Abstract
Background
To mitigate pelvic wound issues following perineal excision of rectal or anal cancer, a number of techniques have been suggested as an alternative to primary closure. These methods include the use of a biological/dual mesh, omentoplasty, muscle flap, and/or pelvic peritoneum closure. The aim of this network analysis was to compare all the available surgical techniques used in the attempt to mitigate issues associated with an empty pelvis.
Methods
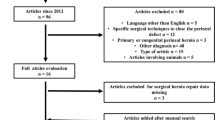
An electronic systematic search using MEDLINE databases (PubMed), EMBASE, and Web of Science was performed (Last date of research was March 15th, 2023). Studies comparing at least two of the aforementioned surgical techniques for perineal wound reconstruction during abdominoperineal resection, pelvic exenteration, or extra levator abdominoperineal excision were included. The incidence of primary healing, complication, and/or reintervention for perineal wound were evaluated. In addition, the overall incidence of perineal hernia was assessed.
Results
Forty-five observational studies and five randomized controlled trials were eligible for inclusion reporting on 146,398 patients. All the surgical techniques had a comparable risk ratio (RR) in terms of primary outcomes. The pooled network analysis showed a lower RR for perineal wound infection when comparing primary closure (RR 0.53; Crl 0.33, 0.89) to muscle flap. The perineal wound dehiscence RR was lower when comparing both omentoplasty (RR 0.59; Crl 0.38, 0.95) and primary closure (RR 0.58; Crl 0.46, 0.77) to muscle flap.
Conclusions
Surgical options for perineal wound closure have evolved significantly over the last few decades. There remains no clear consensus on the “best” option, and tailoring to the individual remains a critical factor.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249
Heald RJ, Ryall RD (1986) Recurrence and survival after total mesorectal excision for rectal cancer. Lancet 1(8496):1479–1482
Kamrava A, Mahmoud NN (2013) Prevention and management of nonhealing perineal wounds. Clin Colon Rectal Surg 26(2):106–111
Spinelli A, Foppa C, Carvello M et al (2021) Transanal transection and single-stapled anastomosis (TTSS): a comparison of anastomotic leak rates with the double-stapled technique and with transanal total mesorectal excision (TaTME) for rectal cancer. Eur J Surg Oncol 47(12):3123–3129
Blok RD, de Jonge J, de Koning MA et al (2019) Propensity score adjusted comparison of pelviperineal morbidity with and without omentoplasty following abdominoperineal resection for primary rectal cancer. Dis Colon Rectum 62(8):952–959
Sayers AE, Patel RK, Hunter IA (2015) Perineal hernia formation following extralevator abdominoperineal excision. Colorectal Dis 17(4):351–355
Blok RD, Musters GD, Borstlap WAA, Buskens CJ, Bemelman WA, Tanis PJ (2018) Snapshot study on the value of omentoplasty in abdominoperineal resection with primary perineal closure for rectal cancer. Ann Surg Oncol 25(3):729–736
Howell AM, Jarral OA, Faiz O, Ziprin P, Darzi A, Zacharakis E (2013) How should perineal wounds be closed following abdominoperineal resection in patients post radiotherapy–primary closure or flap repair? Best evidence topic (BET). Int J Surg 11(7):514–517
Hawkins AT, Albutt K, Wise PE et al (2018) Abdominoperineal resection for rectal cancer in the twenty-first century: indications, techniques, and outcomes. J Gastrointest Surg 22(8):1477–1487
Chan S, Miller M, Ng R et al (2010) Use of myocutaneous flaps for perineal closure following abdominoperineal excision of the rectum for adenocarcinoma. Colorectal Dis 12(6):555–560
