Abstract
Purpose
Cisplatin is a low-cost clinical anti-tumor drug widely used to treat solid tumors. However, its use could damage cochlear hair cells, leading to irreversible hearing loss. Currently, there appears one drug approved in clinic only used for reducing ototoxicity associated with cisplatin in pediatric patients, which needs to further explore other candidate drugs.
Methods
Here, by screening 1967 FDA-approved drugs to protect cochlear hair cell line (HEI-OC1) from cisplatin damage, we found that Tedizolid Phosphate (Ted), a drug indicated for the treatment of acute infections, had the best protective effect. Further, we evaluated the protective effect of Ted against ototoxicity in mouse cochlear explants, zebrafish, and adult mice. The mechanism of action of Ted was further explored using RNA sequencing analysis and verified. Meanwhile, we also observed the effect of Ted on the anti-tumor effect of cisplatin.
Results
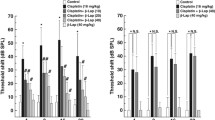
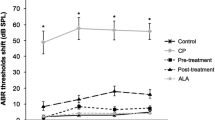
Ted had a strong protective effect on hair cell (HC) loss induced by cisplatin in zebrafish and mouse cochlear explants. In addition, when administered systemically, it protected mice from cisplatin-induced hearing loss. Moreover, antitumor studies showed that Ted had no effect on the antitumor activity of cisplatin both in vitro and in vivo. RNA sequencing analysis showed that the otoprotective effect of Ted was mainly achieved by inhibiting phosphorylation of ERK. Consistently, ERK activator aggravated the damage of cisplatin to HCs.
Conclusion
Collectively, these results showed that FDA-approved Ted protected HCs from cisplatin-induced HC loss by inhibiting ERK phosphorylation, indicating its potential as a candidate for preventing cisplatin ototoxicity in clinical settings.







Similar content being viewed by others
Availability of Data and Material
The data sets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- Ted:
-
Tedizolid Phosphate
- FDA:
-
Food and Drug Administration
- HCs:
-
Hair cells
- OHCs:
-
Outer hair cells
- ROS:
-
Reactive oxygen species
- CIHL:
-
Cisplatin-induced hearing loss
- MET:
-
Mechanoelectrical transducer
- BBB:
-
Blood-brain barrier
- CCK8:
-
Cell Counting Kit-8
References
Ramkumar V, Mukherjea D, Dhukhwa A, Rybak LP (2021) Oxidative stress and inflammation caused by cisplatin ototoxicity. Antioxidants 10(12):1919
Small W, Bacon MA, Bajaj A, Chuang LT, Gaffney DK (2017) Cervical cancer: a global health crisis. Obstet Gynecol Surv 72(11):654–655
He YZ, Zheng ZW, Liu C, Li W, Zhao LP, Nie GH, Li HW (2022) Inhibiting DNA methylation alleviates cisplatin- induced hearing loss by decreasing oxidative stress-induced mitochondria-dependent apoptosis via the LRP1-PI3K/AKT pathway. Acta Pharmaceutica Sinica B 12(3):1305–1321
Karasawa T, Steyger PS (2015) An integrated view of cisplatin-induced nephrotoxicity and ototoxicity. Toxicol Lett 237(3):219–227
Paken J, Govender CD, Pillay M, Sewram V (2022) Cisplatin-associated ototoxicity: perspectives from a single institution cervical cancer cohort and implications for developing a locally responsive monitoring programme in a public healthcare setting. BMC Health Serv Res 22(1):1–14
Tang Q, Wang X, Jin H, Mi Y, Zou Z (2021) Cisplatin-induced ototoxicity: updates on molecular mechanisms and otoprotective strategies. Eur J Pharm Biopharm 163(7):60–71
