Abstract
Background
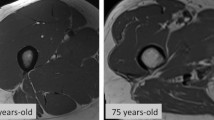
Sarcopenia is a poor prognostic factor in cancer patients. In recent years, there have been reports that serum creatinine and cystatin C (Cr/CysC) ratio is associated with sarcopenia. However, the prognostic value of the Cr/CysC ratio in biliary tract cancer is unclear. We evaluated the impact of the Cr/CysC ratio on sarcopenia and prognosis in biliary tract cancer.
Methods
We retrospectively reviewed the records of 190 patients with biliary tract cancer who had undergone surgical resection from January 2017 to March 2023. Frozen serum samples collected at the time of surgery were used to measure CysC. We calculated the Cr/CysC ratio and investigated the relationship with sarcopenia and the prognostic significance.
Results
We calculated the cutoff value of the Cr/CysC ratio for low skeletal muscle index (SMI) (< 42 cm2/m2 for males and < 38 cm2/m2 for females). The optimal cutoff value of the Cr/CysC ratio was 0.848. The low Cr/CysC ratio group was significantly associated with higher preoperative CRP and lower albumin, lower SMI, lower handgrip strength, and higher intramuscular adipose tissue content. In multivariate analysis, patients with a low Cr/CysC ratio showed poorer overall survival (hazard ratio 2.60, 95% confidence interval 1.07–6.29, p = 0.033), which was significantly worse than in those with a high Cr/CysC ratio.
Conclusions
In patients with biliary tract cancer, the Cr/CysC ratio showed weak correlation with sarcopenic indicators. However, the Cr/CysC ratio could be strong prognostic factor in biliary tract cancer.

Similar content being viewed by others
References
Ioka T, Shindo Y, Ueno M et al (2023) Current progress in perioperative chemotherapy for biliary tract cancer. Ann Gastroenterol Surg 7(4):565–571
Nakachi K, Ikeda M, Konishi M et al (2023) Adjuvant S-1 compared with observation in resected biliary tract cancer (JCOG1202, ASCOT): a multicentre, open-label, randomised, controlled, phase 3 trial. Lancet 401(10372):195–203
Simonsen C, Pieter DH, Eik DB et al (2018) Sarcopenia and postoperative complication risk in gastrointestinal surgical oncology: a meta-analysis. Ann Surg 268(1):58–69
Harimoto N, Shirabe K, Yamashita Y et al (2013) Sarcopenia as a predictor of prognosis in patients following hepatectomy for hepatocellular carcinoma. Br J Surg 100(11):1523–1530
Sugimura K, Miyata H, Kanemura T et al (2022) Impact of preoperative skeletal muscle mass and physical performance on short-term and long-term postoperative outcomes in patients with esophageal cancer after esophagectomy. Ann Gastroenterol Surg 6(5):623–632
Cruz-Jentoft AJ, Bahat G, Bauer J et al (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48(1):16–31
Chen LK, Liu LK, Woo J et al (2014) Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc 15(2):95–101
Filler G, Bokenkamp A, Hofmann W et al (2005) Cystatin C as a marker of GFR–history, indications, and future research. Clin Biochem 38(1):1–8
Tlemsani C, Durand JP, Raynard B et al (2022) Relationship between the creatinine/cystatin C ratio and muscle mass measured by CT-scan in cancer patients. Clin Nutr ESPEN 51:412–418
Yang J, Zhang T, Feng D et al (2019) A new diagnostic index for sarcopenia and its association with short-term postoperative complications in patients undergoing surgery for colorectal cancer. Colorectal Dis 21(5):538–547
Kashani LB, Frazee EN, Kukralova L et al (2017) Evaluating muscle mass by using markers of kidney function: development of the sarcopenia index. Crit Care Med 45(1):e23–e29
Magister S, Kos J (2013) Cystatins in immune system. J Cancer 4(1):45–56
Kos J, Krasovec M, Cimerman N et al (2000) Cysteine proteinase inhibitors stefin A, stefin B, and cystatin C in sera from patients with colorectal cancer: relation to prognosis. Clin Cancer Res 6(2):505–511
Wu J, Liang Y, Chen R et al (2022) Association of plasma cystatin C with all-cause and cause-specific mortality among middle-aged and elderly individuals: a prospective community-based cohort study. Sci Rep 12(1):22265
Harimoto N, Araki K, Yamanaka T et al (2022) The ratio of creatinine and cystatin C estimated glomerular filtration rates as a surrogate marker in patients with hepatocellular carcinoma undergoing hepatic resection. J Hepatobiliary Pancreat Sci 29(9):964–973
Tsukagoshi M, Watanabe A, Araki K et al (2023) Usefulness of serum creatinine and cystatin C ratio as a screening tool for predicting prognosis in patients with pancreatic cancer. Ann Gastroenterol Surg 7(5):784–792
Brierly JD, Gospodarowicz MK, Wittekind C (eds) (2017) TNM classification of malignant tumors, 8th edn. Wiley-Liss, New York
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Baumgartner RN, Koehler KM, Gallagher D et al (1998) Epidemiology of sarcopenia among the elderly in New Mexico. Am J Epidemiol 147(8):755–763
Nishikawa H, Shiraki M, Hisamatsu A et al (2016) Japan Society of Hepatology guidelines for sarcopenia in liver disease (1st edition): Recommendation from the working group for creation of sarcopenia assessment criteria. Hepatol Res 46(10):951–963