Foster JD, Pathak S, Smart NJ et al (2012) Reconstruction of the perineum following extralevator abdominoperineal excision for carcinoma of the lower rectum: a systematic review. Colorectal Dis 14(9):1052–1059
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700
Sterne JA, Hernan MA, Reeves BC et al (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919
Higgins JP, Altman DG, Gøtzsche PC et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Mills EJ, Thorlund K, Ioannidis JP (2013) Demystifying trial networks and network meta-analysis. BMJ 346:f2914
Rausa E, Kelly ME, Sgroi G et al (2019) Quality of life following ostomy reversal with purse-string vs linear skin closure: a systematic review. Int J Colorectal Dis 34(2):209–216
Warn DE, Thompson SG, Spiegelhalter DJ (2002) Bayesian random effects meta-analysis of trials with binary outcomes: methods for the absolute risk difference and relative risk scales. Stat Med 21(11):1601–1623
Aiolfi A, Asti E, Siboni S et al (2019) Impact of spleen-preserving total gastrectomy on postoperative infectious complications and 5-year overall survival: systematic review and meta-analysis of contemporary randomized clinical trials. Curr Oncol 26(2):e202–e209
Dias S, Sutton AJ, Ades AE, Welton NJ (2013) Evidence synthesis for decision making 2: a generalized linear modeling framework for pairwise and network meta-analysis of randomized controlled trials. Med Decis Making 33(5):607–617
Friede T, Rover C, Wandel S, Neuenschwander B (2017) Meta-analysis of few small studies in orphan diseases. Res Synth Methods 8(1):79–91
Turner RM, Davey J, Clarke MJ, Thompson SG, Higgins JP (2012) Predicting the extent of heterogeneity in meta-analysis, using empirical data from the cochrane database of systematic reviews. Int J Epidemiol 41(3):818–827
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560
Smith BJ (2007) boa: an R package for MCMC output convergence assessment and posterior inference. J Stat Softw 21:1–37. https://doi.org/10.18637/jss.v021.i11
Salanti G, Del Giovane C, Chaimani A, Caldwell DM, Higgins JP (2014) Evaluating the quality of evidence from a network meta-analysis. PLoS One 9(7):e99682
Plummer M (2003) JAGS: a program for analysis of Bayesian graphical models using Gibbs sampling. Proceedings of the 3rd International Workshop on Distributed Statistical Computing; Vienna, Austria. pp 20–22
R Core Team. R: A language and environment for statistical computing. R foundation for statistical computing; Vienna, Austria. http://www.R-project.org/. Accessed 5 Apr 2018.
Hultman CS, Sherrill MA, Halvorson EG et al (2010) Utility of the omentum in pelvic floor reconstruction following resection of anorectal malignancy: patient selection, technical caveats, and clinical outcomes. Ann Plast Surg 64(5):559–562
John H, Buchmann P (1991) Improved perineal wound healing with the omental pedicle graft after rectal excision. Int J Colorectal Dis 6(4):193–196
Poston GJ, Smith SR, Baker WN (1991) Retrocolic pelvic omentoplasty in abdominoperineal excision of the rectum. Ann R Coll Surg Engl 73(4):229–232
Shen Y, Yang T, Zeng H, Meng W, Wang Z (2021) Efficacy of pelvic peritoneum closure after laparoscopic extralevator abdominoperineal excision for rectal cancer. J Gastrointest Surg 25(10):2668–2678
Yan X, Su H, Zhang S et al (2021) Pelvic peritoneum closure reduces postoperative complications of laparoscopic abdominoperineal resection: 6-year experience in single center. Surg Endosc 35(1):406–414
Althumairi AA, Canner JK, Ahuja N, Sacks JM, Safar B, Efron JE (2016) Time to chemotherapy after abdominoperineal resection: comparison between primary closure and perineal flap reconstruction. World J Surg 40(1):225–230
