Abstract
Background and aim
The infusion of chronic angiotensin II (Ang II) has been shown to promote renal interstitial fibrosis. To evaluate the pathophysiological significance of the natriuretic peptide-GC-A system, we infused Ang II (1.0 mg/kg/day) in GC-A-deficient mice (GC-A-KO).
Methods
We used 5 groups (Wild-Saline n = 12, Wild-Ang II n = 14, GC-A-KO-Saline n = 11, GC-A-KO-Ang II n = 13, and GC-A-KO-Ang II-Hydralazine n = 10). Saline or Ang II was infused subcutaneously using an osmotic minipump for 3 weeks. Hydralazine was administered orally (0.05 g/L in drinking water).
Results
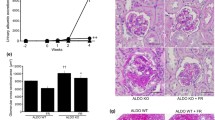
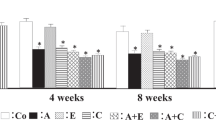
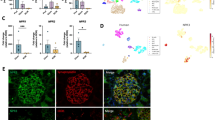
Systolic blood pressure was significantly higher in the GC-A-KO-Saline group (130 ± 12 mmHg) than in the Wild-Saline group (105 ± 30 mmHg), and was similar to that in the Wild-Ang II (141 ± 17 mmHg) and GC-A-KO-Ang II-Hydralazine (140 ± 20 mmHg) groups. Systolic blood pressure was significantly higher in the GC-A-KO-Ang II group (159 ± 21 mmHg) than in the 4 other groups. Renal tubular atrophy and interstitial fibrosis were significantly more severe in the GC-A-KO-Ang II group (atrophy 13.4 %, fibrosis 12.0 %) than in the Wild-Saline (0, 2.0 %), Wild-Ang II (2.9, 4.4 %), and GC-A-KO-Saline (0, 2.6 %) groups. Hydralazine could not inhibit this aggravation (GC-A-KO-Ang II-Hydralazine 13.5, 11.3 %). The expression of monocyte chemotactic protein-1 in tubular cells, and F4/80 and alpha-smooth muscle actin in the interstitium was clearly detected in the Ang II-infused wild and GC-A-KO groups and was associated with renal tubular atrophy and interstitial fibrosis. The expression of E-cadherin in tubular cells was absent in the Ang II-infused wild and GC-A-KO groups and was associated with renal tubular atrophy.
Conclusions
The natriuretic peptide-GC-A system may play an inhibitory role in Ang II-induced renal tubular atrophy, interstitial fibrosis, and phenotypic transformation in renal tubular cells and fibroblasts.






Similar content being viewed by others
References
Kasahara M, Mukoyama M, Sugawara A, et al. Ameliorated glomerular injury in mice overexpressing brain natriuretic peptide with renal ablation. J Am Soc Nephrol. 2000;11:1691–701.
Suganami T, Mukoyama M, Sugawara A, et al. Overexpression of brain natriuretic peptide in mice ameliorates immune-mediated renal injury. J Am Soc Nephrol. 2001;12:2652–63.
Makino H, Mukoyama M, Mori K, et al. Transgenic overexpression of brain natriuretic peptide prevents the progression of diabetic nephropathy in mice. Diabetologia. 2006;49:2514–24.
Saito Y. Roles of atrial natriuretic peptide and its therapeutic use. J Cardiol. 2010;56:262–70.
Lopez MJ, Wong SK, Kishimoto I, et al. Salt-resistant hypertension in mice lacking the guanylyl cyclase-A receptor for atrial natriuretic peptide. Nature. 1995;378:65–8.
Sabrane K, Kruse MN, Fabritz L, et al. Vascular endothelium is critically involved in the hypotensive and hypovolemic actions of atrial natriuretic peptide. J Clin Invest. 2005;115:1666–74.
Chen W, Gassner B, Börner S, et al. Atrial natriuretic peptide enhances microvascular albumin permeability by the caveolae-mediated transcellular pathway. Cardiovasc Res. 2012;93:141–51.
