Abstract
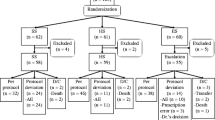
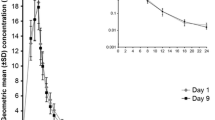
The adequacy of sitafloxacin clinical dose regimens was assessed by comparing the efficacy of the administration of 100 mg sitafloxacin once daily (100 mg qd group) and 50 mg sitafloxacin twice daily (50 mg bid group). Patients with respiratory tract infections caused by pneumococci were orally treated with sitafloxacin (100 mg qd or 50 mg bid) for 7 days. The clinical efficacy, pneumococci eradication rate, safety, and pharmacokinetic and pharmacodynamic indices of the two groups were then assessed. The clinical efficacy was 93.5 % in both groups. The pneumococci eradication rate was 98.2 % in the 100 mg qd group and 92.7 % in the 50 mg bid group. The mean of the free AUC0–24h divided by the minimum inhibitory concentration (MIC) (fAUC0–24h/MIC) did not differ significantly between the 100 mg qd (103.24) and the 50 mg bid groups (105.25). The mean of the free C peak divided by the MIC (fC peak/MIC) was higher in the 100 mg qd group (10.19) than in the 50 mg bid group (6.53). The pathogen eradication rate was 98.9 % (89/90) when the fAUC0–24h/MIC was greater than 30, and the eradication rate was 98.9 % (89/90) when the fC peak/MIC was greater than 2. The incidences of adverse drug reactions were 33.7 % in the 100 mg qd group and 40.4 % in the 50 mg bid group. No obvious differences in the efficacy and safety were observed between the dosage groups. For cases in which a sufficiently high C peak is necessary to ensure the susceptibility of the pathogens to the drug, 100 mg sitafloxacin once daily should be administered.
Similar content being viewed by others
References
Craig WA. Does the dose matter? Clin Infect Dis. 2001;33(suppl 3):S233–7.
Saito A, Tanigawara Y, Watanabe A, Aoki N, Niki Y, Kohno S, et al. Open study of sitafloxacin in patients with respiratory tract infections—PK/PD study (in Japanese). Jpn J Chemother. 2008;56(S-1):63–80.
Yamaguchi K, Ohno A, Ishii Y, Tateda K, Iwata M, Kanda M, et al. In vitro susceptibilities to levofloxacin and various antibacterial agents of 18,639 clinical isolates obtained from 77 centers in 2004 (in Japanese). Jpn J Antibiot. 2006;59:428–51.
Yamaguchi K, Ohno A, Ishii Y, Tateda K, Iwata M, Kanda M, et al. In vitro susceptibilities to levofloxacin and various antibacterial agents of 12,919 clinical isolates obtained from 72 centers in 2007 (in Japanese). Jpn J Antibiot. 2009;62:346–70.
Wikler MA, Low DA, Cookerill FR, Sheehan DJ, Craig WA, Tenover FC, et al. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically: approved standard. Seventh edition. CLSI (NCCLS). 2006;26:M7–A7.
Blondeau JM, Zhao X, Hansen G, Drlica K. Mutant prevention concentrations of fluoroquinolones for clinical isolates of Streptococcus pneumoniae. Antimicrob Agents Chemother. 2001;45:433–8.
Saito A, Miki F, Oizumi K, Rikitomi N, Watanabe A, Koga H, et al. Clinical evaluation methods for new antimicrobial agents to treat respiratory infections: report of the Committee for the Respiratory System, Japan Society of Chemotherapy. J Infect Chemother. 1999;5:110–23.
Japanese Society of Chemotherapy. Criteria for assessment of adverse reactions and abnormal laboratory values associated with antimicrobial agents in study subjects (in Japanese). Jpn J Chemother. 1991;39:687–9.
Japanese Society of Chemotherapy. About the partial modification of ‘‘Criteria for assessment of adverse reactions and abnormal laboratory values associated with antimicrobial agents in study subjects” (in Japanese). Jpn J Chemother. 1995;43 (opening article).
Mouton JW, Dudley MN, Cars O, Derendorf H, Drusano GL. Standardization of pharmacokinetic/pharmacodynamic (PK/PD) terminology for anti-infective drugs. Int J Antimicrob Agents. 2002;19:355–8.
Matsumoto T, Uchino K, Yamaguchi H, et al. Study on the safety and efficacy of sitafloxacin: results of the use-results survey (in Japanese). Jpn J Antibiot. 2011;64:319–37.