Althumairi AA, Canner JK, Gearhart SL et al (2016) Risk factors for wound complications after abdominoperineal excision: analysis of the ACS NSQIP database. Colorectal Dis 18(7):O260–O266
Althumairi AA, Canner JK, Gearhart SL, Safar B, Sacks J, Efron JE (2016) Predictors of perineal wound complications and prolonged time to perineal wound healing after abdominoperineal resection. World J Surg 40(7):1755–1762
Butler CE, Gündeslioglu AO, Rodriguez-Bigas MA (2008) Outcomes of immediate vertical rectus abdominis myocutaneous flap reconstruction for irradiated abdominoperineal resection defects. J Am Coll Surg 206(4):694–703
Chessin DB, Hartley J, Cohen AM et al (2005) Rectus flap reconstruction decreases perineal wound complications after pelvic chemoradiation and surgery: a cohort study. Ann Surg Oncol 12(2):104–110
Curran T, Poylin V, Nagle D (2016) Real world dehiscence rates for patients undergoing abdominoperineal resection with or without myocutaneous flap closure in the national surgical quality improvement project. Int J Colorectal Dis 31(1):95–104
Davidge KM, Raghuram K, Hofer SO et al (2014) Impact of flap reconstruction on perineal wound complications following ablative surgery for advanced and recurrent rectal cancers. Ann Surg Oncol 21(6):2068–2073
Ferenschild FT, Vermaas M, Hofer SO, Verhoef C, Eggermont AM, de Wilt JH (2005) Salvage abdominoperineal resection and perineal wound healing in local recurrent or persistent anal cancer. World J Surg 29(11):1452–1457
Han JG, Wang ZJ, Qian Q et al (2014) A prospective multicenter clinical study of extralevator abdominoperineal resection for locally advanced low rectal cancer. Dis Colon Rectum 57(12):1333–1340
Christensen HK, Nerstrøm P, Tei T, Laurberg S (2011) Perineal repair after extralevator abdominoperineal excision for low rectal cancer. Dis Colon Rectum 54(6):711–717
Han JG, Wang ZJ, Gao ZG et al (2019) Perineal wound complications after extralevator abdominoperineal excision for low rectal cancer. Dis Colon Rectum 62(12):1477–1484
Jacombs AS, Rome P, Harrison JD, Solomon MJ (2013) Assessment of the selection process for myocutaneous flap repair and surgical complications in pelvic exenteration surgery. Br J Surg 100(4):561–567
Khoo AK, Skibber JM, Nabawi AS et al (2001) Indications for immediate tissue transfer for soft tissue reconstruction in visceral pelvic surgery. Surgery 130(3):463–469
Kokosis G, Sun Z, Avashia YJ et al (2017) V-Y fasciocutaneous flap closure technique is a safe and efficacious alternative to primary closure of the perineal wound following abdominoperineal resection. Am J Surg 213(2):371–376
Lefevre JH, Parc Y, Kernéis S et al (2009) Abdomino-perineal resection for anal cancer: impact of a vertical rectus abdominis myocutaneus flap on survival, recurrence, morbidity and wound healing. Ann Surg 250(5):707–711
O’Dowd V, Burke JP, Condon E et al (2014) Vertical rectus abdominis myocutaneous flap and quality of life following abdominoperineal excision for rectal cancer: a multi-institutional study. Tech Coloproctol 18(10):901–906
Radice E, Nelson H, Mercill S, Farouk R, Petty P, Gunderson L (1999) Primary myocutaneous flap closure following resection of locally advanced pelvic malignancies. Br J Surg 86(3):349–354
Sancho-Muriel J, Ocaña J, Cholewa H et al (2020) Biological mesh reconstruction versus primary closure for preventing perineal morbidity after extralevator abdominoperineal excision: a multicentre retrospective study. Colorectal Dis 22(11):1714–1723
Sheckter CC, Shakir A, Vo H, Tsai J, Nazerali R, Lee GK (2016) Reconstruction following abdominoperineal resection (APR): indications and complications from a single institution experience. J Plast Reconstr Aesthet Surg 69(11):1506–1512