Breglio AM, Rusheen AE, Shide ED, Fernandez KA, Spielbauer KK, McLachlin KM, Hall MD, Amable L, Cunningham LL (2017) Cisplatin is retained in the cochlea indefinitely following chemotherapy. Nat Commun 8(1):1654
Wang WL, Chen EF, Ding XR, Lu PH, Chen JW, Ma PW, Lu LJ (2022) N-acetylcysteine protect inner hair cells from cisplatin by alleviated celluar oxidative stress and apoptosis. Toxicol In Vitro 81:105354
Gentilin E, Simoni E, Candito M, Cazzador D, Astolfi L (2019) Cisplatin-induced ototoxicity: updates on molecular targets. Trends Mol Med 25(12):1123–1132
Zallocchi M, Hati S, Xu Z, Hausman W, Zuo J (2021) Characterization of quinoxaline derivatives for protection against iatrogenic-induced hearing loss. JCI Insight 6(5):e141561
Rybak LP (2007) Mechanisms of cisplatin ototoxicity and progress in otoprotection. Curr Opin Otolaryngol Head Neck Surg 15(5):364–369
Yu D, Gu J, Chen Y, Kang W, Wu H (2020) Current strategies to combat cisplatin-induced ototoxicity. Front Pharmacol 11:999
He FA, Huang XT, Wei GK, Lin XR, Zhang WJ, Zhuang W, He WH, Zhan T, Hu H, Yang HD (2022) Regulation of ACSL4-catalyzed lipid peroxidation process resists cisplatin ototoxicity. Oxid Med Cell Longev 3080263
Mukherjea D, Dhukhwa A, Sapra A, Bhandari P, Rybak L (2020) Strategies to reduce the risk of platinum containing antineoplastic drug-induced ototoxicity. Expert Opin Drug Metab Toxicol 16(10):965–982: PMID:32757852
Bhatta P, Dhukhwa A, Sheehan K, Al Aameri RFH, Borse V, Ghosh S, Sheth S, Mamillapalli C, Rybak L, Ramkumar V et al (2019) Capsaicin protects against cisplatin ototoxicity by changing the STAT3/STAT1 ratio and activating cannabinoid (CB2) receptors in the cochlea. Sci Rep 9(1):4131
Kim HJ, Oh GS, Lee JH, Lyu AR, Ji HM, Lee SH, Song J, Park SJ, You YO, Sul JD (2011) Cisplatin ototoxicity involves cytokines and STAT6 signaling network. Cell Res 21(6):13
So H, Kim H, Kim Y, Kim E, Pae HO, Chung HT, Kim HJ, Kwon KB, Lee KM, Lee HY et al (2008) Evidence that cisplatin-induced auditory damage is attenuated by downregulation of pro-inflammatory cytokines via Nrf2/HO-1. Jaro-Journal of the Association for Research in Otolaryngology 9(3):290–306
Volker N, Kwang P, Rahul J, Arwa K, Ryan AF (2017) An antioxidant screen identifies candidates for protection of cochlear hair cells from gentamicin toxicity. Front Cell Neurosci 11:242
Tian CJ, Kim YJ, Kim SW, Lim HJ, Kim YS, Choung YH (2013) A combination of cilostazol and ginkgo biloba extract protects against cisplatin-induced cochleo-vestibular dysfunction by inhibiting the mitochondrial apoptotic and ERK pathways. J Micro/Nanolithogr MEMS MOEMS 4(2):e509
Tan XD, Zhou YJ, Agarwal A, Lim M, Xu YY, Zhu YM, O’Brien J, Tran E, Zheng J, Gius D et al (2020) Systemic application of honokiol prevents cisplatin ototoxicity without compromising its antitumor effect. Am J Cancer Res 10(12):4416
Rolland V, Meyer F, Guitton MJ, Bussières R, Philippon D, Bairati I, Leclerc M, Côté M (2019) A randomized controlled trial to test the efficacy of trans-tympanic injections of a sodium thiosulfate gel to prevent cisplatin-induced ototoxicity in patients with head and neck cancer. J Otolaryngol Head Neck Surg 48(1):4
Sarafraz Z, Ahmadi A, Daneshi A (2018) Transtympanic injections of N-acetylcysteine and dexamethasone for prevention of cisplatin-induced ototoxicity: double blind randomized clinical trial. International Tinnitus Journal 22(1):40
