks and doves to self-consistent games of territorial behavior. Am Nat 167:901–912
Lindström K (1992) The effect of resource holding potential, nest size and information about resource quality on the outcome of intruder-owner conflicts in the sand goby. Behav Ecol Sociobiol 30:53–58
López P, Martín J (2002) Effects of female presence on intrasexual aggression in male lizards, Podarcis hispanicus. Anim Behav 28:491–498
Maynard Smith J, Parker GA (1976) The logic of asymmetric contests. Anim Behav 24:159–175
McMann S (2000) Effects of residence time on displays during territory establishment in a lizard. Anim Behav 59:513–522
Mohamad R, Monge J-P, Goubault M (2010) Can subjective resource value affect aggressiveness and contest outcome in parasitoid wasps? Anim Behav 80:629–636
Nijman VV, Heuts BA (2000) Effect of environmental enrichment upon resource holding power in fish in prior residence situations. Behav Proc 49:77–83
Nosil P (2002) Food fights in house crickets, Acheta domesticus, and the effects of body size and hunger level. Can J Zool 80:409–417
Olsson M (1992) Contest success in relation to size and residency in male sand lizards. Anim Behav 44:386–388
Olsson M, Shine R (2000) Ownership influences the outcome of male–male contests in the scincid lizard, Niveoscincus microlepidotus. Behav Ecol 11:587–590
Osborne L (2005a) Information content of male agonistic displays in the territorial tawny dragon (Ctenophorus decresii). J Ethol 23:189–197
Osborne L (2005b) Rival recognition in the territorial tawny dragon (Ctenophorus decresii). Acta Ethol 8:45–50
Payne RJH (1998) Gradually escalating fights and displays: the cumulative assessment model. Anim Behav 56:651–662
Peters RA, Ord TJ (2003) Display response of the jacky dragon, Amphibolurus muricatus (Lacertilia: Agamidae), to intruders: a semi-Markovian process. Austral Ecol 28:499–506
Radder RS, Saidapur SK, Shine R, Shanbhag BA (2006) The language of lizards: interpreting the function of visual displays of the Indian rock lizard, Psammophilus dorsalis (Agamidae). J Ethol 24:275–283
Rand WM, Rand AS (1976) Agonistic behavior in nesting iguanas: a stochastic analysis of dispute settlement dominated by the minimization of energy cost. Zeitschr f Tierpsychol 40:279–299
Stamps JA (1987) The effect of familiarity with a neighborhood on territory acquisition. Behav Ecol Sociobiol 21:273–277
Stocker AM, Huber R (2001) Fighting strategies in crayfish Orconectes rusticus (Decapoda, Cambaridae) differ with hunger state and the presence of food cues. Ethology 107:727–736
Stuart-Fox D (2006) Testing game theory models: fighting ability and decision rules in chameleon contests. Proc Roy Soc B 273:1555–1561
Stuart-Fox DM, Johnston GR (2005) Experience overrides colour in lizard contests. Behaviour 142:329–350
Számadó S (2008) How threat displays work: species-specific fighting techniques, weaponry and proximity risk. Anim Behav 76:1455–1463
Tachon G, Murray A, Gray D, Cade WH (1999) Agonistic displays and the benefits of fighting in the field cricket, Gryllus bimaculatus. J Insect Behav 12:533–543
Taylor PW, Hasson O, Clark DL (2001) Initiation and resolution of jumping spider contests: roles for size, proximity, and early detection of rivals. Behav Ecol Sociobiol 50:403–413
Turner G (1994) The fighting tactics of male mouthbrooding cichlids: the effects of size and residency. Anim Behav 47:655–662
Vitt LJ, Cooper WE Jr (1985) The evolution of sexual dimorphism in the skink Eumeces laticeps: an example of sexual selection. Can J Zool 63:995–1002
Whiting MJ (1999) When to be neighbourly: differential agonistic responses in the lizard Platysaurus broadleyi. Behav Ecol Sociobiol 46:210–214
Acknowledgments
The authors would like to thank Patricia RY Backwell for comments on experimental design and on the manuscript. The Animal Experimentation Ethics Committee of the Australian National University approved the research presented here under Protocol No. F.BTZ.37.01. Collection of animals was conducted within the guidelines of South Australia National Parks and Wildlife Service under permit M24494. Animals were housed under the guidelines of Environment ACT (Australian Capital Territory) under permit K8164. The experiment complies with all current laws in Australia, and approval was successfully sought from all necessary sources.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Osborne, L., Umbers, K.D.L. & Keogh, J.S. The effects of perch height, time in residence and distance from opponent on aggressive display in male lizards. acta ethol 16, 41–46 (2013). https://doi.org/10.1007/s10211-012-0135-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10211-012-0135-z