Abstract
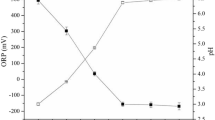
The overall objective of this study was to evaluate the use of controlled bio-acidification prior to land application as a decontamination process to remove heavy metals from sludge. The sulfur-oxidizing bacteria were naturally available in the sludge samples and were activated by providing sulfur and aeration at 28°C–30°C. Activation resulted in bio-acidification to pH 2 within 5–11 days. Successive inoculation of fresh sludges with 5% acidified samples reduced the acidification time to 2–3 days in most samples. Bio-acidification resulted in dissolving significant quantities of heavy metals from all sludge types tested. The maximum solubilization results were: 86%–97% for Ni; 48%–98% for Pb; 26%–71% for Cr; 18%–91% for Zn; 16%–90% for Cu; 7%–60% for Cd. Limited metal solubilization results were observed in the various control samples that accompanied the bio-acidified samples. The leaching results in the control samples were limited to 2%–19% for Ni, 0%–7% for Pb, 0%–5% for Cr, 0.3%–4% for Zn, 0.2%–4% for Cu and 0%–3% for Cd. The results confirmed that Ni and Pb were the easiest metals to dissolve from the various sludge types. On the other hand, the lowest solubilization results were observed for Cu and Cd, and moderate solubilization results were achieved for Cr. The bio-acidification process resulted in moderate gains in terms of improving the suitability of tested sludges for land application.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: April 19, 1999 / Accepted: November 4, 1999
Rights and permissions
About this article
Cite this article
Shanableh, A., Ginige, P. Acidic bioleaching of heavy metals from sewage sludge. J Mater Cycles Waste Manag 2, 43–50 (2000). https://doi.org/10.1007/s10163-999-0019-0
Issue Date:
DOI: https://doi.org/10.1007/s10163-999-0019-0