Abstract
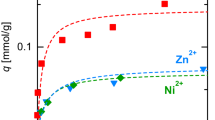
Virgin activated carbon (VAC) was chemically activated by hydrochloric acid and aqueous ammonia to obtain different surface functionalities and nanopore distributions. The N-tailored AC (NAC) acquired by alkali treatment significantly enhanced the cadmium sorption capacity, while the Cl-tailored AC obtained by acidic modification decreased the sorption capacity compared with that of VAC. With increasing ammonia concentration in the basic treatment, the NAC surface area was increased with declining pore volume and diameter, which increased the adsorption capacity for Cd(II) removal. However, increasing HCl concentration in the acidic modification decreased the surface area, with increasing pore volume and diameter, which decreased the cadmium adsorption capacity. The basic treatment created more alkaline-rich sites such as nitrogen-containing functional groups, on the carbon surface, thereby improving cadmium removal efficiency. However, the acidic treatment generated a stronger acidic chemical structure and much more the formation of the acyl- and alkyl chloride groups that greatly inhibited the Cd(II) sorption capacity.











Similar content being viewed by others
References
Gaya UI, Otene E, Abdullah AH (2015) Adsorption of aqueous Cd (II) and Pb(II) on activated carbon nanopores prepared by chemical activation of doum palm shell. Springerplus 4(1):1–18
Su Y et al. (2016) Direct synthesis of novel and reactive sulfide-modified nano iron through nanoparticle seeding for improved cadmium-contaminated water treatment. Sci Rep 6(24358):1–14
Su Y et al (2014) Simultaneous removal of cadmium and nitrate in aqueous media by nanoscale zerovalent iron (nZVI) and Au doped nZVI particles. Water Res 63:102–111
Boparai HK, Joseph M, O’Carroll DM (2013) Cadmium (Cd2+) removal by nano zerovalent iron: surface analysis, effects of solution chemistry and surface complexation modeling. Environ Sci Pollut Res 20(9):6210–6221
Park JH, Chon HT (2016) Characterization of cadmium biosorption by Exiguobacterium sp. isolated from farmland soil near Cu–Pb–Zn mine. Environ Sci Pollut Res 23(12):11814–11822
Chen C et al. (2016) Cadmium exposure and risk of lung cancer: a meta-analysis of cohort and case–control studies among general and occupational populations. J Expo Sci Environ Epidemiol 5(17976):1–8
Shi W et al (2016) Ocean acidification increases cadmium accumulation in marine bivalves: a potential threat to seafood safety. Sci Rep 6:20197
Yin Y et al (2015) Cadmium accumulation and apoplastic and symplastic transport in Boehmeria nivea (L.) Gaudich on cadmium-contaminated soil with the addition of EDTA or NTA. RSC Adv 5(59):47584–47591
Waalkes MP (2000) Cadmium carcinogenesis in review. J Inorg Biochem 79(1):241–244
Aoshima K (2016) Itai-itai disease: renal tubular osteomalacia induced by environmental exposure to cadmium—historical review and perspectives. Soil Sci Plant Nutr 62(4):319–326
Dianati-Tilaki R et al (2004) Study of cadmium removal from environmental water by biofilm covered granular activated carbon. Iran J Publ Health 33(4):43–52
Pratte-Santos R, Ribeiro A, Oliveira J (2016) Guidelines for recreation water quallity in Brazil, USA and Canada: enteric viruses as faecal pollution indicators. J Trop Dis 4(195):2
Munagamage T et al. (2016) Sensitivity of four cyanobacterial isolates from tropical freshwaters to environmentally realistic concentrations of Cr6+, Cd2+ and Zn2+. Bull Environ Contam Toxicol 96(6):816–821
Soto Hidalgo KT et al (2016) Photoelectrochemical solar cells prepared from nanoscale zerovalent iron used for aqueous Cd2+ removal. ACS Sustain Chem Eng 4(3):738–745
