Abstract
Objective
To conduct a systematic review of cost-effectiveness, cost-utility, and cost-benefit studies of DPP-4 inhibitors for diabetes treatment versus other antidiabetics.
Methods
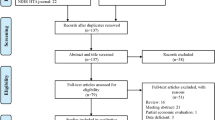
Three investigators searched the CRD York, Tufts CEA Registry, and MEDLINE databases through 2015. We reviewed all potentially relevant titles and abstracts, and screened full-text articles, according to inclusion criteria. We established a quality score for each study based on a 35-item list.
Results
A total of 295 studies were identified, of which 20 were included. The average quality score was 0.720 on a 0–1 scale. All studies were performed in high- and middle-income countries, using a 3rd-party payer perspective and randomized clinical trials to measure effectiveness. Sitagliptin, saxagliptin and vildagliptin had an ICER below 25,000 €/QALY, as second-line and as add-ons to metformin, in comparison to sulfonylureas. When compared with sitagliptin, liraglutide (GLP-1 receptor agonist) had an ICER of up to 22,724 €/QALY for the 1.2-mg dosage, and up to 32,869 €/QALY for the 1.8-mg dosage. Insulin glargine was dominant when compared with sitagliptin.
Conclusions
According to the WHO threshold applied to the country and year of each study, DPP-4 inhibitors were highly cost-effective as second-line, as add-ons to metformin, in comparison with sulfonylureas. More recent therapies (GLP-1 receptor agonists and insulin glargine) were highly cost-effective in comparison to DPP-4 inhibitors. These results were obtained, however, on the basis of a limited number of studies, relying on the same few clinical trials, and financed by manufacturers. Further independent research is needed to confirm these findings.

Similar content being viewed by others
References
International Diabetes Federation. IDF Diabetes atlas, 7th edition. Brussels, Belgium: International Diabetes Federation, 2015. http://www.diabetesatlas.org. Accessed 20 Apr 2016
Kanavos, P., Aardweg, S., Schurer, W.: Diabetes expenditure, burden of disease and management in 5 EU countries. LSE Health, London School of Economics, London (2012)
American Diabetes Association. Economic costs of diabetes in the U.S. in 2012. Diabetes Care 2013. http://care.diabetesjournals.org/content/early/2013/03/05/dc12-2625.full.pdf. Accessed 20 Apr 2016
Inzucchi, S.E., Bergenstal, R.M., Buse, J.B., Diamant, M., Ferrannini, E., Nauck, M., Peters, A.L., Tsapas, A., Wender, R., Matthews, D.R.: Management of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach. Update to a position statement of the American Diabetes Association and the European Association for the study of diabetes. Diabetes Care 38, 140–149 (2015). doi:10.2337/dc14-2441
Liberati, A., Altman, D.G., Tetzlaff, J., Mulrow, C., Gøtzsche, P.C., Ioannidis, J.P.A., Clarke, M., Devereaux, P.J., Kleijnen, J., Moher, D.: The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann. Int. Med. 151, W65–W94 (2009)
Centre for Reviews and Dissemination: Systematic reviews. CRD’s guidance for undertaking reviews in healthcare. CRD University of York, York (2008)
Drummond, M., Jefferson, T.: Guidelines for authors and peer reviewers of economic submissions to the BMJ. The BMJ Economic Evaluation Working Party. BMJ 313, 275–283 (1996)
World Bank. World Bank Country and Lending Groups Data. https://datahelpdesk.worldbank.org/knowledgebase/articles/906519#High_income (2016). Accessed 6 Jul 2016
