Abstract
Background
Our previous in vitro studies suggested that cyclin AMP (cAMP) signaling protects against podocyte injury. However, the molecular mechanisms remain unknown. The aim of the present study was to explore the role of forskolin, an agonist for adenylate cyclase, on ezrin/radixin/moesin (ERM) phosphorylation and chloride intracellular channel 5 (CLIC5) expressions in injured podocytes.
Methods
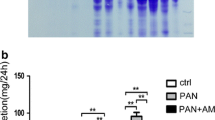
ADR nephrosis model were induced by adriamycin (ADR) injection in BalB/C mice. Parts of ADR nephrosis mice were pretreated with forskolin. Albuminuria was estimated by urine Coomassie blue stain. Nephrin, synaptopodin, CLIC5, phosphorylated ERM and podocalyxin were measured by confocal microscopy. CLIC5 and phosphorylated ERM also were studied using western blotting. RhoA and Rac1 were estimated by G-Lisa kit.
Results
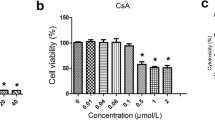
We found that forskolin partially alleviated albuminuria and width of foot processes. Nephrin, synaptopodin, phosphorylated-ERM (p-ERM) and CLIC5 expression were decreased in ADR mice, which were improved by forskolin pretreatment. In vitro studies, pretreatment of podocytes with pCPT-cAMP(PKA-selective cAMP analogue)prevented puromycin aminonucleoside (PAN)-induced CLIC5 downregulation. 8-pCPT-2′-O-Me-cAMP (2Me-cAMP, an Epac-selective cAMP analogue) blocked PAN-induced p-ERM downregulation. PAN inhibited RhoA activation in podocytes, which could be prevented by pCPT-cAMP pretreatment. Y-27632, a Rho inhibitor, decreased CLIC5 expression in podocytes.
Conclusion
Activation cAMP signaling might attenuate albuminuria in ADR-induced nephrosis mice. Different downstream signaling pathway might mediate cAMP protection on CLIC5 and p-ERM expression, respectively.




Similar content being viewed by others
References
Yu D, Petermann A, Kunter U, Rong S, Shankland SJ, Floege J. Urinary podocyte loss is a more specific marker of ongoing glomerular damage than proteinuria. J Am Soc Nephrol. 2005;16:1733–41.
Ding G, Reddy K, Kapasi AA, Franki N, Gibbons N, Kasinath BS, Singhal PC. Angiotensin II induces apoptosis in rat glomerular epithelial cells. Am J Renal Physiol. 2002;283:F173–80.
Kerjaschki D. Caught flat-footed: podocyte damage and the molecular bases of focal glomerulosclerosis. J Clin Invest. 2001;108:1583–7.
Pavenstadt H, Kriz W, Kretzler M. Cell biology of the glomerular podocyte. Physiol Rev. 2003;83:253–307.
Nielsen JS, McNagny KM. The role of podocalyxin in health and disease. J Am Soc Nephrol. 2009;20:1669–76.
Koop K, Eikmans M, Baelde HJ, Kawachi H, De Heer E, Paul LC, Bruijn JA. Expression of podocyte-associated molecules in acquired human kidney disease. J Am Soc Nephrol. 2003;14:2063–71.
Kerjaschki D, Vernillo AT, Farquhar MG. Reduced sialylation of podocalyxin-the major sialoprotein of the rat kidney glomerulus- in aminonucleoside nephrosis. Am J Pathol. 1985;118:343–9.
Pierchala BA, Murioz MR, Tsui CC. Proteomic analysis of the slit diaphragm complex: CLIC5 is a protein critial for podocyte morphology and function. Kidney Int. 2010;78:868–82.
Wegner B, Al-Momany A, Kulak SC, Kozlowski K, Obeidat M, Jahroudi N, Paes J, Berryman M, Ballermann BJ. CLIC5A, a component of the ezrin-podocalyxin complex in glomeruli, is a determinant of podocyte integrity. Am J Physiol Renal Physiol. 2010;298:1492–503.
Gu L, Liang X, Wang L, Yan Y, Ni Z, Dai H, Gao J, Mou S, Wang Q, Chen X, Wang L, Qian J. Functional metabotropic glutamate receptors 1 and 5 are expressed in murine podocytes. Kidney Int. 2012;81:458–68.
Gu L, Dai Y, Xu J, Mallipattu S, Kaufman L, Klotman PE, He JC, Chuang PY. Deletion of podocytes STAT3 mitigates the entire spectrum of HIV-1-associated nephropathy. AIDS. 2013;27:1091–8.