Ogawa Y, Mukoyama M, Yokoi H, et al. Natriuretic peptide receptor guanylyl cyclase-a protects podocytes from aldosterone-induced glomerular injury. J Am Soc Nephrol. 2012;23:1198–209.
Blasi ER, Rocha R, Rudolph AE, Blomme EA, Polly ML, McMahon EG. Aldosterone/salt induces renal inflammation and fibrosis in hypertensive rats. Kidney Int. 2003;63:1791–800.
Faulkner JL, Szcykalski LM, Springer F, Barnes JL. Origin of interstitial fibroblasts in an accelerated model of angiotensin II-induced renal fibrosis. Am J Pathol. 2005;167:1193–205.
Mitani H, Ishizaka N, Aizawa T, et al. In vivo klotho gene transfer ameliorates angiotensin II-induced renal damage. Hypertension. 2002;39:838–43.
Johnson RJ, Alpers CE, Yoshimura A, et al. Renal injury from angiotensin II-mediated hypertension. Hypertension. 1992;19:464–74.
Lombardi DM, Viswanathan M, Vio CP, Saavedra JM, Schwartz SM, Johnson RJ. Renal and vascular injury induced by exogenous angiotensin II is AT1 receptor-dependent. Nephron. 2001;87:66–74.
Kitayama H, Maeshima Y, Takazawa Y, et al. Regulation of angiogenic factors in angiotensin II infusion model in association with tubulointerstitial injuries. Am J Hypertens. 2006;19:718–27.
Ozawa Y, Kobori H, Suzaki Y, Navar LG. Sustained renal interstitial macrophage infiltration following chronic angiotensin II infusions. Am J Physiol Renal Physiol. 2007;292:F330–9.
Giachelli CM, Pichler R, Lombardi D, et al. Osteopontin expression in angiotensin II-induced tubulointerstitial nephritis. Kidney Int. 1994;45:515–24.
Nath KA. Tubulointerstitial changes as a major determinant in the progression of renal damage. Am J Kidney Dis. 1995;20:1–17.
Desmoulière A, Darby IA, Gabbiani G. Normal and pathologic soft tissue remodeling: role of the myofibroblast, with special emphasis on liver and kidney fibrosis. Lab Invest. 2003;83:1689–707.
Tokudome T, Horio T, Kishimoto I, et al. Calcineurin-nuclear factor of activated T cells pathway-dependent cardiac remodeling in mice deficient in guanylyl cyclase A, a receptor for atrial and brain natriuretic peptides. Circulation. 2005;111:3095–104.
Quesada A, Vargas F, Montoro-Molina S, et al. Urinary aminopeptidase activities as early and predictive biomarkers of renal dysfunction in cisplatin-treated rats. PLoS ONE. 2012;7:e40402.
Li P, Oparil S, Novak L, et al. ANP signaling inhibits TGF-beta-induced Smad2 and Smad3 nuclear translocation and extracellular matrix expression in rat pulmonary arterial smooth muscle cells. J Appl Physiol. 2007;102:390–8.
Weber NC, Blumenthal SB, Hartung T, Vollmar AM, Kiemer AK. ANP inhibits TNF-alpha-induced endothelial MCP-1 expression–involvement of p38 MAPK and MKP-1. J Leukoc Biol. 2003;74:932–41.
Suzuki K, Han GD, Miyauchi N, et al. Angiotensin II type 1 and type 2 receptors play opposite roles in regulating the barrier function of kidney glomerular capillary wall. Am J Pathol. 2007;170:1841–53.
Baltatu O, Silva JA Jr, Ganten D, Bader M. The brain renin-angiotensin system modulates angiotensin II-induced hypertension and cardiac hypertrophy. Hypertension. 2000;35(part 2):409–12.
