Abstract
Background
The optimal therapeutic regimen for Helicobacter pylori (H. pylori) infection has not been established in end-stage renal disease (ESRD) patients receiving hemodialysis. We investigated the efficacy and safety of a 7-day omeprazole-based triple therapy with low doses of amoxicillin and clarithromycin (OAC) for eradication of H. pylori infection in ESRD patients receiving hemodialysis.
Methods
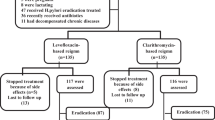
Thirty-three hemodialysis patients and 55 patients with normal renal function underwent upper gastrointestinal endoscopy. For eradication of H. pylori infection, a 7-day triple therapy with low-dose OAC (omeprazole 40 mg daily, amoxicillin 500 mg daily, and clarithromycin 500 mg daily) regimen was used. Four weeks after the completion of the OAC regimen, the success of the H. pylori eradication therapy was determined by histological examination and rapid urease test.
Results
The prevalence of H. pylori infection was 36.4% in hemodialysis patients and 65.5% in non-uremic patients (p = 0.0150). The mean duration of hemodialysis in H. pylori-positive and -negative patients was 56.8 ± 26.9 versus 66.4 ± 26.1 months, respectively (p = 0.3196). Eradication was successful in 83.4% of hemodialysis patients and 81.0% of non-uremic patients (p = 1.000). All patients completed the eradication therapy without any serious adverse effects.
Conclusion
A 7-day triple therapy with a low-dose OAC regimen was effective and safe for eradication of H. pylori infection in hemodialysis patients.
Similar content being viewed by others
References
Sugimoto M, Sakai K, Kita M, Imanishi J, Yamaoka Y. Prevalence of Helicobacter pylori infection in long-term hemodialysis patients. Kidney Int. 2009;75:96–103.
Al-Mueilo SH. Gastroduodenal lesions and Helicobacter pylori infection in hemodialysis patients. Saudi Med J. 2004;25:1010–4.
Moustafa FE, Khalil A, Abdel Wahab M, Sobh MA. Helicobacter pylori and uremic gastritis: a histopathologic study and a correlation with endoscopic and bacteriologic findings. Am J Nephrol. 1997;17:165–71.
Malfertheiner P, Megraud F, O’Morain C, Bazzoli F, El-Omar E, Graham D, et al. Current concepts in the management of Helicobacter pylori infection: the Maastricht III consensus report. Gut. 2007;56:772–81.
Mak SK, Loo CK, Wong AM, Wong PN, Lo KY, Tong GM, et al. Efficacy of a 1-week course of proton-pump inhibitor-based triple therapy for eradicating Helicobacter pylori in patients with and without chronic renal failure. Am J Kidney Dis. 2002;40:576–81.
Chey WD, Wong BC. Practice Parameters Committee of the American College of Gastroenterology. American College of Gastroenterology guideline on the management of Helicobacter pylori infection. Am J Gastroenterol. 2007;102:1808–25.
Tamura H, Tokushima H, Murakawa M, Matsumura O, Itoyama S, Sekine S, et al. Eradication of Helicobacter pylori in patients with end-stage renal disease under dialysis treatment. Am J Kidney Dis. 1997;29:86–90.
Mak SK, Loo CK, Wong PN, Lo KY, Tong GM, Lam EK, et al. A retrospective study on efficacy of proton-pump inhibitor-based triple therapy for eradication of Helicobacter pylori in patients with chronic renal failure. Singapore Med J. 2003;44:74–8.
Wang YL, Sheu BS, Huang JJ, Yang HB. Noninvasive stool antigen assay can effectively screen Helicobacter pylori Infection and assess success of eradication therapy in hemodialysis patients. Am J Kidney Dis. 2001;38:98–103.
Tsukada K, Miyazaki T, Katoh H, Masuda N, Ojima H, Fukai Y, et al. Seven-day triple therapy with omeprazole, amoxycillin and clarithromycin for Helicobacter pylori infection in hemodialysis patients. Scand J Gastroenterol. 2002;37:1265–8.
Itatsu T, Miwa H, Nagahara A, Kubota M, Miyazaki A, Sato N, et al. Eradication of Helicobacter pylori in hemodialysis patients. Ren Fail. 2007;29:97–102.
Veldhuyzen van Zanten S, Chiba N, Barkun A, Fallone C, Farley A, Cockeram A, et al. A randomized trial comparing 7-day ranitidine bismuth citrate and clarithromycin dual therapy to 7-day omeprazole, clarithromycin and amoxicillin triple therapy for the eradication of Helicobacter pylori. Can J Gastroenterol. 2003;17:533–8.
Arancibia A, Drouguett MT, Fuentes G, González G, González C, Thambo S, et al. Pharmacokinetics of amoxicillin in subjects with normal and impaired renal function. Int J Clin Pharmacol Ther Toxicol. 1982;20:447–53.
Fraschini F, Scaglione F, Demartini G. Clarithromycin clinical pharmacokinetics. Clin Pharmacokinet. 1993;25:189–204.
Fischbach LA, Goodman KJ, Feldman M, Aragaki C. Sources of variation of Helicobacter pylori treatment success in adults worldwide: a meta-analysis. Int J Epidemiol. 2002;31:128–39.
Fuccio L, Minardi ME, Zagari RM, Grilli D, Magrini N, Bazzoli F. Meta-analysis: duration of first-line proton-pump inhibitor based triple therapy for Helicobacter pylori eradication. Ann Intern Med. 2007;147:553–62.
Klotz U. Pharmacokinetic considerations in the eradication of Helicobacter pylori. Clin Pharmacokinet. 2000;38:243–70.
Humbert G, Spyker DA, Fillastre JP, Leroy A. Pharmacokinetics of amoxicillin: dosage nomogram for patients with impaired renal function. Antimicrob Agents Chemother. 1979;15:28–33.
Aydemir S, Boyacioglu S, Gur G, Demirbilek M, Can FK, Korkmaz M, et al. Helicobacter pylori infection in hemodialysis patients: susceptibility to amoxicillin and clarithromycin. World J Gastroenterol. 2005;14:842–5.
Wermeille J, Cunningham M, Dederding JP, Girard L, Baumann R, Zelger G, et al. Failure of Helicobacter pylori eradication: is poor compliance the main cause? Gastroenterol Clin Biol. 2002;26:216–9.
Harris AW, Misiewicz JJ. Treating Helicobacter pylori—the best is yet to come? Gut. 1996;39:781–3.
Acknowledgments
This paper was supported by the Second-Phase of BK-21 Project in 2010 of Konkuk University.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Chang, WC., Jo, YI., Park, HS. et al. Helicobacter pylori eradication with a 7-day low-dose triple therapy in hemodialysis patients. Clin Exp Nephrol 14, 469–473 (2010). https://doi.org/10.1007/s10157-010-0319-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-010-0319-7