Hsueh PR. Study for monitoring antimicrobial resistance trends (SMART) in the Asia-Pacific region, 2002–2010. Int J Antimicrob Agents. 2012;40(S1):S1–3.
Tiengrim S, Phiboonbanakit D, Thunyaharn S, Tantisiriwat W, Santiwatanakul S, Susaengrat W, et al. Comparative in vitro activity of sitafloxacin against bacteria isolated from Thai patients with urinary tract infections and lower respiratory tract infections. J Med Assoc Thai. 2012;95(S2):S6–17.
Yamaguchi K, Ohno A, Ishii Y, Tateda K, Iwata M, Akizawa H, et al. In vitro susceptibilities to levofloxacin and various antibacterial agents of 12,866 clinical isolates obtained from 72 centers in 2010 (in Japanese). Jpn J Antibiot. 2012;65:181–206.
Nakashima M, Uematsu T, Kosuge K, Umemura K, Hakusui H, Tanaka M. Pharmacokinetics and tolerance of DU-6859a, a new fluoroquinolone, after single and multiple oral doses in healthy volunteers. Antimicrob Agents Chemother. 1995;39:170–4.
Keating GM. Sitafloxacin: in bacterial infections. Drugs. 2011;71(6):731–44.
Kawada Y, Ishihara S, Matsui T, Tsugawa M, Matsumoto T, Watanabe K, et al. Comparative study on sitafloxacin and levofloxacin in complicated urinary tract infections (in Japanese). Jpn J Chemother. 2008;56(S-1):81–91.
Kawada Y, Yasuda M, Tanaka K, Monden K, Akasaka S, Egashira T, et al. Dose-comparative study of sitafloxacin in complicated urinary tract infections (in Japanese). Jpn J Chemother. 2008;56(S-1):92–102.
Baba S, Suzuki K, Yamanaka N, Yamashita H, Kurono Y, Hori S. Efficacy and safety of sitafloxacin in patients with otorhinolaryngological infections and its tissue distribution in the otorhinolaryngological field (in Japanese). Jpn J Chemother. 2008;56(S-1):110–20.
Sasaki J, Hori S. Oral tissue distribution, efficacy, and safety of sitafloxacin in patients with dentistry and oral surgery infection (in Japanese). Jpn J Chemother. 2008;56(S-1):121–9.
Kawada Y, Matsumoto T, Onodera S, Kaku M, Hori S. Clinical study of sitafloxacin in male nongonococcal urethritis (in Japanese). Jpn J Chemother. 2008;56(S-1):130–8.
Matsuda S, Noguchi M, Yasuda J, Hori S. Clinical study of sitafloxacin in treatment of cervicitis with Chlamydia trachomatis (in Japanese). Jpn J Chemother. 2008;56(S-1):139–45.
Acknowledgments
Based on the results of this study, the clinical use of 100 mg once daily was approved, in addition to the original dose regimen, in Japan in August 2010. This work received financial support from Daiichi Sankyo Co., Ltd., Tokyo, Japan. We thank Daiichi Sankyo Co., Ltd., Tokyo, Japan, for providing editorial assistance.
Conflict of interest
Dr. Kohno has received honoraria for speaking and grant support from Daiichi Sankyo Co., Ltd. Dr. Niki has received speaker’s honoraria and an endowed chair from Daiichi Sankyo Co., Ltd. Dr. Yanagihara has received speaker’s honoraria from Daiichi Sankyo Co., Ltd. Dr. Kaku has received speaker’s honoraria from Daiichi Sankyo Co., Ltd. Dr. Watanabe has received speaker’s honoraria and an endowed chair from Daiichi Sankyo Co., Ltd. Dr. Aoki has received speaker’s honoraria from Daiichi Sankyo Co., Ltd. Dr. Hori has received speaker’s honoraria from Daiichi Sankyo Co., Ltd. Dr. Fujita has received speaker’s honoraria and grant support from Daiichi Sankyo Co., Ltd. All other authors report no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kohno, S., Niki, Y., Kadota, Ji. et al. Clinical dose findings of sitafloxacin treatment: pharmacokinetic–pharmacodynamic analysis of two clinical trial results for community-acquired respiratory tract infections. J Infect Chemother 19, 486–494 (2013). https://doi.org/10.1007/s10156-012-0543-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10156-012-0543-z