Abstract
Background
Anemia in chronic renal failure results from inadequate production of erythropoietin and decrease in its biological activity, and the reduced activity of erythropoietin is caused by the presence of plasma inhibitors of erythropoietin. It is reported that one of the inhibitors of erythropoietin is cyanate, a potential uremic toxin formed spontaneously from increased urea due to decreased renal function, and erythropoietin loses its biological activity due to negatively charged cyanate. The purpose of this study is to investigate the protective effects of amino acids, positively charged amino groups, and albumin binding of several toxins on erythropoietin carbamoylation.
Methods
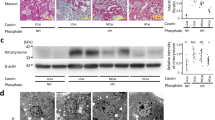
The degree of change in erythropoietin structure by cyanate was measured by the trinitrobenzenesulphonate reaction and Western blotting. The loss of biological activity of erythropoietin caused by cyanate was measured by injecting erythropoietin into rats with chronic renal failure.
Results
The free amino groups in erythropoietin decreased under cyanate treatment in a time- and concentration-dependent manner. In the cyanate treatment group, of the twenty amino acids, phenylalanine, valine, tryptophan, threonine, and lysine prevented the structural modification of erythropoietin, according to Western blot analysis. In addition, of the three proteins, albumin prevented the structural modification of erythropoietin. As for the cyanate with erythropoietin treatment group, only lysine and albumin prevented the loss of biological activity of erythropoietin in the rats.
Conclusion
The results of this study suggest that lysine and albumin may play a protective role against renal anemia by erythropoietin carbamoylation in chronic renal failure.






Similar content being viewed by others
References
Thorp ML, Eastman L, Smith DH, et al. Managing the burden of chronic kidney disease. Dis Manag. 2006;9:115–21.
IV. NKF-K/DOQI Clinical Practice Guidelines for Anemia of Chronic Kidney Disease: update 2000. Am J Kidney Dis 2001; 37: S182–S238.
Wallner SF, Vautrin RM. Evidence that inhibition of erythropoiesis is important in the anemia of chronic renal failure. J Lab Clin Med. 1981;97:170–8.
Mun KC, Golper TA. Impaired biological activity of erythropoietin by cyanate carbamylation. Blood Purif. 2000;18:13–7.
Park KD, Mun KC, Chang EJ, et al. Inhibition of erythropoietin activity by cyanate. Scand J Urol Nephrol. 2004;38:69–72.
Marier JR, Rose D. Determination of cyanate, and a study of its accumulation in aqueous solutions of urea. Anal Biochem. 1964;7:304–14.
Fluckiger R, Harmon W, Meier W, et al. Hemoglobin carbamylation in uremia. N Engl J Med. 1981;304:823–7.
Kraus LM, Jones MR, Kraus AP Jr. Essential carbamoyl-amino acids formed in vivo in patients with end-stage renal disease managed by continuous ambulatory peritoneal dialysis: isolation, identification, and quantitation. J Lab Clin Med. 1998;131:425–31.
Stim J, Shaykh M, Anwar F, et al. Factors determining hemoglobin carbamylation in renal failure. Kidney Int. 1995;48:1605–10.
Balion CM, Draisey TF, Thibert RJ. Carbamylated hemoglobin and carbamylated plasma protein in hemodialyzed patients. Kidney Int. 1998;53:488–95.
Nilsson L, Lundquist P, Kagedal B, et al. Plasma cyanate concentrations in chronic renal failure. Clin Chem. 1996;42:482–3.
Horkko S, Savolainen MJ, Kervinen K, et al. Carbamylation-induced alterations in low-density lipoprotein metabolism. Kidney Int. 1992;41:1175–81.
Allebban Z, Gibson LA, Lange RD, et al. Effects of spaceflight on rat erythroid parameters. J Appl Physiol. 1996;81:117–22.
Vanholder R, Argiles A, Baurmeister U, et al. Uremic toxicity: present state of the art. Int J Artif Organs. 2001;24:695–725.
Vanholder R, De Smet R, Glorieux G, et al. Review on uremic toxins: classification, concentration, and interindividual variability. Kidney Int. 2003;63:1934–43.
Roxborough HE, Millar CA, McEneny J, et al. Carbamylation inhibits the ferroxidase activity of ceruloplasmin. Biochem Biophys Res Commun. 1995;214:1073–8.
Wynckel A, Randoux C, Millart H, et al. Kinetics of carbamylated haemoglobin in acute renal failure. Nephrol Dial Transplant. 2000;15:1183–8.
Harding JJ. Cyanate derived from urea in uraemia. Lancet. 1992;339:492.
Kraus LM, Kraus AP Jr. Carbamoylation of amino acids and proteins in uremia. Kidney Int Suppl. 2001;78:S102–7.
Stark GR. On the reversible reaction of cyanate with sulfhydryl groups and the determination of NH2-terminal cysteine and cystine in proteins. J Biol Chem. 1964;239:1411–4.
Stark GR, Smyth DG. The use of cyanate for the determination of NH2-terminal residues in proteins. J Biol Chem. 1963;238:214–26.
Stark GR. Reactions of cyanate with functional groups of proteins. 3. Reactions with amino and carboxyl groups. Biochemistry. 1965;4:1030–6.
Satake R, Kozutsumi H, Takeuchi M, et al. Chemical modification of erythropoietin: an increase in in vitro activity by guanidination. Biochim Biophys Acta. 1990;1038:125–9.
Park KD, Park SB, Choi HJ, et al. Effect of amino acids and albumin of damage induced by cyanate in osteoblast. Korean J Nephrol. 2007;26:404–13.
Acknowledgments
This work was supported by the 2012 Inje University research grant.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Jin, K. Effects of amino acids and albumin on erythropoietin carbamoylation. Clin Exp Nephrol 17, 575–581 (2013). https://doi.org/10.1007/s10157-012-0751-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-012-0751-y