Abstract
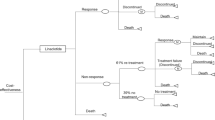
Presently, linaclotide is the only EMA-approved therapy indicated for the treatment of irritable bowel syndrome with constipation (IBS-C). This study sought to determine the cost-effectiveness of linaclotide compared to antidepressants for the treatment of adults with moderate to severe IBS-C who have previously received antispasmodics and/or laxatives. A Markov model was created to estimate costs and QALYs over a 5-year time horizon from the perspective of NHS Scotland. Health states were based on treatment satisfaction (satisfied, moderately satisfied, not satisfied) and mortality. Transition probabilities were based on satisfaction data from the linaclotide pivotal studies and Scottish general all-cause mortality statistics. Treatment costs were calculated from the British National Formulary. NHS resource use and disease-related costs for each health state were estimated from Scottish clinician interviews in combination with NHS Reference costs. Quality of life was based on EQ-5D data collected from the pivotal studies. Costs and QALYs were discounted at 3.5 % per annum. Uncertainty was explored through extensive deterministic and probabilistic sensitivity analyses. Over a 5-year time horizon, the additional costs and QALYs generated with linaclotide were £659 and 0.089, resulting in an incremental cost-effectiveness ratio of £7370 per QALY versus antidepressants. Based on the probabilistic sensitivity analysis, the likelihood that linaclotide was cost-effective at a willingness to pay of £20,000 per QALY was 73 %. Linaclotide can be a cost-effective treatment for adults with moderate-to-severe IBS-C who have previously received antispasmodics and/or laxatives in Scotland.



Similar content being viewed by others
References
NICE clinical guideline 61: Irritable bowel syndrome in adults. Diagnosis and management of irritable bowel syndrome in primary care. https://www.nice.org.uk/guidance/cg61 (2008). Accessed June 2014
Mearin, F., Badia, X., Balboa, A., et al.: Predictive factors of irritable bowel syndrome improvement: 1-year prospective evaluation in 400 patients. Aliment. Pharmacol. Ther. 23, 815–826 (2006)
NHS National Services Scotland: An investigation into the prevalence, management and healthcare burden of irritable bowel syndrome in Scotland. Information Services Division. http://www.isdscotland.org/Health-Topics/Prescribing-and-medicines/ISD-Guide-to-Prescribing-Services-Web-Accessible-Version-V1.pdf (2012). Accessed June 2014
Wilson, S., Roberts, L., Roalfe, A., et al.: Prevalence of irritable bowel syndrome: a community survey. Br. J. Gen. Pract. 54, 495–502 (2004)
Layer, P., Andresen, V., Pehl, C., et al.: Irritable bowel syndrome: German consensus guidelines on definition, pathophysiology and management. German Society of Digestive and Metabolic Diseases (DGVS) and German Society of Neurogastroenterology and motility (DGNM). Z. Gastroenterol. 49, 237–293 (2011)
Dibonaventura, M.D., Prior, M., Prieto, P., et al.: Burden of constipation-predominant irritable bowel syndrome (IBS-C) in France, Italy, and the United Kingdom. Clin. Exp. Gastroenterol. 5, 203–212 (2012)
Hungin, A.P., Whorwell, P.J., Tack, J., et al.: The prevalence, patterns and impact of irritable bowel syndrome: an international survey of 40,000 subjects. Aliment. Pharmacol. Ther. 17(5), 643–650 (2003)
Maxion-Bergemann, S., Thielecke, F., Abel, F., et al.: Costs of irritable bowel syndrome in the UK and US. Pharmacoeconomics 24(1), 21–37 (2006)
Spiegel, B.M., Kanwal, F., Naliboff, B., et al.: The impact of somatization on the use of gastrointestinal health-care resources in patients with irritable bowel syndrome. Am. J. Gastroenterol. 100(10), 2262–2273 (2005)
Gralnek, I.M., Hays, R.D., Kilbourne, A., et al.: The impact of irritable bowel syndrome on health-related quality of life. Gastroenterology 119, 654–660 (2000)
Andersson, E., Ljotsson, B., Smit, F., et al.: Cost-effectiveness of Internet-based cognitive behaviour therapy for irritable bowel syndrome: results from a randomized controlled trial. BMC Public Health 11, 215 (2011)
Tack, J., Muller-Lissner, S., Bytzer, P., et al.: A randomised controlled trial assessing the efficacy and safety of repeated tegaserod therapy in women with irritable bowel syndrome with constipation. Gut 54(12), 1707–1713 (2005)
