Abstract
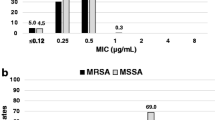
Glycopeptide antibiotics, such as vancomycin and teicoplanin, have been used worldwide to treat infection caused by methicillin-resistant Staphylococcus aureus (MRSA). Generic teicoplanin products were manufactured by many companies in 2009. We investigated the susceptibility of 147 MRSA strains to brand-name teicoplanin (TEIC-1) and seven generic products (TEIC-2 to TEIC-8). The MIC90 of generic TEIC-5 and TEIC-7 was 8 μg/ml whereas that of TEIC-1 and other generic products was 4 μg/ml. The potency equivalent of generic TEIC-5 and TEIC-7 was lower than that of TEIC-1, and TEIC content (%) per potency equivalent (200 mg) in a vial of these two generic products varied greatly compared with the other products. Although the potency equivalent of the TEIC used in this study was within the range stipulated in the Japanese Pharmacopeia, these results showed that the potency equivalent and susceptibility of two of the seven generic products differed from that of TEIC-1. The predicted AUC0–72 value of those two generic products was 84–85% in comparison with that of TEIC-1. Among generic drugs, there may be products whose antimicrobial effect is not equal to that of the brand teicoplanin.

Similar content being viewed by others
References
Mastoraki E, Michalopoulos A, Kriaras I, Mouchtouri E, Falagas ME, Karatza D, et al. Incidence of postoperative infections in patients undergoing coronary artery bypass grafting surgery receiving antimicrobial prophylaxis with original and generic cefuroxime. J Infect. 2008;56:35–9.
Rodriguez CA, Agudelo M, Catano JC, Zuluaga AF, Vesga O. Potential therapeutic failure of generic vancomycin in a liver transplant patient with MRSA peritonitis and bacteremia. J Infect. 2009;59:277–80.
Fujimura S, Fuse K, Kikuchi T, Gomi K, Tokue Y, Watanabe A. In vitro susceptibility of clinical isolates of methicillin-resistant Staphylococcus aureus (MRSA) to manufactured generic drugs compared with the brand vancomycin. Int J Antimicrob Agents. 2008;31:391–2.
Clinical and Laboratory Standards Institute (2009) Perfomance standards for antimicrobial susceptibility testing, 19th edn. Approved standard M100-S19. CLSI, Wayne, PA.
Ministry of Health, Labour and Welfare (2006) Teicoplanin. The Japanese pharmacopoeia, 15th edn. Ministry of Health, Labour and Welfare, Tokyo, Japan, pp 733–735.
Nakayama K, Gemma H, Kaibara A, Niwa T. Population pharmacokinetics of teicoplanin in adult patients. Jpn J Chemother. 2006;54:1–6.
Kuti JL, Kiffer CRV, Mendes CMF, Nicolau DP. Pharmacodynamic comparison of linezolid, teicoplanin and vancomycin against clinical isolates of Staphylococcus aureus and coagulase-negative staphylococci collected from hospitals in Brazil. Clin Microbiol Infect. 2008;14:116–23.
Schito GC, Keenan MH. Predicting the clinical efficacy of generic formulations of ceftriaxone. J Chemother. 2005;17(suppl 2):33–40.
Martin M, Quilici S, File T, Garau J, Kureishi A, Kubin M. Cost-effectiveness of empirical prescribing of antimicrobials in community-acquired pneumonia in three countries in the presence of resistance. J Antimicrob Chemother. 2007;59:977–89.
Japan Antibiotics Research Association (2000) Teicoplanin for injection. Requirements for antibiotic products of Japan. Jiho Co., Tokyo, Japan, p II-39.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Fujimura, S., Fuse, K., Takane, H. et al. Antibacterial effects of brand-name teicoplanin and generic products against clinical isolates of methicillin-resistant Staphylococcus aureus . J Infect Chemother 17, 30–33 (2011). https://doi.org/10.1007/s10156-010-0094-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10156-010-0094-0