Yi W et al (2016) Removal of cadmium from contaminated lentinan by complexation coagulation method. J Food Saf Qual 7(1):383–388
Wang J et al. (2016) Cadmium removal from urban stormwater runoff via bioretention technology and effluent risk assessment for discharge to surface water. J Contam Hydrol 42(50):185–186
Gao J et al (2016) Green modification of outer selective P84 nanofiltration (NF) hollow fiber membranes for cadmium removal. J Membr Sci 499:361–369
Prapagdee S, Piyatiratitivorakul S, Petsom A (2016) Physico-chemical activation on rice husk biochar for enhancing of cadmium removal from aqueous solution. Asian J Water Environ Pollut 13(1):27–34
González M et al (2016) Capturing Cd (ii) and Pb (ii) from contaminated water sources by electro-deposition on hydrotalcite-like compounds. Phys Chem Chem Phys 18(3):1838–1845
Zhang D et al (2016) Preparation of calcium titanate based on the cotton template method and its simultaneous removal performance to heavy metals and organic pollutants in water. J Adv Oxid Technol 19(1):9–18
Sharaf El-Deen S, Zhang FS (2016) Immobilisation of TiO2-nanoparticles on sewage sludge and their adsorption for cadmium removal from aqueous solutions. J Exp Nanosci 11(4):239–258
Alguacil F et al (2016) Cadmium (II) transfer using (TiOAC) ionic liquid as carrier in a smart liquid membrane technology. Chem Eng Process 99:192–196
Bhattacharya K et al. (2015) Mesoporous magnetic secondary nanostructures as versatile adsorbent for efficient scavenging of heavy metals. Sci Rep 5(17072):1–9
Li B et al. (2014) Mercury nano-trap for effective and efficient removal of mercury (II) from aqueous solution. Nature communications 5(35537):1–7
Cao CY et al (2012) Low-cost synthesis of flowerlike α-Fe2O3 nanostructures for heavy metal ion removal: adsorption property and mechanism. Langmuir 28(9):4573–4579
Yang J et al (2014) High-content, well-dispersed γ-Fe2O3 nanoparticles encapsulated in macroporous silica with superior arsenic removal performance. Adv Funct Mater 24(10):1354–1363
Wang T et al (2013) Controllable synthesis of hierarchical porous Fe3O4 particles mediated by poly (diallyldimethylammonium chloride) and their application in arsenic removal. ACS Appl Mater Interfaces 5(23):12449–12459
Moreno-Castilla C et al (1998) Effects of non-oxidant and oxidant acid treatments on the surface properties of an activated carbon with very low ash content. Carbon 36(1):145–151
Chen JP, Wu S, Chong KH (2003) Surface modification of a granular activated carbon by citric acid for enhancement of copper adsorption. Carbon 41(10):1979–1986
Babel S, Kurniawan TA (2004) Cr (VI) removal from synthetic wastewater using coconut shell charcoal and commercial activated carbon modified with oxidizing agents and/or chitosan. Chemosphere 54(7):951–967
Park G et al (2002) Effect of two-step surface modification of activated carbon on the adsorption characteristics of metal ions in wastewater. I. Equilibrium and batch adsorptions. Carbon Sci 3:219–225
Moreno-Tovar R, Terrés E, Rangel-Mendez JR (2014) Oxidation and EDX elemental mapping characterization of an ordered mesoporous carbon: Pb(II) and Cd (II) removal. Appl Surf Sci 303:373–380
Mostafa M (1997) Adsorption of mercury, lead and cadmium ions on modified activated carbons. Adsorpt Sci Technol 15(8):551–557
Nadeem M et al (2006) Sorption of lead from aqueous solution by chemically modified carbon adsorbents. J Hazard Mater 138(3):604–613
Toles CA, Marshall WE, Johns MM (1999) Surface functional groups on acid-activated nutshell carbons. Carbon 37(8):1207–1214