Freyer DR, Chen L, Krailo MD, Knight K, Villaluna D, Bliss B, Pollock BH, Ramdas J, Lange B, Van Hoff D (2017) Effects of sodium thiosulfate versus observation on development of cisplatin-induced hearing loss in children with cancer (ACCL0431): a multicentre, randomised, controlled, open-label, phase 3 trial. Lancet Oncology 18(1):63
Brock PR, Maibach R, Childs M, Rajput K, Neuwelt EA (2018) Sodium thiosulfate for protection from cisplatin-induced hearing loss. N Engl J Med 378(25):191
Kros CJ, Steyger PS (2019) Aminoglycoside- and cisplatin-induced ototoxicity: mechanisms and otoprotective strategies. Cold Spring Harbor Laboratory Press 9(11):a033548
Abada P (2011) SB H: Regulation of cisplatin cytotoxicity by cu influx transporters. Metal Based Drugs 2010:317581
Kros CJ, Steyger PS (2019) Aminoglycoside- and cisplatin-induced ototoxicity: mechanisms and otoprotective strategies. Cold Spring Harb Perspect Med 9(11):a033548
Ciarimboli G, Deuster D, Knief A, Sperling M, Holtkamp M, Edemir B, Pavenstädt H, Lanvers-Kaminsky C, Zehnhoff-Dinnesen AA, Schinkel AH et al (2010) Organic cation transporter 2 mediates cisplatin-induced oto- and nephrotoxicity and is a target for protective interventions. Am J Pathol 176(3):1169–1180
Ingersoll MA, Malloy EA, Caster LE, Holland EM, Teitz T (2020) BRAF inhibition protects against hearing loss in mice. Sci Adv 6(49):eabd0561
Kitcher SR, Kirkwood NK, Camci ED, Wu P, Gibson RM, Redila VA, Simon JA, Rubel EW, Raible DW, Richardson GP et al (2019) ORC-13661 protects sensory hair cells from aminoglycoside and cisplatin ototoxicity. Jci Insight 4(15):e126764
Li M, Liu JW, Liu D, Duan XC, Zhang QC, Wang DW, Zheng QY, Bai XH, Lu ZM (2021) Naringin attenuates cisplatin- and aminoglycoside-induced hair cell injury in the zebrafish lateral line via multiple pathways. J Cell Mol Med 25(2):975–989
Rocha-Sanchez SM, Fuson O, Tarang S, Goodman L, Pyakurel U, Liu HZ, He DZ, Zallocchi M (2019) Quinoxaline protects zebrafish lateral line hair cells from cisplatin and aminoglycosides damage (vol 8, 15119, 2018). Sci Rep 8(1):15119
Fernandes VT, Lin VYW (2014) Development of an ototoxicity model in the adult CBA/CaJ mouse and determination of a golden window of corticosteroid intervention for otoprotection. Journal of Otolaryngology-Head & Neck Surgery 43(1):12
Garcia-Alcantara F, Murillo-Cuesta S, Pulido S, Bermudez-Munoz JM, Martinez-Vega R, Milo M, Varela-Nieto I, Rivera T (2018) The expression of oxidative stress response genes is modulated by a combination of resveratrol and N-acetylcysteine to ameliorate ototoxicity in the rat cochlea. Hear Res 358:10–21
Teitz T, Fang J, Goktug AN, Bonga JD, Diao SY, Hazlitt RA, Iconaru L, Morfouace M, Currier D, Zhou YM et al (2018) CDK2 inhibitors as candidate therapeutics for cisplatin-and noise-induced hearing loss. J Exp Med 215(4):1187–1203
Dhillon S (2023) Sodium thiosulfate: pediatric first approval. Pediatr Drugs 25(2):239–244
Kisgen JJ, Mansour H, Unger NR, Childs LM (2014) Tedizolid: a new oxazolidinone antimicrobial. Am J Health Syst Pharm 71(8):621–633
Mikamo H, Nagashima M, Kusachi S, Fujimi S, Oshima N, De Anda C, Takase A (2022) Efficacy and safety of tedizolid for the treatment of ventilated gram-positive hospital-acquired or ventilator-associated bacterial pneumonia in Japanese patients: results from a subgroup analysis of a phase 3, randomized, double-blind study comparing tedizolid and linezolid. J Infect Chemother 28(9):1235–1241
