Abstract
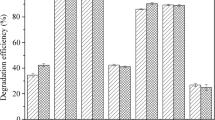
In the present work, we investigate the destruction efficiency of hexachlorobenzene (HCB) by milling with various reagents in a planetary ball mill under different milling conditions. Under the same conditions of mill rotary rate and charge ratio, the mixture of magnesium powder and aluminum oxide (Mg/Al2O3) was found best in promoting the destruction of HCB, which can be completed destroyed after 90 min grinding at a charge ratio of 20:1 (reagent/HCB, m/m), a ball mass/reagent mass ration of 30:1 and a mill rotation speed of 550 rpm. The ground samples were characterized and analyzed by X-ray fluorescence, gas chromatography (GC), X-ray diffraction and ion chromatography. The intermediate products, such as pentachlorobenzene, tetrachlorobenzene, trichlorobenzene isomers, dichlorobenzene and monochlorobenzene were detected by GC. Then the main dechlorination path way for HCB was proposed. With a series of verification experiments, the final degradation products of HCB were amorphous carbon and inorganic chlorine. Based on this study, Mg/Al2O3 has the potential to complete the innocuous treatment of chlorinated compounds.










Similar content being viewed by others
References
Stockholm Convention, http://www.pops.int/documents/pops/default.htm
Bailey RE (2001) Global hexanchlorobenzene emissions. Chemosphere
Randi AS, Cocca C, Carbone V, Nuñez M, Croci M, Gutiérrez A, Bergoc R, de Pisarev DLK (2006) Hexachlorobenzene is a tumor co-carcinogen and induces alterations in insulin-growth factors signaling pathway in the rat mammary gland. Toxicol Sci 89:83–92
Barber JL, Sweetman AJ, Van Wijk D, Jones KC (2005) Hexachlorobenzene in the global environment: emissions, levels, distribution, trends and processes. Sci Total Environ 349:1–44
(2007) National implementation plan for the Stockholm Convention on persistent organic pollutants, The People’s Republic of China. http://www.chem.unep.ch
Wikström E, Marklund S (2000) Secondary formation of chlorinated dibenzo-p-dioxins, dibenzofurans, biphenyls, benzenes, and phenols during MSW combustion. Environ Sci Technol 34:604–609
Guo X, Xiang D, Duan G, Mou P (2010) A review of mechanochemistry applications in waste management. Waste Manag 30:4–10
Nomura Y, Nakai S, Hosomi M (2005) Elucidation of degradation mechanism of dioxins during mechanochemical treatment. Environ Sci Technol 39:3799–3804
Zhang T, Huang J, Zhang W, Yu YF, Deng SB, Wang B, Yu G (2013) Coupling the dechlorination of aqueous 4-CP with the mechanochemical destruction of solid PCNB using Fe-Ni-SiO2. J Hazard Mater 250:175–180
Zhang K, Huang J, Yu G, Zhang Q, Deng S, Wang B (2013) Destruction of perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) by ball milling. Environ Sci Technol 47:6471–6477
Yu Y, Huang J, Zhang W, Zhang K, Deng S, Yu G (2013) Mechanochemical destruction of mirex co-ground with iron and quartz in a planetary ball mill. Chemosphere 90:1729–1735
Di Leo P, Pizzigallo MDR, Ancona V, Di Benedetto F, Mesto E, Schingaro E, Ventruti G (2013) Mechanochemical degradation of pentachlorophenol onto birnessite. J Hazard Mater 244:303–310
Zhang K, Huang J, Zhang W, Yu Y, Deng S, Yu G (2012) Mechanochemical degradation of tetrabromobisphenol A: performance, products and pathway. J Hazard Mater 243:278–285
Tongamp W, Zhang QW, Shoko M, Saito F (2009) Generation of hydrogen from polyvinyl chloride by milling and heating with CaO and Ni(OH)(2). J Hazard Mater 167:1002–1006
Aresta M, Dibenedetto A, Pastore T (2005) Mechanochemistry: an old technology with new applications to environmental issues. Decontamination of polychlorobiphenyl-contaminatecl soil by high-energy milling in the solid state with ternary hydrides. Environ Chem Green Chem Pollut Ecosyst 553–559
Mulas G, Loiselle S, Schiffini L, Cocco G (1997) The mechanochemical self-propagating reaction between hexachlorobenzene and calcium hydride. J Solid State Chem 129:263–270
Hall AK, Harrowfield JM, Hart RJ, McCormick PG (1996) Mechanochemical reaction of DDT with calcium oxide. Environ Sci Technol 30:3401–3407
Zhang QW, Saito F, Ikoma T, Tero-Kubota S (2001) Effects of quartz addition on the mechanochemical dechlorination of chlorobiphenyl by using CaO. Environ Sci Technol 35:4933–4935
Zhang QW, Matsumoto H, Saito F, Baron M (2002) Debromination of hexabromobenzene by its co-grinding with CaO. Chemosphere 48:787–793
Lu S, Huang J, Peng Z, Li X, Yan J (2012) Ball milling 2,4,6-trichlorophenol with calcium oxide: dechlorination experiment and mechanism considerations. Chem Eng J 195:62–68
Loiselle S, Branca M, Mulas G, Cocco G (1997) Selective mechanochemical dehalogenation of chlorobenzenes over calcium hydride. Environ Sci Technol 31:261–265
Zhang W, Wang H, Jun H, Yu M, Wang F, Zhou L, Yu G (2014) Acceleration and mechanistic studies of the mechanochemical dechlorination of HCB with iron powder and quartz sand. Chem Eng J 239:185–191
Delogu F, Deidda C, Mulas G et al (2004) A quantitative approach to mechanochemical processes [J]. J Material Sci 39(16–17):5121–5124
Zhang L, Zheng M, Liu W, Zhang B, Su G (2008) A method for decomposition of hexachlorobenzene by γ-alumina. J Hazard Mater 150:831–834
Simagina V, Likholobov V, Bergeret G, Gimenez MT, Renouprez A (2003) Catalytic hydrodechlorination of hexachlorobenzene on carbon supported Pd-Ni bimetallic catalysts. Appl Catal B Environ 40:293–304
Yamada S, Naito Y, Takada M, Nakai S, Hosomi M (2008) Photodegradation of hexachlorobenzene and theoretical prediction of its degradation pathways using quantum chemical calculation. Chemosphere 70:731–736
Gao XB, Wang W, Liu X (2009) Dechlorination reaction of hexachlorobenzene with calcium oxide at 300–400 °C. J Hazard Mater 169:279–284
Yang Y, Yu G, Deng S, Wang S, Xu Z, Huang J, Wang B (2012) Catalytic oxidation of hexachlorobenzene in simulated gas on V2O5-WO3/TiO2 catalyst. Chem Eng J 192:284–291
Xiao Y, Jiang J, Yang Y, Gao G (2011) Base-catalyzed destruction of hexachlorobenzene with zero-valent iron. Chem Eng J 173:415–421
Tanaka Y, Zhang Q, Saito F (2003) Mechanochemical dechlorination of trichlorobenzene on oxide surfaces. J Phys Chem B 107:11091–11097
Acknowledgments
Support for this research by the Open Fund of Shanghai Cooperative Centre for WEEE Recycling (B50ZS120003) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ren, Y., Kang, S. & Zhu, J. Mechanochemical degradation of hexachlorobenzene using Mg/Al2O3 as additive. J Mater Cycles Waste Manag 17, 607–615 (2015). https://doi.org/10.1007/s10163-015-0398-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-015-0398-3