Li F et al (2016) Preparation and characterization of biochars from Eichornia crassipes for cadmium removal in aqueous solutions. PLoS One 11(2):e0148132
Cui X et al (2016) Potential mechanisms of cadmium removal from aqueous solution by Canna indica derived biochar. Sci Total Environ 562:517–525
Mopoung R, Kengkhetkit N (2016) Lead and cadmium removal efficiency from aqueous solution by NaOH treated pineapple waste. Int J Appl Chem 12(1):23–35
Wang K et al (2016) Removal of cadmium (II) from aqueous solution by granular activated carbon supported magnesium hydroxide. J Taiwan Inst Chem Eng 61:287–291
Zahabi SR, Hosseini Ravandi SA, Allafchian A (2016) Removal of nickel and cadmium heavy metals using nanofiber membranes functionalized with (3-mercaptopropyl) trimethoxysilane (TMPTMS). J Water Health 14(4):630–639
Jafari SA, Jamali A (2016) Continuous cadmium removal from aqueous solutions by seaweed in a packed-bed column under consecutive sorption-desorption cycles. Korean J Chem Eng 33(4):1296–1304
Xu H et al. (2016) Controlled synthesized natroalunite microtubes applied for cadmium (II) and phosphate co–removal. J Hazard Mater 314(2016):249–259
Ahmad MF, Haydar S (2016) Evaluation of a newly developed biosorbent using packed bed column for possible application in the treatment of industrial effluents for removal of cadmium ions. J Taiwan Inst Chem Eng 62:122–131
Iqbal M et al (2016) Response surface methodology application in optimization of cadmium adsorption by shoe waste: a good option of waste mitigation by waste. Ecol Eng 88:265–275
Ince OK et al. (2016) Optimization of cadmium removal from water by hydroxyapatite using experimental design methodology. Anal Lett 8(2016):4293–4299
Tsechansky L, Graber E (2014) Methodological limitations to determining acidic groups at biochar surfaces via the Boehm titration. Carbon 66:730–733
Boehm H (1994) Some aspects of the surface chemistry of carbon blacks and other carbons. Carbon 32(5):759–769
Kim YS et al (2012) Effects of carbon dioxide and acidic carbon compounds on the analysis of Boehm titration curves. Carbon 50(4):1510–1516
Zhao H et al. (2013) Green “planting” nanostructured single crystal silver. Sci Rep 3(1511):1–7
Mekatel H et al (2012) Treatment of polluted aqueous solutions by Ni2+, Pb2+, Zn2+, Cr+6, Cd+2 and Co+2 ions by ion exchange process using faujasite zeolite. Proc Eng 33:52–57
Zabihi M, Ahmadpour A, Asl AH (2009) Removal of mercury from water by carbonaceous sorbents derived from walnut shell. J Hazard Mater 167(1):230–236
Shavandi MA et al (2012) Continuous metal and residual oil removal from palm oil mill effluent using natural zeolite-packed column. J Taiwan Inst Chem Eng 43(6):934–941
Dawson EA, Parkes GM, Branton P (2012) Synthesis of vegetable-based activated carbons with mixed micro-and mesoporosity for use in cigarette filters. Adsorpt Sci Technol 30(10):859–866
Gong H et al (2015) Surface modification of activated carbon for siloxane adsorption. Renew Energy 83:144–150
Shen W (2007) Surface chemical functional groups modification of porous carbon. Recent Pat Chem Eng 1(2007):27–40
Youngs C et al (1957) Preparation of long-chain fatty acid chlorides. J Am Oil Chem Soc 34(3):107–108
He C, Hu X (2011) Anionic dye adsorption on chemically modified ordered mesoporous carbons. Ind Eng Chem Res 50(24):14070–14083
Yang Z et al (2014) Nitrogen-doped, carbon-rich, highly photoluminescent carbon dots from ammonium citrate. Nanoscale 6(3):1890–1895
Joshi S, Pokharel BP (2014) Preparation and characterization of activated carbon from Lapsi (Choerospondias axillaris) seed stone by chemical activation with potassium hydroxide. J Inst Eng 9(1):79–88