Shibata D, Hyland W, Busse P et al (1999) Immediate reconstruction of the perineal wound with gracilis muscle flaps following abdominoperineal resection and intraoperative radiation therapy for recurrent carcinoma of the rectum. Ann Surg Oncol 6(1):33–37
Wang ED, Conkling N, Xu X et al (2015) Perineal flap reconstruction following oncologic anorectal extirpation: an outcomes assessment. Plast Reconstr Surg 135(1):176e–184e
Wang YL, Zhang X, Mao JJ et al (2018) Application of modified primary closure of the pelvic floor in laparoscopic extralevator abdominal perineal excision for low rectal cancer. World J Gastroenterol 24(30):3440–3447
Woodfield J, Hulme-Moir M, Ly J (2017) A comparison of the cost of primary closure or rectus abdominis myocutaneous flap closure of the perineum after abdominoperineal excision. Colorectal Dis 19(10):934–941
Kapoor V, Cole J, Isik FF, Sinanan M, Flum D (2005) Does the use of a flap during abdominoperineal resection decrease pelvic wound morbidity? Am Surg 71(2):117–122
Hardt J, Mai S, Weiß C, Kienle P, Magdeburg J (2016) Abdominoperineal resection and perineal wound healing in recurrent, persistent, or primary anal carcinoma. Int J Colorectal Dis 31(6):1197–1203
Peacock O, Pandya H, Sharp T et al (2012) Biological mesh reconstruction of perineal wounds following enhanced abdominoperineal excision of rectum (APER). Int J Colorectal Dis 27(4):475–482
Hannes S, Reinisch A, Bechstein WO, Habbe N (2016) Salvage abdominoperineal excisions in recurrent anal cancer–impact of different reconstruction techniques on outcome, morbidity, and complication rates. Int J Colorectal Dis 31(3):653–659
Chaudhry A, Oliver JD, Vyas KS et al (2019) Comparison of outcomes in oncoplastic pelvic reconstruction with VRAM versus omental flaps: a large cohort analysis. J Reconstr Microsurg 35(6):425–429
Zeiderman MR, Nuño M, Sahar DE, Farkas LM (2021) Trends in flap reconstruction of pelvic oncologic defects: analysis of the national inpatient sample. J Plast Reconstr Aesthet Surg 74(9):2085–2094
Billig JI, Hsu JJ, Zhong L, Wang L, Chung KC, Kung TA (2019) Comparison of effective cost and complications after abdominoperineal resection: primary closure versus flap reconstruction. Plast Reconstr Surg 144(5):866e-e875
Nichols DS, Satteson E, Harbor P, DeFranzo A, David L, Thompson JT 2nd (2020) Factors associated with the use of vertical rectus abdominus myocutaneous flap reconstruction following abdominoperineal resection for anorectal cancer. J Surg Oncol 122(5):923–927
Spasojevic M, Mariathasan AB, Goscinski M et al (2018) Vertical rectus abdominis musculocutaneous flap repair improves perineal wound healing after abdominoperineal resection for irradiated locally advanced rectal cancer. Ann Surg Oncol 25(5):1357–1365
Sharabiany S, van Dam JJW, Sparenberg S et al (2021) A comparative multicentre study evaluating gluteal turnover flap for wound closure after abdominoperineal resection for rectal cancer. Tech Coloproctol 25(10):1123–1132
Cataneo JL, Mathis SA, Faqihi S et al (2023) Comparison of perineal closure techniques after abdominoperineal resections for carcinoma of the anus. Am Surg 89(2):238–246
Cataneo JL, Mathis SA, Del Valle DD et al (2023) Outcomes of perineal wound closure techniques after abdominoperineal resections in rectal cancer: an NSQIP propensity score matched study. J Plast Surg Hand Surg 57(1–6):399–407
Nagata M, Matsuda T, Hasegawa H et al (2020) Usefulness of omentoplasty to reduce perineal wound complications in abdominoperineal resection after neoadjuvant chemoradiotherapy. Anticancer Res 40(11):6539–6543
