Abstract
Background
Heavy proteinuria at diagnostic renal biopsy has been reported as an independent risk factor for deteriorating renal function in benign nephrosclerosis (BNS). However, studies investigating the relationship between the amount of proteinuria during follow-up and long-term renal prognosis in BNS are limited. This study aimed to assess the relationship between time-averaged proteinuria (TAP) and renal prognosis in BNS.
Methods
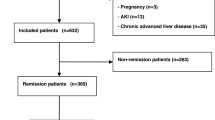
The study participants included 98 patients with biopsy-proven BNS (average age 52 ± 13 years, estimated glomerular filtration rate (eGFR) 53 ± 25 ml/min/1.73 m2, urine protein excretion at baseline 1.34 ± 1.30 g/gCr) from the Jikei University Hospital. Multivariate analysis was used to investigate the effects of TAP and other clinicopathological findings on the risk for renal outcome in biopsy-proven BNS (a 30% decline in eGFR from baseline or end-stage renal disease). Proteinuria was measured every 6 months and the mean value was used as an indicator of TAP.
Results
The average observation period was 56 ± 43 months. In the unadjusted model, higher levels of TAP and urinary protein at baseline, glomerulosclerosis, and tubulointerstitial damage were associated with renal prognosis. The adjusted model demonstrated a significant association between TAP and renal outcomes (hazard ratio 5.45, 95% confidence interval 3.02–10.7), which was independent of higher baseline proteinuria, glomerulosclerosis, and tubulointerstitial damage.
Conclusions
TAP is an independent risk factor for renal prognosis in patients with BNS, indicating the significance of urinary protein excretion during follow-up for the progression of BNS. Clinicians should understand the importance of follow-up evaluation for proteinuria in patients with BNS.


Similar content being viewed by others
Data availability
The datasets generated and analyzed during the study are not publicly available due to the terms of consent to which the participants agreed, but the datasets generated and/or analyzed during the current study are available upon request from the corresponding author.
References
Ruilope LM, van Veldhuisen DJ, Ritz E, Luscher TF. Renal function: the Cinderella of cardiovascular risk profile. J Am Coll Cardiol. 2001;38:1782–7.
Shoji T, Abe T, Matsuo H, Egusa G, Yamasaki Y, Kashihara N, et al. Chronic kidney disease, dyslipidemia, and atherosclerosis. J Atheroscler Thromb. 2012;19:299–315.
Iseki K, Ikemiya Y, Iseki C, Takishita S. Proteinuria and the risk of developing end-stage renal disease. Kidney Int. 2003;63:1468–74.
Liyanage T, Ninomiya T, Jha V, Neal B, Patrice H, Okpechi I, et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet. 2015;385:1975–82.
Ritz E. Minor renal dysfunction: an emerging independent cardiovascular risk factor. Heart. 2003;89:963–4.
Shiraishi N, Kitamura K, Kohda Y, Iseki K, Tomita K. Prevalence and risk factor analysis of nephrosclerosis and ischemic nephropathy in the Japanese general population. Clin Exp Nephrol. 2014;18:461–8.
Sumida K, Hoshino J, Ueno T, Mise K, Hayami N, Suwabe T, et al. Effect of proteinuria and glomerular filtration rate on renal outcome in patients with biopsy-proven benign nephrosclerosis. PLoS ONE. 2016;11:e0147690.
Barbour SJ, Cattran DC, Espino-Hernandez G, Hladunewich MA, Reich HN. Identifying the ideal metric of proteinuria as a predictor of renal outcome in idiopathic glomerulonephritis. Kidney Int. 2015;88:1392–401.
Reich HN, Troyanov S, Scholey JW, Cattran DC. The toronto glomerulonephritis registry. Remission of proteinuria improves prognosis in IgA nephropathy. J Am Soc Nephrol. 2007;18:3177–83.
Nishi S, Ubara Y, Utsunomiya Y, Okada K, Obata Y, Kai H, et al. Evidence-based clinical practice guidelines for nephrotic syndrome 2014. Clin Exp Nephrol. 2016;20:342–70.
Anonymous. Manual for pathological diagnosis of diabetic nephropathy and hypertensive nephrosclerosis. Nihon Jinzo Gakkai Shi. 2015;57:649–725. (in Japanese)
Vikse BE, Aasarød K, Bostad L, Iversen BM. Clinical prognostic factors in biopsy-proven benign nephrosclerosis. Nephrol Dial Transpl. 2003;18:517–23.
