Abstract
Objective
The Hemraude study was conducted to describe the profile of patients with HA, disease management, and economic burden in a collective perspective.
Methods
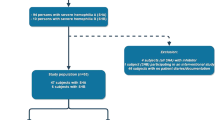
This retrospective study was conducted using the French administrative healthcare claims database SNIIRAM/SNDS. Male patients treated for hemophilia A with a long-term illness (ALD) status or invalidity were included in the study between January 1, 2016 and December 31, 2017. Patients were classified in six treatment groups: no treatment, on-demand FVIII, prophylactic FVIII, FVIII in immune tolerance induction (ITI) protocol, on-demand bypassing agents, and prophylactic bypassing agents. Patients treated with FVIII in ITI protocol and those treated with bypassing agents are deemed to have developed inhibitors. HA patients were compared to a control population without coagulation disorder and matched (ratio 1:3) on age and sex.
Results
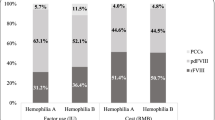
A total of 4172 patients were included in the analysis, aged on average 35.2 years, 5.3% had HIV infection, and 8.8% had hepatitis B or C. In 2017, half of the patients received no treatment for HA, 46.7% were treated with FVIII (25% on demand, 20.6% with prophylaxis, and 1.1% ITI), 1.5% with bypassing agents. Patients treated with prophylactic treatments, either inhibitor or non-inhibitor, were less likely to be hospitalized for severe bleeding compared to patients receiving on-demand treatments. The average annual costs for HA management per patient were 72,209.60 €. The highest costs were observed in patients treated with FVIII in ITI protocol and those receiving prophylactic bypassing agents.
Conclusion
Direct costs of HA treatments for HA may be very high especially in the small percentage of patients developing inhibitors or treated with ITI protocol.
Similar content being viewed by others
References
Kloosterman, F., Zwagemaker, A.F., Abdi, A., Gouw, S., Castaman, G., Fijnvandraat, K.: Hemophilia management: huge impact of a tiny difference. Res Pract Thromb Haemost 4(3), 377–385 (2020). https://doi.org/10.1002/rth2.12314.PMID:32211572;PMCID:PMC7086468
Wong, T., Recht, M.: Current options and new developments in the treatment of haemophilia. Drugs 71(3), 305–320 (2011)
Mannucci, P.M., Tuddenham, E.G.: The hemophilias–from royal genes to gene therapy. N Engl J Med 344, 1773–1779 (2001)
World Federation of Hemophilia: Report on the Annual Global Survey 2019. October 2020. https://elearning.wfh.org/resource/report-on-the-annual-global-survey-2019/ (2020). Accessed 03 Nov 2020
Doncarli, A., Demiguel, V., Guseva Canu, I., Goulet, V., Bayart, S., Calvez, T., Castet, S., Dalibard, V., Demay, Y., Frotscher, B., Goudemand, J., Lambert, T., Milien, V., Oudot, C., Sannié, T., Chambost, H.: FranceCoag network. FranceCoag: a 22-year prospective follow-up of the national French cohort of patients with inherited bleeding disorders. Eur J Epidemiol 34(5), 521–532 (2019)
Soucie, J.M., Monahan, P.E., Kulkarni, R., Konkle, B.A., Mazepa, M.A.: US hemophilia treatment center network. The frequency of joint hemorrhages and procedures in non-severe hemophilia A vs B. Blood Adv 2(16), 2136–2144 (2018)
Srivastava, A., Brewer, A.K., Mauser-Bunschoten, E.P., et al.: Guidelines for the management of hemophilia. Haemophilia 19(1), e1–e47 (2013)
Gringeri, A., Ewenstein, B., Reininger, A.: The burden of bleeding in haemophilia: is one bleed too many? Haemophilia 20(4), 459–463 (2014)
Valentino, L.A.: Blood-induced joint disease: the pathophysiology of hemophilic arthropathy. J Thromb Haemost 8(9), 1895–1902 (2010)