Nyberg S, Abbott NJ, Shi X, Steyger PS, Dabdoub A (2019) Delivery of therapeutics to the inner ear: The challenge of the blood-labyrinth barrier. ence Transl Med 11(482):eaao0935
Gu LQ, Ma MN, Zhang Y, Zhang LJ, Zhang S, Huang MC, Zhang MJ, Xin YF, Zheng GL, Chen SH (2019) Comparative pharmacokinetics of tedizolid in rat plasma and cerebrospinal fluid. Regul Toxicol Pharmacol 107:104420
Wenzler E, Adeel A, Wu T, Jurkovic M, Walder J, Ramasra E, Campion M, Cerny J, Theodoropoulos NM (2021) Inadequate cerebrospinal fluid concentrations of available salvage agents further impedes the optimal treatment of multidrug-resistant Enterococcus faecium meningitis and bacteremia. Infectious disease reports 13(3):843–854
Chen YW, Lear TB, Evankovich JW, Larsen MB, Lin B, Alfaras I, Kennerdell JR, Salminen L, Camarco DP, Lockwood KC et al (2021) A high-throughput screen for TMPRSS2 expression identifies FDA-approved compounds that can limit SARS-CoV-2 entry. Nat Commun 12(1):3907
Jin WJ, Yu JM, Su Y, Lin HC, Liu TF, Chen J, Ge C, Zhao FY, Geng Q, Mao L et al (2023) Drug repurposing flubendazole to suppress tumorigenicity via PCSK9-dependent Inhibition and potentiate lenvatinib therapy for hepatocellular carcinoma. Int J Biol Sci 19(7):2270–2288
Shahab M, Rosati R, Meyers D, Sheilds J, Jamesdaniel S (2021) Cisplatin-induced hair cell loss in zebrafish neuromasts is accompanied by protein nitration and Lmo4 degradation. Toxicol Appl Pharmacol 410(1–2):115342
Drusano GL, Liu W, Kulawy R, Louie A (2011) Impact of granulocytes on the antimicrobial effect of tedizolid in a mouse thigh infection model▿. Antimicrob Agents Chemother 55(11):5300–5305
He Y, Zheng Z, Liu C, Li W, Zhao L, Nie G, Li H (2022) Inhibiting DNA methylation alleviates cisplatininduced hearing loss by decreasing oxidative stress-induced mitochondria-dependent apoptosis via the LRP1-PI3K/AKT pathway. Acta Pharmaceutica Sinica B 003:012
Yu H, Lin Q, Wang Y, He Y, Fu S, Jiang H, Yu Y, Sun S, Chen Y, Shou J et al (2013) Inhibition of H3K9 methyltransferases G9a/GLP prevents ototoxicity and ongoing hair cell death. Cell Death Dis 4(2):e506
Drévillon L, Tanguy G, Hinzpeter A, Arous N, de Becdelièvre A, Aissat A, Tarze A, Goossens M, Fanen P (2011) COMMD1-mediated ubiquitination regulates CFTR trafficking. PLoS ONE 6(3):e18334
Liu JY, Zheng XJ, Li W, Ren LW, Li S, Yang YH, Yang H, Ge BB, Du GH, Shi JY et al (2022) Anti-tumor effects of Skp2 inhibitor AAA-237 on NSCLC by arresting cell cycle at G0/G1 phase and inducing senescence. Pharmacol Res 181:106259
Teitz T, Goktug AN, Chen T, Zuo J (2016) Development of cell-based high-throughput chemical screens for protection against cisplatin-induced ototoxicity. Methods Mol Biol 1427:419–430
Landegger LD, Dilwali S, Stankovic KM (2017) Neonatal murine cochlear explant technique as an in vitro screening tool in hearing research. Jove-Journal of Visualized Experiments 124:55704
Masuda M, Pak K, Chavez E, Ryan AF (2012) TFE2 and GATA3 enhance induction of POU4F3 and myosin VIIa positive cells in nonsensory cochlear epithelium by ATOH1. Dev Biol 372(1):68–80
Lim HW, Pak K, Ryan AF, Kurabi A (2018) Screening mammalian cochlear hair cells to identify critical processes in aminoglycoside-mediated damage. Front Cell Neurosci 12:179