Kriz W, Shirato I, Nagata M, LeHir M, Lemley KV. The podocyte’s response to stress: the enigma of foot process effacement. Am J Physiol Renal Physiol. 2013;304:F333–47.
Mundel P, Kriz W. Structure and function of podocytes: an update. Anat Embryol (Berl). 1995;192:385–97.
Amould T, Mercy L, Houbion A, Vankoningsloo S, Renard P, Pascal T, Ninane N, Demazy C, Raes M. mtCLIC is up-regulated and maintains a mitochondrial membrane potential in mtDNA-depleted L929 cells. FASEB J. 2003;17:2145–7.
Gu C, Li X, Tan Q, Wang Z, Chen L, Liu Y. MiR-183 family regulates chloride intracellular channel 5 expression in inner ear hair cells. Toxicol In Vitro. 2013;27:486–91.
Hugo C, Hugo C, Pichler R, Gordon K, Schmidt R, Amieva M, Couser WG, Furthmayr H, Johnson RJ. The cytoskeletal linking proteins, moesin and radixin, are upregulated by platelet-derived growth factor, but not basic fibroblast growth factor in experimental mesangial proliferative. J Clin Invest. 1996;97:2499–508.
Ostalska-Nowicka D, Zachwieja J, Nowicki M, Kaczmarek E, Siwinska A, Witt M. Ezrin-a useful factor in the prognosis of nephrotic syndrome in children: an immunohistochemical approach. J Clin Pathol. 2006;59:916–20.
Ivetic A, Ridley AJ. Ezrin/radixin/moesin proteins and Rho GTPase signalling in leucocytes. Immunology. 2004;112:165–76.
Hsu HH, Hoffmann S, Endlich N, Velic A, Schwab A, weide T, Schlatter E, Pavenstadt H. Mechanisms of angiotensin II signaling on cytoskeleton of podocytes. J Mol Med (Berl). 2008;86:1379–94.
Niggli V, Rossy J. Ezrin/radixin/moesin: versatile controllers of signaling molecules and of the cortical cytoskeleton. Int J Biochem Cell Biol. 2008;40:344–9.
Chen J, Cohn JA, Mandel LJ. Dephosphorylation of ezrin as an early event in renal microvillar breakdown and anoxic injury. Proc Natl Acad Sci USA. 1995;92:7495–9.
Mosenden R, Tasken K. Cyclic AMP-mediated immune regulation-overview of mechanisms of action in T cells. Cell Signal. 2011;23:1009–16.
Gloerich M, Ponsioen B, Vliem MJ, Zhang Z, Zhao J, Kooistra MR, Price LS, Ritsma L, Zwartkruis FJ, Rehmann H, Jalink K, Bos JL. Spatial regulation of cyclic AMP-Epac1 signaling in cell adhesion by ERM proteins. Mol Cell Biol. 2010;30(22):5421–31.
Ross SH, Post A, Raaijmakers JH, Verlaan I, Gloerich M, Bos JL. Ezrin is required for efficient Rap1-induced cell spreading. J Cell Sci. 2011;124:1808–18.
Oishi A, Makita N, Sato J, Iiri T. Regulation of RhoA signaling by the cAMP-dependent phosphorylation of RhoGDIa. J Biol Chem. 2012;287:38705–15.
Saito Y, Okamura M, Nakajima S, Hayakawa K, Huang T, Yao J, Kitamura M. Suppression of nephrin expression by TNF-α via interfering with the cAMP-retinoic acid receptor pathway. Am J Physiol Renal Physiol. 2010;298:F1436–44.
Acknowledgments
We are grateful to Professor Cijiang He (USA) for the podocyte clones. LG and ZN are supported by National Basic Research Program of China 973 Program 2012CB517602. LG is supported by National Nature Science Foundation Grant of China 30971363 and 81270781, as well as Nature Science Foundation Grant of Shanghai 08ZR1413200.
Conflict of interest
The authors have declared that no conflict of interest exists.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hua Tao and Xiaoying Li have contributed equally for this paper.
About this article
Cite this article
Tao, H., Li, X., Wei, K. et al. Cyclic AMP prevents decrease of phosphorylated ezrin/radixin/moesin and chloride intracellular channel 5 expressions in injured podocytes. Clin Exp Nephrol 19, 1000–1006 (2015). https://doi.org/10.1007/s10157-015-1102-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-015-1102-6