Manco-Johnson, M.J., Abshire, T.C., Shapiro, A.D., et al.: Prophylaxis versus episodic treatment to prevent joint disease in boys with severe hemophilia. N Engl J Med 357(6), 535–544 (2007)
Garagiola, I., Palla, R., Peyvandi, F.: Risk factor for inhibitor development in severe hemophilia A. Thromb Res 168, 20–27 (2018)
Dekoven, M., Wisniewski, T., Petrilla, A., et al.: Health-related quality of life in haemophilia patients with inhibitors and their caregivers. Haemophilia 19(2), 287–293 (2013)
Kroner, B.L.: Comparison of the international immune tolerance registry and the North American immune tolerance registry. Vox Sang. 77(Suppl 1), 33–37 (1999)
Yoo, K.Y., Joo, S.C., Choi, Y.M.: Long-term course of anti-factor VIII antibody in patients with hemophilia A at a single center. Blood Res. 51(1), 37–43 (2016)
Ljung, R.C.R.: How I manage patients with inherited haemophilia A and B and factor inhibitors. Br J Haematol 180(4), 501–510 (2018)
Kitazawa, T., Igawa, T., Sampei, Z., et al.: A bispecific antibody to factors IXa and X restores factor VIII hemostatic activity in a hemophilia A model. Nat Med. 18(10), 1570–1574 (2012)
Harkins Druzgal, C., Kizilocak, H., Brown, J., Sennett, M., Young, G.: Neutralizing antidrug antibody to emicizumab in a patient with severe hemophilia A with inhibitors: new case with detailed laboratory evaluation. J Thromb Haemost (2020). https://doi.org/10.1111/jth.14957
Colowick, A.B., Bohn, R.L., Avorn, J., Ewenstein, B.M.: Immune tolerance induction in hemophilia patients with inhibitors: costly can be cheaper. Blood 96(5), 1698–1702 (2000)
Grimes, D.A., Schulz, K.F.: Compared to what? Finding controls for case-control studies. Lancet 365(9468), 1429–1433 (2005)
Bouillon, K., Bertrand, M., Maura, G., Blotière, P.O., Ricordeau, P., Zureik, M.: Risk of bleeding and arterial thromboembolism in patients with non-valvular atrial fibrillation either maintained on a vitamin K antagonist or switched to a non-vitamin K-antagonist oral anticoagulant: a retrospective, matched-cohort study. Lancet Haematol 2(4), e150–e159 (2015)
O’Hara, J., Hughes, D., Camp, C., Burke, T., Carroll, L., Diego, D.G.: The cost of severe haemophilia in Europe: the CHESS study. Orphanet J Rare Dis 12(1), 106 (2017)
Valentino, L.A., Pipe, S.W., Tarantino, M.D., Ye, X., Xiong, Y., Luo, M.P.: Healthcare resource utilization among haemophilia A patients in the United States. Haemophilia 18, 332–338 (2012)
Guh, S., Grosse, S.D., McAlister, S., Kessler, C.M., Soucie, J.M.: Health care expenditures for Medicaid-covered males with haemophilia in the United States, 2008. Haemophilia 18, 276–283 (2012)
Dalton, D.R.: Hemophilia in the managed care. Am J Manag Care 21(suppl 6), S123–S130 (2015)
Zhou, Z.Y., Koerper, M.A., Johnson, K.A., Riske, B., Baker, J.R., Ullman, M., Curtis, R.G., Poon, J.L., Lou, M., Nichol, M.B.: Burden of illness: direct and indirect costs among persons with hemophilia A in the United States. J Med Econ 18(6), 457–465 (2015). https://doi.org/10.3111/13696998.2015.1016228
Tencer, T., Friedman, H.S., Li-McLeod, J., Johnson, K.: Medical costs and resource utilization for hemophilia patients with and without HIV or HCV infection. J Manag Care Pharm 13(9), 790–798 (2007)
Funding
This study was supported by an unrestricted grant from Roche.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Laurendeau, C., Goudemand, J., Trossaert, M. et al. Costs and management of patients with hemophilia A in France: the Hemraude study. Eur J Health Econ 23, 23–32 (2022). https://doi.org/10.1007/s10198-021-01339-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10198-021-01339-4