Holmgren M, Sheets L (2021) Using the zebrafish lateral line to understand the roles of mitochondria in sensorineural hearing loss. Frontiers in Cell and Developmental Biology 8:628712
Dehne N, Lautermann J, Petrat F, Rauen U, de Groot H (2001) Cisplatin ototoxicity: involvement of iron and enhanced formation of superoxide anion radicals. Toxicol Appl Pharmacol 174(1):27–34
Mohan S, Smyth BJ, Namin A, Phillips G, Gratton MA (2014) Targeted amelioration of cisplatin-induced ototoxicity in guinea pigs. Otolaryngology-Head and Neck Surgery 151(5):836–839
Langer T, Zehnhoff-Dinnesen AA, Radtke S, Meitert J, Zolk O (2013) Understanding platinum-induced ototoxicity. Trends Pharmacol Sci 34(8):458–469
Kim HJ, Lee JH, Kim SJ, Oh GS, Moon HD, Kwon KB, Park C, Park BH, Lee HK, Chung SY et al (2010) Roles of NADPH oxidases in cisplatin-induced reactive oxygen species generation and ototoxicity. J Neurosci 30(11):3933–3946
Kim J, Cho HJ, Sagong B, Kim SJ, Lee JT, So HS, Lee IK, Kim UK, Lee KY, Choo YS (2014) Alpha-lipoic acid protects against cisplatin-induced ototoxicity via the regulation of MAPKs and proinflammatory cytokines. Biochem Biophys Res Commun 449(2):183–189
So H, Kim HJ, Lee JH, Park C, Kim Y, Kim E, Kim JK, Yun KJ, Lee LHY, Al E (2007) isplatin cytotoxicity of auditory cells requires secretions of proinflammatory cytokines via activation of ERK and NF- κ B. JARO-J Assoc Res Otolaryngol 8(3):338–55
Zheng S, Zheng S, Liu C, Liu C, Tang D, Tang D, Zheng Z, Zheng Z, Yan R, Yan R (2022) The protective effect of rutin against the cisplatin-induced cochlear damage in vitro. Neurotoxicology 90:102–111
Lee JS, Kang SU, Hwang HS, Pyun JH, Choung YH, Kim CH (2010) Epicatechin protects the auditory organ by attenuating cisplatin-induced ototoxicity through inhibition of ERK. Toxicol Lett 199(3):308–316
Wang D, Shi S, Ren T, Zhang Y, Guo P, Wang J, Wang W (2021) U0126 pretreatment inhibits cisplatin-induced apoptosis and autophagy in HEI-OC1 cells and cochlear hair cells. Toxicol Appl Pharmacol 415:115447
Kao C, Chao A, Tsai CL, Chuang WC, Huang WP, Chen GC, Lin CY, Wang TH, Wang HS, Lai CH (2014) Bortezomib enhances cancer cell death by blocking the autophagic flux through stimulating ERK phosphorylation. Cell Death Dis 5(11):e1510
Hsin MC, Hsiao YH, Chen PN, Lin CW, Wang PH, Yang SF, Lee CY (2022) Dihydromyricetin inhibited migration and invasion by reducing S100A4 expression through ERK1/2/beta-catenin pathway in human cervical cancer cell lines. Int J Mol Sci 23(23):15106
Astolfi L, Ghiselli S, Guaran V, Chicca M, Simoni E, Olivetto E, Lelli G, Martini A (2013) Correlation of adverse effects of cisplatin administration in patients affected by solid tumours: a retrospective evaluation. Oncol Rep 29(4):1285–1292
Rybak LP, Dhukhwa A, Mukherjea D, Ramkumar V (2019) Local drug delivery for prevention of hearing loss. Front Cell Neurosci 13:300
Hazlitt RA, Min JK, Zuo J (2018) Progress in the development of preventative drugs for cisplatin-induced hearing loss. J Med Chem 61(13):5512–5524
Gu JY, Tong L, Lin X, Chen YM, Wu H, Wang XL, Yu DH (2022) The disruption and hyperpermeability of blood-labyrinth barrier mediates cisplatin-induced ototoxicity. Toxicol Lett 354:56–64
Wang XL, Zhou YY, Wang DL, Wang Y, Zhou ZY, Ma XL, Liu XF, Dong YD (2023) Cisplatin-induced ototoxicity: from signaling network to therapeutic targets. Biomed Pharmacother 157:114045