Hay, J., Smeeding, J., Carrol, N.V., Drummond, M., Garrison, L.P., Mansley, E.C., Mullins, C.D., Mycka, J.M., Seal, B., Shi, L.: Good research practices for measuring drug costs in cost effectiveness analyses: issues and recommendations: the ISPOR Drug Cost Task Force report—part I. Value Health 13, 3–7 (2010)
Sachs, J.D.: Macroeconomics and health: investing in health for economic development. Report of the Commission on Macroeconomics and Health. World Health Organization, Geneva (2003)
Baltussen, R.M.P.M., Adam, T., Tan-Torres, E.T., Hutubessy, R.C.W., Acharya, A., Evans, D.B., Murray, C.J.L.: Making choices in health: WHO guide to cost-effectiveness analysis. World Health Organization, Geneva (2003)
Schwarz, B., Gouveia, M., Chen, J., Nocea, G., Jameson, K., Cook, J., Krishnarajah, G., Alemao, E., Yin, D., Sintonen, H.: Cost-effectiveness of sitagliptin-based treatment regimens in European patients with type 2 diabetes and haemoglobin A1c above target on metformin monotherapy. Diabetes Obes. Metab. 10(Supplement 1), 43–55 (2008)
Pereira, R., Gouveia, M., Martins, A.P.: Análise custo-efectividade de sitagliptina quando adicionada a metformina em doentes com diabetes tipo 2 em Portugal. Revista Portuguesa de diabetes 7(1), 13–23 (2012)
Elgart, J.F., Caporale, J.E., Gonzalez, L., Aiello, E., Waschbusch, M., Gagliardino, J.J.: Treatment of type 2 diabetes with saxagliptin: a pharmacoeconomic evaluation in Argentina. Health Econ. Rev. 3(1), 11 (2013)
Erhardt, W., Bergenheim, K., Duprat-Lomon, I., McEwan, P.: Cost effectiveness of saxagliptin and metformin versus sulfonylurea and metformin in the treatment of type 2 diabetes mellitus in Germany: a Cardiff Diabetes Model analysis. Clin. Drug Investig. 32(3), 189–202 (2012)
Granstrom, O., Bergenheim, K., McEwan, P., Sennfalt, K., Henriksson, M.: Cost-effectiveness of saxagliptin (Onglyza) in type 2 diabetes in Sweden. Primary Care Diabetes 6(2), 127–136 (2012)
Bergenheim, K., Williams, S.A., Bergeson, J.G., Stern, L., Sriprasert, M.: US cost-effectiveness of saxagliptin in type 2 diabetes mellitus. Am. J. Pharm. Benefits 4(1), 20–28 (2012)
Carvalho, D., Contente, M., Silva, C., Trindade, R.: Saxagliptin in the treatment of diabetes mellitus type 2 in Portugal: a study of cost-utility in the perspective of society. Revista Portuguesa da Diabetes 9(2), 60–72 (2014)
Grzeszczak, W., Czupryniak, L., Kolasa, K., Sciborski, C., Lomon, I.D., McEwan, P.: The cost-effectiveness of saxagliptin versus NPH insulin when used in combination with other oral antidiabetes agents in the treatment of type 2 diabetes mellitus in Poland. Diabetes Technol. Therap. 14(1), 65–73 (2012)
Nita, M.E., Eliaschewitz, F.G., Ribeiro, E., Asano, E., Barbosa, E., Takemoto, M., Donato, B., Rached, R., Rahal, E.: Cost-effectiveness and budget impact of saxagliptin as additional therapy to metformin for the treatment of diabetes mellitus type 2 in the Brazilian private health system. Rev. Assoc. Med. Bras. 58(3), 294–301 (2011)
Viriato, D., Calado, F.; Gruenberger, J.B., Ong, S.H., Carvalho, D., Silva-Nunes, J., Johal, S., Viana, R.: Cost-effectiveness of metformin plus vildagliptin compared with metformin plus sulphonylurea for the treatment of patients with type 2 diabetes mellitus: a Portuguese healthcare system perspective. J. Med. Econ. 17(7), 499–507 (2014)
Li, Q., Chitnis, A., Hammer, M., Langer, J.: Real-world clinical and economic outcomes of liraglutide versus sitagliptin in patients with type 2 diabetes mellitus in the United States. Diabetes Ther. 5(2), 579–590 (2014)
Carlsson, K.S., Persson, U.: Cost-effectiveness of add-on treatments to metformin in a Swedish setting: liraglutide vs sulphonylurea or sitagliptin J. Med. Econ. 17(9), 658–669 (2014). doi:10.3111/13696998.2014.933110. (Epub 2014 Jun 26)
