Abstract
Background
In living kidney transplantation, predicting the risk of end-stage kidney disease in the organ donors though crucial remains to be resolved. Thus, any useful biomarker to predict kidney outcome would be highly desirable to safeguard donors.
Methods
This retrospective study was conducted at Nagoya Daini Red Cross Hospital to confirm whether an increase in preserved kidney volume (PKV) was a predict marker of proteinuria. A change of PKV before and 1 year after kidney donation was measured, and its association with proteinuria 3 years after the donation was analyzed.
Results
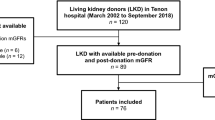
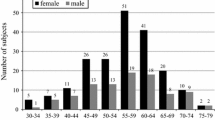
A total of 119 kidney donors who met the Japanese donor guideline were enrolled. The mean age was 57.4 years, 46.2% were male. The mean values of the variables before kidney donation (baseline) were: BMI levels: 23.4 kg/m2, BSA-adjusted PKV: 132.9 cm3/1.73 m2, and estimated glomerular filtration rate (eGFRave): 82.9 mL/min/1.73 m2. A positive correlation was noted between BSA-adjusted PKV and eGFRave (r = 0.61, p < 0.001). BSA-adjusted PKV increased by 19.5% 1 year after donation, and the median urine protein was 0.04 g/gCre. Linear regression analyses showed that change of PKV and BSA-adjusted PKV before the donation were significantly associated with proteinuria 3 years after donation.
Conclusion
Change of PKV and BSA-adjusted PKV before donation is important factors for proteinuria after donation under the Japanese donor guidelines. Further studies are needed to confirm whether these factors are associated with renal survival after donation.


Similar content being viewed by others
Abbreviations
- ESKD:
-
End-stage kidney disease
- RFR:
-
Renal functional reserve
- GFR:
-
Glomerular filtration rate
- PKV:
-
Preserved kidney volume
- CKD:
-
Chronic kidney disease
- CT:
-
Computed tomography
- Cre:
-
Creatinine
- BSA:
-
Body surface area
- HT:
-
Hypertension
- HLP:
-
Hyperlipidemia
- DM:
-
Diabetes mellitus
- HbA1c:
-
Hemoglobin A1c
- eGFRcre:
-
Estimated creatinine-based GFR
- eGFRcys:
-
Estimated cystatin C-based GFR
- eGFRave:
-
Averaged value of eGFRcre and eGFRcys
- BP:
-
Blood pressure
- BMI:
-
Body mass index
References
Mjøen G, Hallan S, Hartmann A, Foss A, Midtvedt K, Øyen O, et al. Long-term risks for kidney donors. Kidney Int. 2014;86(1):162–7.
O’Keeffe LM, Ramond A, Oliver-Williams C, Willeit P, Paige E, Trotter P, et al. Mid-and long-term health risks in living kidney donors: a systematic review and meta-analysis. Ann Intern Med. 2018;168(4):276–84.
Wainright JL, Robinson AM, Wilk AR, Klassen DK, Cherikh WS, Stewart DE. Risk of ESRD in prior living kidney donors. Am J Transplant. 2018;18(5):1129–39.
Maggiore U, Budde K, Heemann U, Hilbrands L, Oberbauer R, Oniscu GC, et al. Long-term risks of kidney living donation: review and position paper by the ERA-EDTA DESCARTES working group. Nephrol Dial Transplant. 2017;32(2):216–23.
Sharma A, Mucino MJ, Ronco C. Renal functional reserve and renal recovery after acute kidney injury. Nephron Clin Pract. 2014;127(1–4):94–100.
Taner T, Iqbal CW, Textor SC, Stegall MD, Ishitani MB. Compensatory hypertrophy of the remaining kidney in medically complex living kidney donors over the long term. Transplantation. 2015;99(3):555–9. https://doi.org/10.1097/TP.0000000000000356.
Karam Z, Tuazon J. Anatomic and physiologic changes of the aging kidney. Clin Geriatr Med. 2013;29(3):555–64.
van Londen M, Kasper N, Hessels NR, Messchendorp AL, Bakker SJ, Sanders J-S, et al. Renal functional reserve capacity before and after living kidney donation. Am J Physiol Renal Physiol. 2018;315(6):F1550–4.
Dicker S, Shirley D. Mechanism of compensatory renal hypertrophy. J Physiol. 1971;219(3):507–23.