Kanda E, Usui T, Kashihara N, Iseki C, Iseki K, Nangaku M. Importance of glomerular filtration rate change as surrogate endpoint for the future incidence of end-stage renal disease in general Japanese population: community-based cohort study. Clin Exp Nephrol. 2018;22:318–27.
Altman DG, Bland JM. Diagnostic tests 3: receiver operating characteristic plots. BMJ. 1994;309:188.
Obuchowski NA, Lieber ML, Wians FH. ROC curves in clinical chemistry: uses, misuses, and possible solutions. Clin Chem. 2004;50:1118–25.
Kanda Y. Investigation of the freely available easy-to-use software 'EZR' for medical statistics. Bone Marrow Transpl. 2013;48:452–8.
Matsuzaki K, Suzuki Y, Nakata J, Sakamoto N, Horikoshi S, Kawamura T, et al. Nationwide survey on current treatments for IgA nephropathy in Japan. Clin Exp Nephrol. 2013;17:827–33.
Kee YK, Yoon CY, Kim SJ, Moon SJ, Kim CH, Park JT, et al. Determination of the optimal target level of proteinuria in the management of patients with glomerular diseases by using different definitions of proteinuria. Medicine. 2017;96:e8154.
Momoki K, Kataoka H, Moriyama T, Mochizuki T, Nitta K. Hyperuricemia as a predictive marker for progression of nephrosclerosis: clinical assessment of prognostic factors in biopsy-proven arterial/arteriolar nephrosclerosis. J Atheroscler Thromb. 2017;24:630–42.
Haruhara K, Tsuboi N, Kanzaki G, Koike K, Suyama M, Shimizu A, et al. Glomerular density in biopsy-proven hypertensive nephrosclerosis. Am J Hypertens. 2015;28:1164–71.
Chronic Kidney Disease Prognosis Consortium, Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, et al. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet. 2010;375:2073–81.
Bouchi R, Babazono T, Yoshida N, Nyumara I, Toya K, Hayashi T, et al. Association of albuminuria and reduced estimated glomerular filtration rate with incident stroke and coronary artery disease in patients with type 2 diabetes. Hypertens Res. 2010;33:1298–304.
Remuzzi G, Bertani T. Pathophysiology of progressive nephropathies. N Engl J Med. 1998;339:1448–566.
Eddy AA, Giachelli CM. Renal expression of genes that promote interstitial inflammation and fibrosis in rats with protein-overload proteinuria. Kidney Int. 1995;47:1546–57.
Abbate M, Zoja C, Remuzzi G. How does proteinuria cause progressive renal damage? J Am Soc Nephrol. 2006;17:2974–84.
Mills KT, Chen J, Yang W, Appel LJ, Kusek JW, Alper A, et al. Sodium excretion and the risk of cardiovascular disease in patients with chronic kidney disease. JAMA. 2016;315:2200–10.
Slagman MC, Waanders F, Hemmelder MH, Woittiez A-J, Janssen WMT, Lambers Heerspink HJ, et al. Moderate dietary sodium restriction added to angiotensin-converting enzyme inhibition compared with dual blockade in lowering proteinuria and blood pressure: randomized controlled trial. BMJ. 2011;343:d4366.
Yamanouchi M, Hoshino J, Ubara Y, Takaichi K, Kinowaki K, Fujii T, et al. Clinicopathological predictors for progression of chronic kidney disease in nephrosclerosis: a biopsy-based cohort study. Nephrol Dial Transplant. 2018. https://doi.org/10.1093/ndt/gfy121.
Acknowledgements
We thank all staff, who were part of the Division of Nephrology and Hypertension, The Jikei University School of Medicine, for their comments and technical assistance.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee at the Jikei University hospital (approval number 29-110), where the studies were conducted and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
All patients included in this study provided their written informed consent for performing the renal biopsy and analyzing the data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Amano, H., Koike, K., Haruhara, K. et al. Time-averaged proteinuria during follow-up and renal prognosis in patients with biopsy-proven benign nephrosclerosis. Clin Exp Nephrol 24, 688–695 (2020). https://doi.org/10.1007/s10157-020-01885-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-020-01885-w