Gultekin S, Gartrell R, Lu L et al (2022) Outcomes of perineal reconstruction with inferior gluteal artery myocutaneous flaps and primary closure following abdominoperineal resection. ANZ J Surg 92(11):2968–2973
Musters GD, Klaver CEL, Bosker RJI et al (2017) Biological mesh closure of the pelvic floor after extralevator abdominoperineal resection for rectal cancer: a multicenter randomized controlled trial (the BIOPEX-study). Ann Surg 265(6):1074–1081
Irvin TT, Goligher JC (1975) A controlled clinical trial of three different methods of perineal wound management following excision of the rectum. Br J Surg 62(4):287–291
Touny A, Othman H, Maamoon S, Ramzy S, Elmarakby H (2014) Perineal reconstruction using pedicled vertical rectus abdominis myocutaneous flap (VRAM). J Surg Oncol 110(6):752–757
Cui J, Ma JP, Xiang J et al (2009) Prospective study of reconstructing pelvic floor with GORE-TEX Dual Mesh in abdominoperineal resection. Chin Med J (Engl) 122(18):2138–2141
Blok RD, Sharabiany S, Stoker J et al (2022) Cumulative 5-year results of a randomized controlled trial comparing biological mesh with primary perineal wound closure after extralevator abdominoperineal resection (BIOPEX-study). Ann Surg 275(1):e37–e44
Mjoli M, Sloothaak DA, Buskens CJ, Bemelman WA, Tanis PJ (2012) Perineal hernia repair after abdominoperineal resection: a pooled analysis. Colorectal Dis 14(7):e400–e406
Christian CK, Kwaan MR, Betensky RA, Breen EM, Zinner MJ, Bleday R (2005) Risk factors for perineal wound complications following abdominoperineal resection. Dis Colon Rectum 48(1):43–48
Thomas PW, Blackwell JEM, Herrod PJJ et al (2019) Long-term outcomes of biological mesh repair following extra levator abdominoperineal excision of the rectum: an observational study of 100 patients. Tech Coloproctol 23(8):761–767
Smith RL, Bohl JK, McElearney ST et al (2004) Wound infection after elective colorectal resection. Ann Surg 239(5):599–605
Burstein HJ, Krilov L, Aragon-Ching JB et al (2017) Clinical cancer advances 2017: annual report on progress against cancer from the American Society of Clinical Oncology. J Clin Oncol 35(12):1341–1367
Funding
This work was not supported.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10151_2023_2868_MOESM3_ESM.tif
Supplementary file3 Node-split analysis for A, Primary healing of perineal wound; B, perineal hernia; C, incidence of perineal complications; D, reintervention related to perineal wound; (TIF 341 KB)
10151_2023_2868_MOESM4_ESM.tiff
Supplementary file4 Network geometry for studies reporting E, incidence of perineal infection; F, abscess/collection; G, dehiscence; and H, development of chronic sinus (TIFF 213 KB)
10151_2023_2868_MOESM5_ESM.tiff
Supplementary file5 Forest plots of network meta-analysis estimates the risk ratio for E, incidence of perineal infection; F, abscess/collection; G, dehiscence; and H, development of chronic sinus (TIFF 4813 KB)
10151_2023_2868_MOESM6_ESM.tiff
Supplementary file6 A rank plot created using the rankogram function from the gemtc R package applied to the three surgical approaches illustrating empirical probabilities that each treatment is ranked first through fourth (left to right) for E, incidence of perineal infection; F, abscess/collection; G, dehiscence; and H, development of chronic sinus (TIFF 4565 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Riva, C.G., Kelly, M.E., Vitellaro, M. et al. A comparison of surgical techniques for perineal wound closure following perineal excision: a systematic review and network meta-analysis. Tech Coloproctol 27, 1351–1366 (2023). https://doi.org/10.1007/s10151-023-02868-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10151-023-02868-1