Abstract
Background
The Kidney Disease: Improving Global Outcomes group (KDIGO) defined acute kidney injury (AKI) as an elevation of serum creatinine (sCR) exceeding 0.3 mg/dl within 48 h. The widely used adverse events criteria for chemotherapy, Common Toxicity Criteria for Adverse Events Version 4.0 (CTCAE v4.0), also defined AKI as sCR exceeding 0.3 mg/dl, but with no provision of a time course. Here, we attempted to clarify the impact of AKI (CTCAE v4.0) during cisplatin-based chemotherapy on clinical outcome of patients with advanced urothelial cancer (UC).
Methods
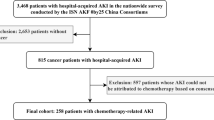
This multicenter retrospective study included 230 UC patients who received cisplatin-based chemotherapy.
Results
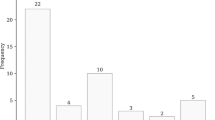
During the first chemotherapy course, AKI (CTCAE v4.0) episodes were observed in 61 patients (26.5 %), whereas only four patients (1.5 %) experienced AKI (KDIGO) episodes. Both the pretreatment estimated glomerular filtration rate (eGFR) and creatinine clearance by Cockcroft–Gault formula were not efficient predictors for the development of AKI (CTCAE v4.0). AKI (CTCAE v4.0) impacted renal function: at the start of second-course chemotherapy, the average eGFR of the patients with AKI (CTCAE v4.0) was 54.1 ml/min/1.73 m2, significantly lower than that of patients without AKI (CTCAE v4.0) (63.4 ml/min/1.73 m2). As a result, only 57.4 % of patients with AKI (CTCAE v4.0) received the planned treatment at the second course. The survival of the patients who developed AKI (CTCAE v4.0) was significantly worse than that of the patients who did not. The 3-year OSs were 10.3 and 21.4 %, respectively (P = 0.02).
Conclusion
The present study demonstrated that AKI (CTCAE v4.0) during chemotherapy had a negative impact on both the intensity of subsequent chemotherapy and oncological outcomes.


Similar content being viewed by others
References
von der Maase H, Hansen SW, Roberts JT, et al. Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol. 2000;18(17):3068–77.
Sternberg CN, Yagoda A, Scher HI, et al. Methotrexate, vinblastine, doxorubicin, and cisplatin for advanced transitional cell carcinoma of the urothelium. Efficacy and patterns of response and relapse. Cancer. 1989;64(12):2448–58.
Dash A, Galsky MD, Vickers AJ, et al. Impact of renal impairment on eligibility for adjuvant cisplatin-based chemotherapy in patients with urothelial carcinoma of the bladder. Cancer. 2006;107(3):506–13.
Launay-Vacher V, Ayllon J, Janus N, et al. Drug management of prostate cancer: prevalence and consequences of renal insufficiency. Clin Genitourin Cancer. 2009;7(3):E83–9.
Vaughn DJ. Chemotherapeutic options for cisplatin-ineligible patients with advanced carcinoma of the urothelium. Cancer Treat Rev. 2008;34(4):328–38.
Ichioka D, Miyazaki J, Inoue T, et al. Impact of renal function of patients with advanced urothelial cancer on eligibility for first-line chemotherapy and treatment outcomes. Jpn J Clin Oncol. 2015;45(9):867–73.
Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract. 2012;120(4):c179–84.
Matsuo S, Imai E, Horio M, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53(6):982–92.
Coresh J, Astor BC, Greene T, Eknoyan G, Levey AS. Prevalence of chronic kidney disease and decreased kidney function in the adult US population: Third National Health and Nutrition Examination Survey. Am J Kidney Dis. 2003;41(1):1–12.
Cockcroft DW, Gault MH. Prediction of Creatinine Clearance from Serum Creatinine. Nephron. 1976;16(1):31–41.
Jin JO, Lehmann J, Taxy J, et al. Phase II trial of adjuvant gemcitabine plus cisplatin-based chemotherapy in patients with locally advanced bladder cancer. Clin Genitourin Cancer. 2006;5(2):150–4.
Maeda T, Takahashi A, Hirobe M, et al. Adverse events of MVAC chemotherapy in patients with advanced urothelial cancer of the bladder. Hinyokika Kiyo. 2007;53(4):213–9.
Canter D, Viterbo R, Kutikov A, et al. Baseline renal function status limits patient eligibility to receive perioperative chemotherapy for invasive bladder cancer and is minimally affected by radical cystectomy. Urology. 2011;77(1):160–5.
Tsao CK, Moshier E, Seng SM, et al. Impact of the CKD-EPI equation for estimating renal function on eligibility for cisplatin-based chemotherapy in patients with urothelial cancer. Clin Genitourin Cancer. 2012;10(1):15–20.
Powles T, Eder JP, Fine GD, et al. MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature. 2014;515(7528):558–62.
Soares M, Salluh JI, Carvalho MS, Darmon M, Rocco JR, Spector N. Prognosis of critically ill patients with cancer and acute renal dysfunction. J Clin Oncol. 2006;24(24):4003–10.
Christiansen CF, Johansen MB, Langeberg WJ, Fryzek JP, Sorensen HT. Incidence of acute kidney injury in cancer patients: a Danish population-based cohort study. Eur J Intern Med. 2011;22(4):399–406.
Cosmai L, Porta C, Gallieni M, Perazella MA. Onco-nephrology: a decalogue. Nephrol Dial Transpl. 2016;31(4):515–9.
Cantiello F, Cicione A, Salonia A, et al. Association between metabolic syndrome, obesity, diabetes mellitus and oncological outcomes of bladder cancer: a systematic review. Int J Urol. 2015;22(1):22–32.
Montella M, Di Maso M, Crispo A, et al. Metabolic syndrome and the risk of urothelial carcinoma of the bladder: a case-control study. BMC Cancer. 2015;15:720.
James MT, Grams ME, Woodward M, et al. A meta-analysis of the association of estimated GFR, albuminuria, diabetes mellitus, and hypertension with acute kidney injury. Am J Kidney Dis. 2015;66(4):602–12.
Acknowledgments
This work was supported in part by a Grant-in-Aid for Scientific Research (C) (26462397) and a Grant-in-Aid for Scientific Research (B) (26293349).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee at which the studies were conducted (IRB approval number H23-33) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent as opt-out was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Ishitsuka, R., Miyazaki, J., Ichioka, D. et al. Impact of acute kidney injury defined by CTCAE v4.0 during first course of cisplatin-based chemotherapy on treatment outcomes in advanced urothelial cancer patients. Clin Exp Nephrol 21, 732–740 (2017). https://doi.org/10.1007/s10157-016-1327-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-016-1327-z