Abstract
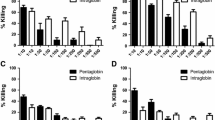
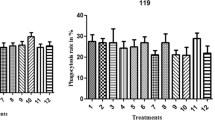
We have used the ability of opsonized bacteria to stimulate luminol-enhanced chemiluminescence (CL) of human polymorphonuclear leukocytes (PMN) to examine the opsonic capabilities of commercially available human intravenous immunoglobulin (IVIG) preparations. The method was tested against 14 strains of drug-resistant gram-positive bacteria (including methicillin-resistant Staphylococcus aureus, hetero-vancomycin-resistant S. aureus, vancomycin-resistant enterococci, penicillin-resistant Streptococcus pneumoniae), and 23 strains of gram-negative bacteria (including extended-spectrum β-lactamase-producing bacteria, metallo-β-lactamase-producing bacteria, β-lactamase-negative ampicillin-resistant Haemophilus influenzae). An Fc-intact IVIG preparation treated with polyethylene glycol (PEG) was evaluated for opsonization effectiveness against these bacteria in vitro. The opsonization of these organisms was enhanced by an Fc-intact IVIG, and the opsonic activity was dose dependent. A pepsin-treated IVIG preparation exhibited poor opsonic activity for all bacteria tested. These results suggest that Fc-intact IVIG, which augments opsonic activity against various drug-resistant bacteria, will be a useful addition to the treatment of severe bacterial infections in immunocompromised patients with impaired serum opsonic capacity.
Article PDF
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ono, Y., Ito, T., Watanabe, T. et al. Opsonic activity assessment of human intravenous immunoglobulin preparations against drug-resistant bacteria. J Infect Chemother 10, 234–238 (2004). https://doi.org/10.1007/s10156-004-0325-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10156-004-0325-3