Abstract
Background
Cisplatin-induced injury of renal proximal tubular cells results basically from increased apoptosis via mitochondrial damage, and is mitigated by appropriate enhancement of autophagy. Peroxisome proliferator-activated receptor-delta (PPAR-δ) reportedly protects against not only mitochondrial damages but also enhances autophagy. Thus, PPAR-δ may protect against cisplatin-induced kidney injury.
Methods
We examined the protective effects of PPAR-δ activation on cisplatin-induced cellular injury and their detailed mechanisms in a murine renal proximal tubular (mProx) cell line using GW0742, an authentic PPAR-δ activator. Cisplatin-induced cell damages were evaluated by TUNEL assay and immunoblot analyses for p53, 14-3-3, Bax, Bcl2, cytochrome C, and activated caspases. Autophagy status was examined by immunoblot analyses for p62 and LC3.
Results
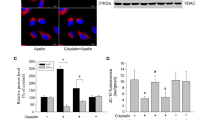
GW0742 suppressed cisplatin-induced apoptosis of mProx cells by reducing the activation of caspase-3 via attenuating the phosphorylation of p53 and 14-3-3, mitochondrial Bax accumulation, cytochrome C release from mitochondria to the cytosol and ensuing cytosolic caspase-9 activation. In contrast, GW0742 did not diminish cisplatin-enhanced activation of caspases-8 or -12 as extrinsic or endothelium reticulum apoptotic pathways, respectively. The inhibitory effect of GW0742 on cisplatin-induced caspase-3 activation was significantly diminished by silencing of the PPAR-δ gene expression. GW0742 itself had no influence on starvation-stimulated or cisplatin-induced autophagy in mProx cells, suggesting that the protective effects were not mediated by autophagy modification.
Conclusion
Our results indicate that GW0742 may serve as a candidate agent to mitigate cisplatin nephrotoxicity via inhibiting the mitochondrial apoptotic pathway considerably depending on PPAR-δ, without modulating autophagy.




Similar content being viewed by others
References
Ozkok A, Edelstein CL. Pathophysiology of cisplatin-induced acute kidney injury. Biomed Res Int. 2014;2014:967826. https://doi.org/10.1155/2014/967826.
Oh GS, Kim HJ, Shen A, Lee SB, Khadka D, Pandit A, et al. Cisplatin-induced kidney dysfunction and perspectives on improving treatment strategies. Electrolyte Blood Press. 2014;12(2):55–65. https://doi.org/10.5049/EBP.2014.12.2.55.
Park MS, De Leon M, Devarajan P. Cisplatin induces apoptosis in LLC-PK1 cells via activation of mitochondrial pathways. J Am Soc Nephrol. 2002;13(4):858–65.
Nagothu KK, Bhatt R, Kaushal GP, Portilla D. Fibrate prevents cisplatin-induced proximal tubule cell death. Kidney Int. 2005;68(6):2680–93. https://doi.org/10.1111/j.1523-1755.2005.00739.x.
Wei Q, Dong G, Franklin J, Dong Z. The pathological role of Bax in cisplatin nephrotoxicity. Kidney Int. 2007;72(1):53–62. https://doi.org/10.1038/sj.ki.5002256.
Zhang D, Liu Y, Wei Q, Huo Y, Liu K, Liu F, et al. Tubular p53 regulates multiple genes to mediate AKI. J Am Soc Nephrol. 2014;25(10):2278–89. https://doi.org/10.1681/ASN.2013080902.
Yang C, Kaushal V, Shah SV, Kaushal GP. Autophagy is associated with apoptosis in cisplatin injury to renal tubular epithelial cells. Am J Physiol Renal Physiol. 2008;294(4):F777–87. https://doi.org/10.1152/ajprenal.00590.2007.
Takahashi A, Kimura T, Takabatake Y, Namba T, Kaimori J, Kitamura H, et al. Autophagy guards against cisplatin-induced acute kidney injury. Am J Pathol. 2012;180(2):517–25. https://doi.org/10.1016/j.ajpath.2011.11.001.
Li J, Gui Y, Ren J, Liu X, Feng Y, Zeng Z, et al. Metformin protects against cisplatin-induced tubular cell apoptosis and acute kidney injury via AMPKalpha-regulated autophagy induction. Sci Rep. 2016;6:23975. https://doi.org/10.1038/srep23975.
Jiang M, Wei Q, Dong G, Komatsu M, Su Y, Dong Z. Autophagy in proximal tubules protects against acute kidney injury. Kidney Int. 2012;82(12):1271–83. https://doi.org/10.1038/ki.2012.261.
Guan Y, Breyer MD. Peroxisome proliferator-activated receptors (PPARs): novel therapeutic targets in renal disease. Kidney Int. 2001;60(1):14–30. https://doi.org/10.1046/j.1523-1755.2001.00766.x.
Barish GD, Narkar VA, Evans RM. PPAR delta: a dagger in the heart of the metabolic syndrome. J Clin Invest. 2006;116(3):590–7. https://doi.org/10.1172/JCI27955.
Lee CH, Chawla A, Urbiztondo N, Liao D, Boisvert WA, Evans RM, et al. Transcriptional repression of atherogenic inflammation: modulation by PPARdelta. Science. 2003;302(5644):453–7. https://doi.org/10.1126/science.1087344.