Liu B, Preisig PA. Compensatory renal hypertrophy is mediated by a cell cycle-dependent mechanism. Kidney Int. 2002;62(5):1650–8.
Piras D, Masala M, Delitala A, Urru SA, Curreli N, Balaci L, et al. Kidney size in relation to ageing, gender, renal function, birthweight and chronic kidney disease risk factors in a general population. Nephrol Dial Transplant. 2020;35(4):640–7.
Morozumi KIN, Katayama A, Goto N. Living kidney donor guideline in Japan. 2014.
Hiramitsu T, Tomosugi T, Futamura K, Okada M, Tsujita M, Goto N, et al. Preoperative comorbidities and outcomes of medically complex living kidney donors. Kidney Int Rep. 2020;5(1):13–27.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53(6):982–92.
Imai E, Iseki K, Nitta K. Clinical practice guidebook for diagnosis and treatment of chronic kidney disease. Japan Soc Nephrol. 2012;2012:18–21.
Essential points from evidence-based clinical practice guidelines for chronic kidney disease 2018. Clinical and Experimental Nephrology. 2019; 23: 1–15.
Yamamoto T, Watarai Y, Kobayashi T, Matsuda Y, Tsujita M, Hiramitsu T, et al. Kidney volume changes in patients with autosomal dominant polycystic kidney disease after renal transplantation. Transplantation. 2012;93(8):794–8.
Rojas-Canales DM, Li JY, Makuei L, Gleadle JM. Compensatory renal hypertrophy following nephrectomy: when and how? Nephrology (Carlton). 2019;24(12):1225–32. https://doi.org/10.1111/nep.13578.
Chen Z, Fang J, Li G, Zhang L, Xu L, Pan G et al., editors. Compensatory changes in the retained kidney after nephrectomy in a living related donor. Transplantation proceedings; 2012: Elsevier.
Chen J-K, Nagai K, Chen J, Plieth D, Hino M, Xu J, et al. Phosphatidylinositol 3-kinase signaling determines kidney size. J Clin Invest. 2015;125(6):2429–44.
Freedman BI, Iskandar SS, Appel RG. The link between hypertension and nephrosclerosis. Am J Kidney Dis. 1995;25(2):207–21.
Denic A, Lieske JC, Chakkera HA, Poggio ED, Alexander MP, Singh P, et al. The substantial loss of nephrons in healthy human kidneys with aging. J Am Soc Nephrol. 2017;28(1):313–20.
Chang S-G, Kim J-H, Lee S-J, Choi J-M, Huh J-S, editors. Factors influencing contralateral renal hypertrophy after living donor nephrectomy. Transplantation proceedings; 2002.
Jeon HG, Gong IH, Hwang JH, Choi DK, Lee SR, Park DS. Prognostic significance of preoperative kidney volume for predicting renal function in renal cell carcinoma patients receiving a radical or partial nephrectomy. BJU Int. 2012;109(10):1468–73.
Wickman C, Kramer H, editors. Obesity and kidney disease: potential mechanisms. Seminars in nephrology; 2013: Elsevier.
Locke JE, Reed RD, Massie A, MacLennan PA, Sawinski D, Kumar V, et al. Obesity increases the risk of end-stage renal disease among living kidney donors. Kidney Int. 2017;91(3):699–703.
Acknowledgements
We sincerely thank all the staff of the Kidney Disease Center at Nagoya Daini Red Cross Hospital for their cooperation with this study.
Funding
None.
Author information
Authors and Affiliations
Contributions
MT: research idea and study design, data acquisition, data interpretation, statistical analysis, and wrote manuscript. NG: data acquisition. KF: data acquisition. MO: data acquisition. TH: data acquisition. NS: data acquisition. KU: supervision. KM: supervision. YW: data acquisition and supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Research involving human and animal rights
This study was performed per the Declaration of Helsinki, and the Ethics Committee approved the research protocol of the Nagoya Daini Red Cross Hospital Research Institute (Approval no. 1329). Because of a retrospective cohort study, informed consent from individual participants was waived.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Tsujita, M., Goto, N., Futamura, K. et al. The importance of kidney volume as a marker in the assessment of living-donor kidney transplantation in Japan. Clin Exp Nephrol 25, 537–544 (2021). https://doi.org/10.1007/s10157-020-02014-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-020-02014-3