CEGEDIM, Longitudinal Patient Database (LPD) for the UK. https://www.cegedimstrategicdata.com/Corporate/OurSolutions/Patient/Pages/default.aspx (2010). Accessed June 2014
WG Consulting. Identifying current practice for the treatment of moderate to severe irritable bowel syndrome with constipation (IBS-C) in Scotland (2012)
Ruepert, L., Quartero, A.O., de Wit, N.J., et al.: Bulking agents, antispasmodics and antidepressants for the treatment of irritable bowel syndrome (Review). The Cochrane Library. 11 (2011)
Ford, A.C., Vandvik, P.O.: Irritable bowel syndrome. Clin. Evid. 01, 410 (2012)
GRADE Working Group. http://www.gradeworkinggroup.org/index.htm. Accessed July 2013
Rao, S., Lembo, A., Shiff, S., et al.: A 12-week, randomized, controlled trial with a 4-week randomized withdrawal period to evaluate the efficacy and safety of linaclotide in irritable bowel syndrome with constipation. Am. J. Gastroenterol. 107(11), 1714–1724 (2012)
Chey, W.D., Lembo, A.J., Lavins, B.J., et al.: Linaclotide for irritable bowel syndrome with constipation: a 26-week, randomized, double-blind, placebo-controlled trial to evaluate efficacy and safety. Am. J. Gastroenterol. 107(11), 1702–1712 (2012)
Quigley, E.M., Tack, J., Chey, W.D., et al.: Randomised clinical trials: linaclotide Phase 3 studies in IBS-C—a prespecified further analysis based on European Medicines Agency-specified endpoints. Pharmacol. Ther. 37(1), 49–61 (2013)
Summary of Product Characteristics—Constella. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/002490/WC500135622.pdf (2013). Accessed June 2014
Scottish Medicines Consortium, linaclotide hard capsules, 290 micrograms (Constella®) SMC No. (869/13). https://www.scottishmedicines.org.uk/files/advice/linaclotide__Constella__FINAL_May_2013.doc__for_website.pdf (2013). Accessed June 2014
Bracco, A., Jonsson, B., Ricci, J.F., et al.: Economic evaluation of tegaserod vs. placebo in the treatment of patients with irritable bowel syndrome: an analysis of the TENOR study. Value Health. 10(4), 238–246 (2007)
Kantar Health. The IBS—Chronic Constipation Market: UK Core Therapeutic Report. National Health and Wellness Survey. http://www.kantarhealth.com/docs/ebooks/global-health-and-wellness-report.pdf (2010). Accessed June 2014
Office for National Statistics. Interim Life Tables Scotland. 1980–1982 to 2007–2009. http://www.ons.gov.uk/ons/search/index.html?pageSize=50&sortBy=none&sortDirection=none&newquery=interim+lifetables (2009). Accessed June 2014
Dolan, P.: Modelling valuations for health states. Med. Care 35(11), 1095–1108 (1997)
Joint Formulary Committee.: British National Formulary (2013). http://www.bnf.org. Accessed July 2013
Curtis, L.: Unit Costs of Health and Social Care. http://www.pssru.ac.uk/archive/pdf/uc/uc2012/fullwith-covers.pdf (2012). Accessed June 2014
Department of Health.: NHS Reference Costs 2011–2012. https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/213060/2011-12-referencecosts-publication.pdf (2012). Accessed June 2014
Spiller, R., Aziz, Q., Creed, F., et al.: Guidelines on the irritable bowel syndrome: mechanisms and practical management. Gut 56(12), 1770–1798 (2007)
Lembo, A.: Factors contributing to treatment satisfaction in patients with irritable bowel syndrome with constipation (IBS-C) treated with linaclotide versus placebo, pp. 19–22. Poster presented at Digestive Disease Week, San Diego (2012)
Creed, F., Ratcliffe, J., Fernandez, L., et al.: Health-related quality of life and health care costs in severe, refractory irritable bowel syndrome. Ann. Intern. Med. 134(9 Pt 2), 860–868 (2001)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Miguel Moya, Meritxell Falqués, Mark Rance, and Leandro Lindner are/were employees of Almirall, S.A. Mark Fisher previously worked for WG Consulting Healthcare Limited, who was funded by Almirall, S.A. to develop the cost-effectiveness model and write the manuscript. Andrew Walker has worked as a consultant for Almirall, S.A. and other pharmaceutical companies. Douglas Taylor is currently an employee of Ironwood Pharmaceuticals, Inc.
Rights and permissions
About this article
Cite this article
Fisher, M., Walker, A., Falqués, M. et al. Cost-effectiveness of linaclotide compared to antidepressants in the treatment of irritable bowel syndrome with constipation in Scotland. Eur J Health Econ 17, 1091–1100 (2016). https://doi.org/10.1007/s10198-015-0747-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10198-015-0747-0