Cargill RI, Coutie WJ, Lipworth BJ. The effects of angiotensin II on circulating levels of natriuretic peptides. Br J Clin Pharmacol. 1994;38:139–42.
Marin-Grez M, Fleming JT, Steinhausen M. Atrial natriuretic peptide causes pre-glomerular vasodilatation and post-glomerular vasoconstriction in rat kidney. Nature. 1986;324:473–6.
Abbate M, Zoja C, Remuzzi G. How does proteinuria cause progressive renal damage? J Am Soc Nephrol. 2006;17:2974–84.
Manotham K, Tanaka T, Matsumoto M, et al. Transdifferentiation of cultured tubular cells induced by hypoxia. Kidney Int. 2004;65:871–80.
Sun S, Ning X, Zhang Y, et al. Hypoxia-inducible factor-1alpha induces Twist expression in tubular epithelial cells subjected to hypoxia, leading to epithelial-to-mesenchymal transition. Kidney Int. 2009;75:1278–87.
Long DA, Mu W, Price KL, et al. Vascular endothelial growth factor administration does not improve microvascular disease in the salt-dependent phase of post-angiotensin II hypertension. Am J Physiol Renal Physiol. 2006;291:F1248–54.
Parekh N, Zou AP. Role of prostaglandins in renal medullary circulation: response to different vasoconstrictors. Am J Physiol. 1996;271:F653–8.
Zhu Q, Wang Z, Xia M, et al. Silencing of hypoxia-inducible factor-1α gene attenuated angiotensin II-induced renal injury in Sprague-Dawley rats. Hypertension. 2011;58:657–64.
Tokudome T, Kishimoto I, Yamahara K, et al. Impaired recovery of blood flow after hind-limb ischemia in mice lacking guanylyl cyclase-A, a receptor for atrial and brain natriuretic peptides. Arterioscler Thromb Vasc Biol. 2009;29:1516–21.
Sexton PM, Zhuo J, Mendelsohn FA. Localization and regulation of renal receptors for angiotensin II and atrial natriuretic peptide. Tohoku J Exp Med. 1992;166:41–56.
Garg R, Pandey KN. Angiotensin II-mediated negative regulation of Npr1 promoter activity and gene transcription. Hypertension. 2003;41(part 2):730–6.
Arise KK, Pandey KN. Inhibition and down-regulation of gene transcription and guanylyl cyclase activity of NPRA by angiotensin II involving protein kinase C. Biochem Biophys Res Commun. 2006;349:131–5.
Hu P, Wang J, Zhao XQ, Hu B, Lu L, Qin YH. Overexpressed C-type natriuretic peptide serves as an early compensatory response to counteract extracellular matrix remodeling in unilateral ureteral obstruction rats. Mol Biol Rep. 2013;40:1429–41.
Segawa K, Minami K, Jimi N, Nakashima Y, Shigematsu A. C-type natriuretic peptide inhibits rat mesangial cell proliferation by a phosphorylation-dependent mechanism. Naunyn Schmiedebergs Arch Pharmacol. 1998;357:70–6.
Canaan-Kühl S, Ostendorf T, Zander K, Koch KM, Floege J. C-type natriuretic peptide inhibits mesangial cell proliferation and matrix accumulation in vivo. Kidney Int. 1998;53:1143–51.
Acknowledgments
We thank the Howard Hughes Medical Institute for the GC-A knockout mice, Dr. Kyoko Shioya for her support, and Tamaki Mabuchi and Junko Nakamura for their excellent technical assistance. This study was supported by the Program for Promotion of Fundamental Studies in Health Sciences of the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan (No. 22590906).
Conflict of interest
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Yoshihara, F., Tokudome, T., Kishimoto, I. et al. Aggravated renal tubular damage and interstitial fibrosis in mice lacking guanylyl cyclase-A (GC-A), a receptor for atrial and B-type natriuretic peptides. Clin Exp Nephrol 19, 197–207 (2015). https://doi.org/10.1007/s10157-014-0982-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-014-0982-1