Leyva-Ramos R et al (1997) Adsorption of cadmium (II) from aqueous solution onto activated carbon. Water Sci Technol 35(7):205–211
Prahas D et al (2008) Activated carbon from jackfruit peel waste by H 3 PO 4 chemical activation: pore structure and surface chemistry characterization. Chem Eng J 140(1):32–42
Bian Y et al (2015) Adsorption of cadmium ions from aqueous solutions by activated carbon with oxygen-containing functional groups. Chin J Chem Eng 23(10):1705–1711
Jiann K-T, Santschi PH, Presley BJ (2013) Relationships between geochemical parameters (pH, DOC, SPM, EDTA concentrations) and trace metal (Cd Co, Cu, Fe, Mn, Ni, Pb, Zn) concentrations in river waters of Texas (USA). Aquat Geochem 19(2):173–193
Basha S, Mason LG (2007) Focus on hazardous materials research. Nova Science Publ, New York
Tang WZ (2003) Physicochemical treatment of hazardous wastes. CRC Press, Boca Raton
Schmuki P (1997) Proceedings of the international symposium on pits and pores–formation, properties, and significance for advanced luminescent materials. The Electrochemical Society
Zhou J, Huang P, Lin R (1998) Sorption and desorption of Cu and Cd by macroalgae and microalgae. Environ Pollut 101(1):67–75
Mohan D et al (2007) Sorption of arsenic, cadmium, and lead by chars produced from fast pyrolysis of wood and bark during bio-oil production. J Colloid Interface Sci 310(1):57–73
Utomo HD, Hunter K (2006) Adsorption of divalent copper, zinc, cadmium and lead ions from aqueous solution by waste tea and coffee adsorbents. Environ Technol 27(1):25–32
Sheng PX et al (2004) Sorption of lead, copper, cadmium, zinc, and nickel by marine algal biomass: characterization of biosorptive capacity and investigation of mechanisms. J Colloid Interface Sci 275(1):131–141
Sharma AK, Lee BK (2016) Surfactant-aided sol-gel synthesis of TiO2–MgO nanocomposite and their photocatalytic azo dye degradation activity. J Compos Mater. doi:10.1177/0021998316636464
Singh J, Lee B-K (2016) Influence of nano-TiO 2 particles on the bioaccumulation of Cd in soybean plants (Glycine max): a possible mechanism for the removal of Cd from the contaminated soil. J Environ Manage 170:88–96
Febrianto J et al (2009) Equilibrium and kinetic studies in adsorption of heavy metals using biosorbent: a summary of recent studies. J Hazard Mater 162(2):616–645
Tien C, Ramarao BV (2014) Further examination of the relationship between the Langmuir kinetics and the Lagergren and the second-order rate models of batch adsorption. Sep Purif Technol 136:303–308
Ho Y-S, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34(5):451–465
Pentia E et al (2000) The influence of cadmium salt anion on the growth mechanism and on the physical properties of CdS thin films. J Optoelectron Adv Mater 2(5):593–601
Smith GW, Jacobson HW (1956) Characteristics of adsorption of complex metal-ammines and other complex ions of zinc, copper, cobalt, nickel and silver on silica gel. J Phys Chem 60(7):1008–1012
Liu J, Chen J, Huang L (2015) Heavy metal removal from MSS fly ash by thermal and chlorination treatments. Sci Rep 5(17270):1–14
Bandosz TJ (2006) Activated carbon surfaces in environmental remediation, vol 7. Academic Press, Cambridge
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Minh, T.D., Lee, BK. Effects of functionality and textural characteristics on the removal of Cd(II) by ammoniated and chlorinated nanoporous activated carbon. J Mater Cycles Waste Manag 19, 1022–1035 (2017). https://doi.org/10.1007/s10163-016-0570-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-016-0570-4