Abstract
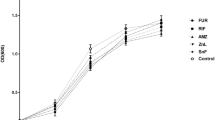
Using β-lactamase-negative ampicillin (ABPC)-susceptible (BLNAS) and β-lactamase-negative ABPC-resistant (BLNAR) nontypeable Haemophilus influenzae (NTHi) strains isolated from otological patients, colony biofilm was prepared on membrane filter substrates. Bactericidal activities of garenoxacin (GRNX), levofloxacin (LVFX), cefditoren (CDTR), and clavulanic acid/amoxicillin (CVA/AMPC) were examined by counting viable cells after drug exposure to biofilm cells for 6 and 24 h and by observation under a scanning electron microscope (SEM). After exposure of biofilm to the 100-fold MIC of GRNX or LVFX for 24 h, GRNX and LVFX showed potent bactericidal activity (∆log10 CFU/ml, ≥5.1). In this case, the drug-exposure AUC, exposure concentration × 24 μg h/ml, was 64–128 % for GRNX and 121 % for LVFX of free AUC at the clinical dosage in humans, respectively. CDTR and CVA/AMPC at 100-fold MIC exhibited little bactericidal activity against biofilm cells. Under an SEM, after exposure of BLNAS and BLNAR biofilms to GRNX or LVFX, most of the biofilm matrices were transformed. Quinolones such as GRNX show potent bactericidal activity against biofilm-forming NTHi at the usual clinical dosage.

Similar content being viewed by others
References
Bluestone CD. Clinical course, complications and sequelae of acute otitis media. Pediatr Infect Dis J. 2000;19:S37–46.
Kalcioglu MT, Durmaz B, Aktas E, Ozturan O, Durmaz R. Bacteriology of chronic maxillary sinusitis and normal maxillary sinuses: using culture and multiplex polymerase chain reaction. Am J Rhinol. 2003;17:143–7.
Murphy TF, Brauer AL, Schiffmacher AT, Sethi S. Persistent colonization by Haemophilus influenzae in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2004;170:266–72.
Hall-Stoodley L, Hu FZ, Gieseke A, Nistico L, Nguyen D, Hayes J, et al. Direct detection of bacterial biofilms on the middle-ear mucosa of children with chronic otitis media. JAMA. 2006;296:202–11.
Faden H, Brodsky L, Bernstein J, Stanievich J, Krystofik D, Shuff C, et al. Otitis media in children: local immune response to nontypeable Haemophilus influenzae. Infect Immun. 1989;57:3555–9.
Bakaletz LO. Bacterial biofilms in otitis media: evidence and relevance. Pediatr Infect Dis J. 2007;26:S17–9.
Sanbongi Y, Suzuki T. Osaki Y, Senju N, Ida T, Ubukata K. Molecular evolution of β-lactam-resistant Haemophilus influenzae: 9-year surveillance of penicillin-binding protein 3 mutations in isolates from Japan. Antimicrob Agents Chemother 2006;50:2487–2492.
García-Cobos S, Campos J, Lázaro E, Román F, Cercenado E, Gárcia-Rey C, et al. Ampicillin-resistant non-β-lactamase-producing Haemophilus influenzae in Spain: recent emergence of clonal isolates with increased resistance to cefotaxime and cefixime. Antimicrob Agents Chemother. 2007;51:2564–73.
American Academy of Pediatrics Subcommittee on Management of Acute Otitis Media. Diagnosis and management of acute otitis media. Pediatrics. 2004;113:1451–65.
Grubb MS, Spaugh DC. Treatment failure, recurrence, and antibiotic prescription rates for different acute otitis media treatment methods. Clin Pediatr (Phila). 2010;49:970–5.
Suzuki K, Kurono Y, Kobayashi T, Nishimura T, Baba S, Harabuchi Y, et al. Antimicrobial susceptibility surveillance of recent isolates from otorhinolaryngological infections to garenoxacin and other antimicrobial drugs (in Japanese). Jpn J Antibiot. 2009;62:71–8.
Jones RN, Fritsche TR, Sader HS, Stilwell MG. Activity of garenoxacin, an investigational des-F(6)-quinolone, tested against pathogens from community-acquired respiratory tract infections, including those with elevated or resistant-level fluoroquinolone MIC values. Diagn Microbiol Infect Dis. 2007;58:9–17.
Gajjar DA, Bello A, Ge Z, Christopher L, Grasela DM. Multiple-dose safety and pharmacokinetics of oral garenoxacin in healthy subjects. Antimicrob Agents Chemother. 2003;47:2256–63.
Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Nineteenth Informational Supplement. M100-S21. Wayne, PA: CLSI; 2011.
Roveta S, Schito AM, Marchese A, Schito GC. Activity of moxifloxacin on biofilms produced in vitro by bacterial pathogens involved in acute exacerbations of chronic bronchitis. Int J Antimicrob Agents. 2007;30:415–21.
Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically. Approved Standard. Eighth Edition. M07-A8. Wayne, PA: CLSI. 2009.
Webster P, Wu S, Gomez G, Apicella M, Plaut AG, St. Geme JW 3rd. Distribution of bacterial proteins in biofilms formed by non-typeable Haemophilus influenzae. J Histochem Cytochem. 2006;54:829–42.
Bello A, Hollenbaugh D, Gajjar D, Christopher L, Grasela D. In: Abstracts of the 41st Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC). Washington, DC: ASM Press; 2001 [Abstract A-45]
Chien SC, Rogge MC, Gisclon LG, Curtin C, Wong F, Natarajan J, et al. Pharmacokinetic profile of levofloxacin following once-daily 500-milligram oral or intravenous doses. Antimicrob Agents Chemother. 1997;41:2256–60.
Zeitlinger MA, Dehghanyar P, Mayer BX, Schenk BS, Neckel U, Heinz G, et al. Relevance of soft-tissue penetration by levofloxacin for target site bacterial killing in patients with sepsis. Antimicrob Agents Chemother. 2003;47:3548–53.
National Committee for Clinical Laboratory Standards. Methods for Determining Bactericidal Activity of Antimicrobial Agents; Approved Guideline. M26-A19 (18). Wayne, PA: National Committee for Clinical Laboratory Standards; 1999.
Post JC, Hiller NL, Nistico L, Stoodley P, Ehrlich GD. The role of biofilms in otolaryngologic infections: update 2007. Curr Opin Otolaryngol Head Neck Surg. 2007;15:347–51.
Anderson GG, O’Toole GA. Innate and induced resistance mechanisms of bacterial biofilms. Curr Top Microbiol Immunol. 2008;322:85–105.
Stewart PS, Costerton JW. Antibiotic resistance of bacteria in biofilms. Lancet. 2001;358:135–8.
Grijalva CG, Nuorti JP, Griffin MR. Antibiotic prescription rates for acute respiratory tract infections in US ambulatory settings. JAMA. 2009;302:758–66.
Moriyama S, Hotomi M, Shimada J, Billal DS, Fujihara K, Yamanaka N. Formation of biofilm by Haemophilus influenzae isolated from pediatric intractable otitis media. Auris Nasus Larynx. 2009;36:525–31.
Baba S, Suzuki K, Yamanaka N, Yajin K. Clinical phase III open-label study of oral garenoxacin in patients with otrhinolaryngological infection (in Japanese). Jpn J Chemother. 2007;55:194–205.
Neu T, Swerhone GD, Lawrence JR. Assessment of lectin-binding analysis for in situ detection of glycoconjugates in biofilm systems. Microbiology. 2001;147:299–313.
Donlan RM, Costerton JW. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev. 2002;15:167–93.
Spoering AL, Lewis K. Biofilms and planktonic cells of Pseudomonas aeruginosa have similar resistance to killing by antimicrobials. J Bacteriol. 2001;183:6746–51.
Poulsen LK, Ballard G, Stahl DA. Use of rRNA fluorescence in situ hybridization for measuring the activity of single cells in young and established biofilms. Appl Environ Microbiol. 1993;59:1354–60.
Kaji C, Watanabe K, Apicella MA, Watanabe H. Antimicrobial effect of fluoroquinolones for the eradication of nontypeable Haemophilus influenzae isolates within biofilms. Tohoku J Exp Med. 2008;214:121–8.
Hong W, Mason K, Jurcisek J, Novotny L, Bakaletz LO, Swords WE. Phosphorylcholine decreases early inflammation and promotes the establishment of stable biofilm communities of nontypeable Haemophilus influenzae strain 86–028NP in a chinchilla model of otitis media. Infect Immun. 2007;75:958–65.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Takahata, M., Sugiura, Y., Shinmura, Y. et al. Bactericidal activity of garenoxacin against in vitro biofilm formed by nontypeable Haemophilus influenzae . J Infect Chemother 19, 441–446 (2013). https://doi.org/10.1007/s10156-012-0502-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10156-012-0502-8