Tzanetakos, C., Melidonis, A., Verras, C., Kourlaba, G., Maniadakis, N.: Cost-effectiveness analysis of liraglutide versus sitagliptin or exenatide in patients with inadequately controlled type 2 diabetes on oral antidiabetic drugs in Greece. BMC Health Serv. Res. 14, 419 (2014)
Langer, J., Hunt, B., Valentine, W.J.: Evaluating the short-term cost-effectiveness of liraglutide versus sitagliptin in patients with type 2 diabetes failing metformin monotherapy in the United States. J Manag. Care Pharm. 19(3), 237–246 (2013)
Perez, A., Raya, P.M., Arellano, A.R., Briones, T., Hunt, B., Valentine, W.J.: Cost-effectiveness analysis of incretin therapy for type 2 diabetes in Spain:1.8 mg liraglutide vs. sitagliptin. Diabetes Ther. 6, 61–74 (2015)
Raya, P.M., Perez, A., Arellano, A.R., Briones, T., Hunt, B., Valentine, W.J.: Incretin therapy for type 2 diabetes in Spain: a cost-effectiveness analysis of liraglutide versus sitagliptin. Diabetes Ther. 4, 417–430 (2013)
Davies, M.J., Chubb, B.D., Smith, I.C., Valentine, W.J.: Cost-utility analysis of liraglutide compared with sulphonylurea or sitagliptin, all as add-on to metformin monotherapy in type 2 diabetes mellitus. Diabet. Med. 29(3), 313–320 (2012)
Lee, W.C., Samyshkin, Y., Langer, J., Palmer, J.L.: Long-term clinical and economic outcomes associated with liraglutide versus sitagliptin therapy when added to metformin in the treatment of type 2 diabetes: a CORE diabetes model analysis. J. Med. Econ. 15(Supplement 2), 28–37 (2012)
Brown, S.T., Grima, D.G., Sauriol, L.: Cost-effectiveness of insulin glargine versus sitagliptin in insulin-naive patients with type 2 diabetes mellitus. Clin. Ther. 36(11), 1576–1587 (2014)
Kiadaliri, A.A., Gerdtham, U.G., Eliasson, B., Carlsson, K.S.: Cost-utility analysis of glucagon-like peptide-1 agonists compared with dipeptidyl peptidase-4 inhibitors or neutral protamine hagedorn basal insulin as add-on to metformin in type 2 diabetes in Sweden. Diabetes Ther. 5, 591–607 (2014)
Chen, J., Alemao, E., Yin, D., Cook, J.: Development of a diabetes treatment simulation model: with application to assessing alternative treatment intensification strategies on survival and diabetes related complications. Diabetes Obes. Metab. 10(47), 1747–1759 (2008)
Clarke, P.M., Gray, A.M., Briggs, A., Farmer, A.J., Fenn, P., Stevens, R.J., Matthews, D.R., Stratton, I.M., Holman, R.R.: A model to estimate the lifetime health outcomes of patients with diabetes type 2: the United Kingdom prospective diabetes study (UKPDS) outcomes model (UKPDS no.68). Diabetologia 47, 1747–1759 (2004)
Palmer, A.J., Mount Hood 5 Modeling Group.: Computer modeling of diabetes and its complications: a report on the fifth Mount Hood challenge meeting. Value Health 16(4), 670–685 (2013)
McEwan, P., Peters, J.R., Bergenheim, K., Currie, C.J.: Evaluation of the costs and outcomes from changes in risk factors in type 2 diabetes using the Cardiff stochastic simulation cost-utility model (DiabForecaster). Curr. Med. Res. Opin. 22, 121–129 (2006)
McEwan, P., Bergenheim, K., Yuan, Y., Tetlow, A.P., Gordon, J.P.: Assessing the relationship between computational speed and precision: a case study comparing an interpreted versus compiled programming language using a stochastic simulation model in healthcare. Pharmacoeconomics 28, 665–674 (2010)
Palmer, A.J., Roze, S., Valentine, W.J., Minshall, M.E., Foos, V., Lurati, F.M., Lammert, M., Spinas, G.A.: The core diabetes model: projecting long-term clinical outcomes, costs and cost-effectiveness of interventions in diabetes mellitus (types 1 and 2) to support clinical and reimbursement decision-making. Curr. Med. Res. Opin. 20(1 Suppl), S5–S26 (2004)