Fu D, Wu J, Lai J, Liu Y, Zhou L, Chen L, Zhang Q (2020) T cell recruitment triggered by optimal dose platinum compounds contributes to the therapeutic efficacy of sequential PD-1 blockade in a mouse model of colon cancer. Am J Cancer Res 10(2):473–490
Kawaguchi Y, Ohshio Y, Watanabe A, Shiratori T, Okamoto K, Ueda K, Kataoka Y, Suzuki T, Hanaoka J (2023) Depletion of tumor-associated macrophages inhibits lung cancer growth and enhances the antitumor effect of cisplatin. Cancer Sci 114(3):750–763
Schacht J, Talaska AE, Rybak LP (2012) Cisplatin and aminoglycoside antibiotics: hearing loss and its prevention. Anatomical Record-advances in Integrative Anatomy & Evolutionary Biology 295(11):1837–1850
Liu Y, Wei M, Mao X, Chen TS, Lin P, Wang W (2021) Key signaling pathways regulate the development and survival of auditory hair cells. Neural Plast 5522717
Roux PP, Blenis J (2004) ERK and p38 MAPK-activated protein kinases: a family of protein kinases with diverse biological functions. Microbiol Mol Biol Rev 68(2):320
Choi BM, Chen XY, Gao SS, Zhu R, Kim BR (2011) Anti-apoptotic effect of phloretin on cisplatin-induced apoptosis in HEI-OC1 auditory cells. Pharmacological Reports Pr 63(3):708–716
Hall MD, Okabe M, Shen DW, Liang XJ, Gottesman MM (2008) The role of cellular accumulation in determining sensitivity to platinum-based chemotherapy. Annu Rev Pharmacol Toxicol 48(1):495–535
Shen DW, Pouliot LM, Hall MD, Gottesman MM (2012) Cisplatin resistance: a cellular self-defense mechanism resulting from multiple epigenetic and genetic changes. Pharmacol Rev 64(3):706–721
Thomas AJ, Hailey DW, Stawicki TM, Wu P, Coffin AB, Rubel EW, Raible DW, Simon JA, Ou HC (2013) Functional mechanotransduction is required for cisplatin-induced hair cell death in the zebrafish lateral line. J Neurosci 33(10):4405–4414
Funding
This research was supported by grants from the National Natural Science Foundation of China (no. 82271175, 82192863, 82171162, 81900937, 82201296, 82201294, 82001204), the Natural Science Foundation from Shandong Province (no. ZR2021QH269 and ZR2022MC216), the Natural Science Foundation from Henan Province (no. 232300420259), and the Scientific and Technological Innovation Plan Project of Medical System Staff of Shandong Province (no. SDYWZGKCJHLH2023096) and Shandong Province medical health science and technology project (no. 202307010965).
Author information
Authors and Affiliations
Contributions
Xiaolong Fu, Daqing Sun, Lei Xu, and Wen Li designed the research. Zhiwei Yao, Yu Xiao, Wen Li, Hailong Tu, Shuhui Kong, Ruifeng Qiao, Lushun Ma, and Siwei Guo conducted experiments and analyzed data. Song Wang and Miao Chang contributed to writing the manuscript. Zhiwei Yao, Xiaolong Fu, Wen Li, and Daqing Sun wrote the manuscript and interpreted the data. Xiaoxu Zhao and Yuan Zhang revised the article.
Corresponding authors
Ethics declarations
Ethics Approval and Consent to Participate
All experimental procedures involving animals were approved by the Tianjin Medical University General Hospital Experimental Animal Administrative Committee and were in accordance with animal welfare principles.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yao, Z., Xiao, Y., Li, W. et al. FDA-Approved Tedizolid Phosphate Prevents Cisplatin-Induced Hearing Loss Without Decreasing Its Anti-tumor Effect. JARO (2024). https://doi.org/10.1007/s10162-024-00945-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10162-024-00945-2