Coll T, Alvarez-Guardia D, Barroso E, Gomez-Foix AM, Palomer X, Laguna JC, et al. Activation of peroxisome proliferator-activated receptor-{delta} by GW501516 prevents fatty acid-induced nuclear factor-{kappa}B activation and insulin resistance in skeletal muscle cells. Endocrinology. 2010;151(4):1560–9. https://doi.org/10.1210/en.2009-1211.
Mikami D, Kimura H, Kamiyama K, Torii K, Kasuno K, Takahashi N, et al. Telmisartan activates endogenous peroxisome proliferator-activated receptor-delta and may have anti-fibrotic effects in human mesangial cells. Hypertens Res. 2014;37(5):422–31. https://doi.org/10.1038/hr.2013.157.
Kimura H, Mikami D, Kamiyama K, Sugimoto H, Kasuno K, Takahashi N, et al. Telmisartan, a possible PPAR-delta agonist, reduces TNF-alpha-stimulated VEGF-C production by inhibiting the p38MAPK/HSP27 pathway in human proximal renal tubular cells. Biochem Biophys Res Commun. 2014;454(2):320–7. https://doi.org/10.1016/j.bbrc.2014.10.077.
Wang YX, Zhang CL, Yu RT, Cho HK, Nelson MC, Bayuga-Ocampo CR, et al. Regulation of muscle fiber type and running endurance by PPARdelta. PLoS Biol. 2004;2(10):e294. https://doi.org/10.1371/journal.pbio.0020294.
Wang P, Liu J, Li Y, Wu S, Luo J, Yang H, et al. Peroxisome proliferator-activated receptor delta is an essential transcriptional regulator for mitochondrial protection and biogenesis in adult heart. Circ Res. 2010;106(5):911–9. https://doi.org/10.1161/CIRCRESAHA.109.206185.
Li Q, Li L, Wang F, Chen J, Zhao Y, Wang P, et al. Dietary capsaicin prevents nonalcoholic fatty liver disease through transient receptor potential vanilloid 1-mediated peroxisome proliferator-activated receptor delta activation. Pflugers Arch. 2013;465(9):1303–16. https://doi.org/10.1007/s00424-013-1274-4.
Palomer X, Capdevila-Busquets E, Botteri G, Salvado L, Barroso E, Davidson MM, et al. PPARbeta/delta attenuates palmitate-induced endoplasmic reticulum stress and induces autophagic markers in human cardiac cells. Int J Cardiol. 2014;174(1):110–8. https://doi.org/10.1016/j.ijcard.2014.03.176.
Okada H, Kikuta T, Inoue T, Kanno Y, Ban S, Sugaya T, et al. Dexamethasone induces connective tissue growth factor expression in renal tubular epithelial cells in a mouse strain-specific manner. Am J Pathol. 2006;168(3):737–47. https://doi.org/10.2353/ajpath.2006.050656.
Lee JM, Wagner M, Xiao R, Kim KH, Feng D, Lazar MA, et al. Nutrient-sensing nuclear receptors coordinate autophagy. Nature. 2014;516(7529):112–5. https://doi.org/10.1038/nature13961.
Wei Y, Pattingre S, Sinha S, Bassik M, Levine B. JNK1-mediated phosphorylation of Bcl-2 regulates starvation-induced autophagy. Mol Cell. 2008;30(6):678–88. https://doi.org/10.1016/j.molcel.2008.06.001.
Kim YS, Jung MH, Choi MY, Kim YH, Sheverdin V, Kim JH, et al. Glutamine attenuates tubular cell apoptosis in acute kidney injury via inhibition of the c-Jun N-terminal kinase phosphorylation of 14-3-3. Crit Care Med. 2009;37(6):2033–44. https://doi.org/10.1097/CCM.0b013e3181a005ba.
Barlaka E, Gorbe A, Gaspar R, Paloczi J, Ferdinandy P, Lazou A. Activation of PPARbeta/delta protects cardiac myocytes from oxidative stress-induced apoptosis by suppressing generation of reactive oxygen/nitrogen species and expression of matrix metalloproteinases. Pharmacol Res. 2015;95–96:102–10. https://doi.org/10.1016/j.phrs.2015.03.008.
Bao XC, Fang YQ, You P, Zhang S, Ma J. Protective role of peroxisome proliferator-activated receptor beta/delta in acute lung injury induced by prolonged hyperbaric hyperoxia in rats. Respir Physiol Neurobiol. 2014;199:9–18. https://doi.org/10.1016/j.resp.2014.04.004.
An YQ, Zhang CT, Du Y, Zhang M, Tang SS, Hu M, et al. PPARdelta agonist GW0742 ameliorates Abeta1-42-induced hippocampal neurotoxicity in mice. Metab Brain Dis. 2016;31(3):663–71. https://doi.org/10.1007/s11011-016-9800-7.
Kaushal GP, Shah SV. Autophagy in acute kidney injury. Kidney Int. 2016;89(4):779–91. https://doi.org/10.1016/j.kint.2015.11.021.
Acknowledgements
This study was supported by Grants-in-Aid of the Japan Society of Promotion of Science (JSPS KAKENHI Grant Numbers: JP15K09252 to H.K., JP15H04836 to M.I.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Shan, J., Kimura, H., Yokoi, S. et al. PPAR-δ activation reduces cisplatin-induced apoptosis via inhibiting p53/Bax/caspase-3 pathway without modulating autophagy in murine renal proximal tubular cells. Clin Exp Nephrol 25, 598–607 (2021). https://doi.org/10.1007/s10157-021-02039-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-021-02039-2