Pratley, R., Nauck, M., Bailey, T., Montanya, E., Cuddihy, R., Filetti, S., Garber, A., Thomses, A.B., Hartvig, H., Davies, M.: One year of liraglutide treatment offers sustained and more effective glycaemic control and weight reduction compared with sitagliptin, both in combination with metformin, in patients with type 2 diabetes: a randomised, parallel-group, open-label trial. Int. J. Clin. Pract. 65, 397–407 (2011)
OECD. OECD.Stat. https://stats.oecd.org/Index.aspx?DataSetCode=PDB_LV## (2015). Accessed 29 Jun 2016
Waugh, N., Cummins, E., Royle, P., Clar, C., Marien, M., Richter, B., Philip, S.: Newer agents for blood glucose control in type 2 diabetes: systematic review and economic evaluation. Health Technol. Assess. 14(36), 1–248 (2010)
Geng, J., Yu, H., Ma, O.Y., Zhang, P., Chen, Y.: Cost effectiveness of dipeptidyl peptidase-4 inhibitors for type 2 diabetes. Pharmacoeconomics 33, 581–597 (2015)
Ahuja, V., Chou, C.H.: Novel therapeutics for diabetes: uptake, usage trends, and comparative effectiveness. Curr. Diabetes Rep. 16, 47 (2016)
Bell, C.H., Urbach, D.R., Ray, J.G., Bayoumi, A., Rosen, A.B., Greenberg, D., Neumann, P.J.: Bias in published cost effectiveness studies: systematic review. BMJ 332(7543), 699–703 (2006)
Turner, L.W., Nartey, D., Stafford, R.S., Singh, S., Alexander, G.C.: Ambulatory treatment of type 2 diabetes in the U.S., 1997–2012. Diabetes Care 37(4), 985–992 (2014). doi:10.2337/dc13-2097
Acknowledgments
This research had no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. The authors have no affiliations or financial involvement that conflicts with the material presented in this paper. The authors are not employed in the pharmaceutical industry and, at the time of this research, had not performed nor were conducting any research study pertaining to the DPP-4 inhibitors group, nor were conducting any research for any of the marketing authorization holders of those therapeutic agents.
Appendix
Appendix
See Table 3.
Template for each study extracted and recorded information:
-
Title [incl. author(s) and journal]
-
Type of economic evaluation
-
Objective
-
Interventions
-
Comparators
-
Methods (Analytical approach; Perspective; Time Horizon; Population; Effectiveness data; Monetary benefit and utility valuation; Measure of benefit; Cost data; Discount rate; Analysis of uncertainty)
-
Results
-
Authors’ Conclusions
-
Limitations
-
Affiliation to pharmaceutical industry
Search strings for search:
-
Sitagliptin OR Saxagliptin OR Vildagliptin OR Alogliptin OR Linagliptin assessed in March 2015
-
Sitatgliptin AND Saxagliptin assessed in March 2015
-
Sitagliptin AND Vildagliptin assessed in March 2015
-
Sitagliptin AND Alogliptin assessed in March 2015
-
Sitagliptin AND Linagliptin assessed in March 2015
-
Saxagliptin AND Vildagliptin assessed in March 2015
-
Saxagliptin AND Alogliptin assessed in March 2015
-
Saxagliptin AND Linagliptin assessed in March 2015
-
Vildagliptin AND Alogliptin assessed in March 2015
-
Vildagliptin AND Linagliptin assessed in March 2015
-
Alogliptin AND Linagliptin assessed in March 2015
Rights and permissions
About this article
Cite this article
Baptista, A., Teixeira, I., Romano, S. et al. The place of DPP-4 inhibitors in the treatment algorithm of diabetes type 2: a systematic review of cost-effectiveness studies. Eur J Health Econ 18, 937–965 (2017). https://doi.org/10.1007/s10198-016-0837-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10198-016-0837-7