Abstract
Background
Hematuria is the first manifestation of urinary abnormality in immunoglobulin A nephropathy (IgAN). Hematuria has recently been reported as a risk factor for deterioration of renal function; however, its cause remains unknown.
Methods
We analyzed the surface marker of peripheral blood mononuclear cells before and immediately after tonsillectomy in IgAN patients and controls (chronic tonsillitis or tonsillar hypertrophy) by flow cytometry and investigated the association with hematuria. To prove our hypothesis that NK cells induce hematuria, we administered IL-12, activator of NK cells, to HIGA mice. In addition, we transferred cultured NK cells to nude rats and transferred the CD16+CD56+ cells, including NK cells, that are derived from the peripheral blood of IgAN patients immediately after tonsillectomy to nude rats to assess the hematuria level and renal histology of the recipients. We also performed cytotoxicity assays against glomerular endothelial cells by NK cells.
Results
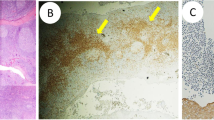
We found that IgAN patients who showed rapid deterioration of hematuria after tonsillectomy also displayed a significant increase in CD16+CD56+ cells in the peripheral blood immediately after tonsillectomy. Exogenous administration of IL-12 to HIGA mice induced hematuria. Adoptive transfer of either cells of an NK cell line, or of CD16+CD56+ cells derived from IgAN patients, into nude rats induced hematuria in the recipients. In vitro analysis showed that NK cells exert cytotoxic activity toward human glomerular endothelial cells in a dose-dependent manner.
Conclusions
CD16+CD56+ cells seem to be responsible for hematuria in IgAN.






Similar content being viewed by others
References
Pettersson E. IgA nephropathy: 30 years on. J Intern Med. 1997;242(5):349–53.
Haas M. IgA nephropathy histologically resembling focal-segmental glomerulosclerosis: a clinicopathologic study of 18 cases. Am J Kidney Dis. 1996;28(3):365–71.
Espinosa M, Ortega R, Gomez-Carrasco JM, et al. Mesangial C4d deposition: a new prognostic factor in IgA nephropathy. Nephrol Dial Transplant. 2009;24(3):886–91.
Haas M. Histologic subclassification of IgA nephropathy: a clinicopathologic study of 244 cases. Am J Kidney Dis. 1997;29(6):829–42.
Gutierrez E, Gonzalez E, Hernandez E, et al. Factors that determine an incomplete recovery of renal function in macrohematuria-induced acute renal failure of IgA nephropathy. Clin J Am Soc Nephrol. 2007;2(1):51–7.
Manno C, Strippoli GF, D’Altri C, et al. A novel simpler histological classification for renal survival in IgA nephropathy: a retrospective study. Am J Kidney Dis. 2007;49(6):763–75.
Ayalon O, Hughes EA, Cresswell P, et al. Induction of transporter associated with antigen processing by interferon gamma confers endothelial cell cytoprotection against natural killer-mediated lysis. Proc Natl Acad Sci USA. 1998;95(5):2435–40.
Nogaki F, Muso E, Kobayashi I, et al. Interleukin 12 induces crescentic glomerular lesions in a high IgA strain of ddY mice, independently of changes in IgA deposition. Nephrol Dial Transplant. 2000;15(8):1146–54.
Kobayashi I, Nogaki F, Kusano H, et al. Interleukin-12 alters the physicochemical characteristics of serum and glomerular IgA and modifies glycosylation in a ddY mouse strain having high IgA levels. Nephrol Dial Transplant. 2002;17(12):2108–16.
Hotta O, Seki S, Taguma Y, et al. CD16+ and CD56+ cells as markers of necrotizing and crescentic glomerulonephritis and active IgA glomerulonephropathy. Nephron. 1991;57(2):239–40.
Antonaci S, Polignano A, Ottolenghi A, et al. Redistribution of natural killer (NK) cell frequency and NK cytotoxic activity in primary IgA nephropathy. Cytobios. 1992;69(276):27–34.
Yildirim I, Okur E, Ciragil P, et al. Bacteraemia during tonsillectomy. J Laryngol Otol. 2003;117(8):619–23.
Maintz L, Novak N. Modifications of the innate immune system in atopic dermatitis. J Innate Immun. 2011;3(2):131–41.
Cox SN, Sallustio F, Serino G, et al. Activated innate immunity and the involvement of CX3CR1-fractalkine in promoting hematuria in patients with IgA nephropathy. Kidney Int. 2012;82(5):548–60.
Muso E, Yoshida H, Takeuchi E, et al. Enhanced production of glomerular extracellular matrix in a new mouse strain of high serum IgA ddY mice. Kidney Int. 1996;50(6):1946–57.
Beziat V, Duffy D, Quoc SN, et al. CD56brightCD16+ NK cells: a functional intermediate stage of NK cell differentiation. J Immunol. 2011;186(12):6753–61.
Yago T, Tsukuda M, Fukushima H, et al. IL-12 promotes the adhesion of NK cells to endothelial selectins under flow conditions. J Immunol. 1998;161(3):1140–5.
Segawa C, Wada T, Takaeda M, et al. In situ expression and soluble form of P-selectin in human glomerulonephritis. Kidney Int. 1997;52(4):1054–63.
Yoneda O, Imai T, Goda S, et al. Fractalkine-mediated endothelial cell injury by NK cells. J Immunol. 2000;164(8):4055–62.
Erdbruegger U, Grossheim M, Hertel B, et al. Diagnostic role of endothelial microparticles in vasculitis. Rheumatology (Oxford). 2008;47(12):1820–5.
Acknowledgments
This study was supported by a Grant-in-Aid for Young Scientists B (18790562).
Conflict of interest
All authors have declared no competing interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Iwatani, H., Nagasawa, Y., Yamamoto, R. et al. CD16+CD56+ cells are a potential culprit for hematuria in IgA nephropathy. Clin Exp Nephrol 19, 216–224 (2015). https://doi.org/10.1007/s10157-014-0968-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-014-0968-z