Abstract
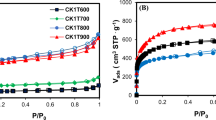
Polychlorinated biphenyl (PCB) residues from the sodium dispersion (SD) process were employed as the raw materials for the production of activated carbon using KOH activation. The pore properties, such as the specific surface area and pore size distribution, were characterized using the Barrett–Joyner–Halenda method and the Horvath–Kawazoe method based on the N2 adsorption isotherm at 77 K. The activated carbon produced showed similar adsorption capacities and specific surface areas to the commercially available product. The effects of the activation conditions on the porosity of the activated carbon produced were studied. The most significant factor affecting the specific surface proved to be the activation temperature. The activated carbon produced from PCB residues from the high-temperature (423–443 K) SD process had a binary pore size distribution well developed in the 4 nm region and in the micropore region. The pore structure of the carbon produced from PCB residues from the low-temperature (333–393 K) SD process had a wide range of micropores and mesopores.
Similar content being viewed by others
References
A Ariizumi T Otsuka M Kamiyama M Hosomi (1997) ArticleTitleDechlorination and decomposition behavior of PCBs by the sodium dispersion process (in Japanese) J Environ Chem Jpn 7 IssueID4 793–799 Occurrence Handle1:CAS:528:DyaK1cXhtFOjsLk%3D
Y-R Lin H Teng (2002) ArticleTitleMesoporous carbons from waste tire char and their application in wastewater discoloration Micropor Mesopor Mater 54 167–174 Occurrence Handle1:CAS:528:DC%2BD38XktlKmuro%3D
S Chitra G Sekaran G Chandrakasan (1996) ArticleTitleAdsorption of a mutant strain of Pseudomonas pictorum on rice bran-based activated carbon J Hazard Mater 48 239–250 Occurrence Handle10.1016/0304-3894(95)00158-1 Occurrence Handle1:CAS:528:DyaK28XitFCgsL0%3D
AR Khan TA Al-Bahri A Al-Haddad (1997) ArticleTitleAdsorption of phenol-based organic pollutants on activated carbon from multi-component dilute aqueous solutions Water Res 31 2102–2112 Occurrence Handle10.1016/S0043-1354(97)00043-2 Occurrence Handle1:CAS:528:DyaK2sXktlSrur8%3D
J Matos J Laine JM Herrmann (2001) ArticleTitleEffect of the type of activated carbons on the photocatalytic degradation of aqueous organic pollutants by UV-irradiated titania J Catal 200 10–20 Occurrence Handle10.1006/jcat.2001.3191 Occurrence Handle1:CAS:528:DC%2BD3MXjtVGlsrg%3D
A Namane A Mekarzia K Benrachedi N Belhaneche-Bensemra A Hellal (2005) ArticleTitleDetermination of the adsorption capacity of activated carbon made from coffee grounds by chemical activation with ZnCl2 and H3PO4 J Hazard Mater 119 189–194 Occurrence Handle10.1016/j.jhazmat.2004.12.006 Occurrence Handle1:CAS:528:DC%2BD2MXhvFymtbY%3D
GN Manju C Raji TS Anirudhan (1998) ArticleTitleEvaluation of coconut husk carbon for the removal of arsenic from water Water Res 32 3062–3070 Occurrence Handle10.1016/S0043-1354(98)00068-2 Occurrence Handle1:CAS:528:DyaK1cXmtVGms7g%3D
Chemical engineering handbook, 6th edn (in Japanese) (1999) Maruzen, The Society of Chemical Engineers, Japan, pp 694
VK Dobruskin (2001) ArticleTitleCorrelation between two- and three-dimensional condensations in cylindrical mesopores of active carbons Carbon 39 583–591 Occurrence Handle10.1016/S0008-6223(00)00172-X Occurrence Handle1:CAS:528:DC%2BD3MXhslOgt7s%3D
H Ma J He DG Evans X Duan (2004) ArticleTitleImmobilization of lipase in a mesoporous reactor based on MCM-41 J Mol Catal 30 209–217 Occurrence Handle10.1016/j.molcatb.2004.04.007 Occurrence Handle1:CAS:528:DC%2BD2cXmt1Kms7o%3D
AK Khattak M Afzal M Saleem G Yasmeen R Ahmad (2000) ArticleTitleSurface modification of alumina by metal doping Colloids Surf A: Physicochem Eng Aspects 162 99–106 Occurrence Handle10.1016/S0927-7757(99)00218-6 Occurrence Handle1:CAS:528:DC%2BD3cXhtFeltLg%3D
F Moro H Böhni (2002) ArticleTitleInk-bottle effect in mercury intrusion porosimetry of cement-based material J Colloid Interface Sci 246 135–149 Occurrence Handle10.1006/jcis.2001.7962 Occurrence Handle1:CAS:528:DC%2BD38XitVOgtA%3D%3D
SJ Gregg KSW Sing (1976) ArticleTitleAdsorption of gases on porous solids Surf Colloid Sci 6 183–186
VM Gun'ko DD Do (2001) ArticleTitleCharacterisation of pore structure of carbon adsorbents using regularisation procedure Colloids Surf A: Physicochem Eng Aspects 193 71–83 Occurrence Handle10.1016/S0927-7757(01)00685-9
JJ Mahle (2002) ArticleTitleAn adsorption equilibrium model for Type 5 isotherms Carbon 40 2753–2759 Occurrence Handle10.1016/S0008-6223(02)00201-4 Occurrence Handle1:CAS:528:DC%2BD38Xot1Wmsrc%3D
Do HD Do DD (2002) ArticleTitleAdsorption of mixtures containing subcritical fluids on activated carbon AIChE J 48 2213–2229 Occurrence Handle10.1002/aic.690481012
Nishimura M (1979) Pore structure of active carbons and adsorption phenomena of hydrocarbons (in Japanese). J Chem Soc Jpn Volume 1979, No. 1:10–15
AJ Juhola EO Wiig (1949) ArticleTitlePore structure in activated charcoal. I. Determination of micropore size distribution J Am Chem 71 2069–2077 Occurrence Handle10.1021/ja01174a050 Occurrence Handle1:CAS:528:DyaG3cXhs1Cj
T Zimny G Finqueneisel L Cossarutto JV Weber (2005) ArticleTitleWater vapor adsorption on activated carbon preadsorbed with naphthalene J Colloid Interface Sci 285 56–60 Occurrence Handle10.1016/j.jcis.2004.11.029 Occurrence Handle1:CAS:528:DC%2BD2MXis1eku7o%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, H., Kobayashi, N., Sakata, M. et al. Production and characterization of KOH-activated carbon derived from polychlorinated biphenyl residue generated by the sodium dispersion process. J Mater Cycles Waste Manag 9, 182–187 (2007). https://doi.org/10.1007/s10163-007-0175-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-007-0175-z