Kitajima Y, Hyogo H, Sumida Y et al (2013) Severity of non-alcoholic steatohepatitis is associated with substitution of adipose tissue in skeletal muscle. J Gastroenterol Hepatol 28(9):1507–1514
Chan AW, Chan SL, Wong GL et al (2015) Prognostic Nutritional Index (PNI) Predicts Tumor Recurrence of Very Early/Early Stage Hepatocellular Carcinoma After Surgical Resection. Ann Surg Oncol 22(13):4138–4148
Akaike H (1974) A new look at statistical model identification. IEEE Trans Automat Contr 19:716–23
Aldo JM, Andres DR, Judith MJ et al (2015) Inclusion of sarcopenia within MELD (MELD-Sarcopenia) and the prediction of mortality in patients with cirrhosis. Clin Transl Gastroenterol 6(7):e102
Nishikawa H, Shiraki M, Hisamitsu A et al (2021) Reduced handgrip strength predicts poorer survival in chronic liver diseases: a large multicenter study in Japan. Hepatol Res 51(9):957–967
Hirai K, Tanaka A, Homma T et al (2021) Serum creatinine/cystatin C ratio as a surrogate marker for sarcopenia in patients with chronic obstructive pulmonary disease. Clin Nutr 40:1274–1280
Jung CY, Kim HW, Han SH et al (2022) Creatinine-cystatin C ratio and mortality in cancer patients: a retrospective cohort study. J Cachexia Sarcopenia Muscle 13(4):2064–2072
Schmeusser BN, Biermann H, Nicaise EH et al (2023) Creatinine to cystatin-C ratio in renal cell carcinoma: a clinically pragmatic prognostic factor and sarcopenia biomarker. Oncologist. https://doi.org/10.1093/oncolo/oyad218
Tang T, Xie L, Hu S et al (2022) Serum creatinine and cystatin C-based diagnostic indices for sarcopenia in advanced non-small cell lung cancer. J Cachexia Sarcopenia Muscle 13(3):1800–1810
Kusunoki H, Tsuji S, Wada Y et al (2018) Relationship between sarcopenia and the serum creatinine/cystatin C ratio in Japanese rural community-dwelling older adults. J Cachexia Sarcopenia Muscle - Clin Rep 3:e00057
Mori J, Tanikawa C, Funauchi Y et al (2016) Cystatin C as a p53-inducible apoptotic mediator that regulates cathepsin L activity. Cancer Sci 107(3):298–306
Zeng Q, Zhao Y, Yang Y et al (2010) Expression of Cystatin C in human stomach neoplasms. Mol Med Rep 3(4):607–611
Lu S, Li S (2022) Association between preoperative serum Cystatin-C levels and postsurgical oncological prognosis in patients with PRCC: a retrospective cohort study. Cancer Med 11(22):4112–4121
Acknowledgements
We thank Mr. Takumi Nagasawa, Ms. Rumi Okazaki, Ms. Azusa Uchida, Ms. Kiyomi Nakajima of Gunma University Hospital laboratory section and Ms.Harumi Kanai and Ms. Yukiko Sudo for measuring cystatin C. We thank Carol Wilsom, PhD, from Edanz (http://jp.edanz.com/ac) for editing a draft of this manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
R.F., N.H., M.T., and K.S. contributed to the conception and design of this study. R.F., T.O., T.S., K.H., S.K., and K.H. contributed to the acquisition of data. R.F., N.H., N.I., and T.I. contributed to the analysis and interpretation of data. R.F., N.H., M.T., K.A., and K.S. contributed to the drafting of the manuscript. R.F., N.H., M.T., K.A., and K.S. contributed to the revision of the manuscript. All authors contributed to the final approval of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this article.
Ethical approval
The study was approved by the Gunma University Ethics Committee (HS2021-064) and complied with institutional guidelines and the Declaration of Helsinki. Patient consent for participation was obtained using the opt-out method.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10147_2024_2539_MOESM1_ESM.pdf
Supplementary file1 (PDF 816 KB)—Figure. 1 Correlation between Cr/CysC ratio and a SMI and b handgrip strength. The Cr/CysC ratio showed a very weak positive correlation with SMI and a weak positive correlation with grip strength. (SMI: R2 = 0.096, p < 0.001; handgrip strength: R2 = 0.20, p < 0.001). Abbreviations: Cr/CysC, creatinine and cystatin C; SMI, skeletal muscle index. Figure. 2 Kaplan–Meier curves for overall survival (OS) and recurrence-free survival (RFS) by the classifications of the creatinine and cystatin C (Cr/CysC) ratio: 0.839. a Patients in the low Cr/CysC ratio group (Cr/CysC ≤ 0.848) had significantly worse OS (p = 0.007) than those in the high Cr/CysC ratio group (Cr/CysC > 0.848). b Patients in the low Cr/CysC ratio group (Cr/CysC ≤ 0.848) had significantly worse RFS (p = 0.010) than those in the high Cr/CysC ratio group (Cr/CysC > 0.848). Abbreviations: Cr/CysC, creatinine and cystatin C. Figure. 3 Kaplan–Meier curves for overall survival (OS) and recurrence-free survival (RFS) by the classifications of the ratio of Cr and CysC estimated glomerular filtration rates (eGFRcre/eGFRcys) ratio: 1.044. a Patients in the high eGFRcre/eGFRcys ratio group (eGFRcre/eGFRcys ≥ 1.044) had significantly worse OS (p = 0.002) than those in the low eGFRcre/eGFRcys ratio group (eGFRcre/eGFRcys < 1.044). b Patients in the high eGFRcre/eGFRcys ratio group (eGFRcre/eGFRcys ≥ 1.044) had significantly worse RFS (p = 0.010) than those in the low eGFRcre/eGFRcys ratio group (eGFRcre/eGFRcys < 1.044). Abbreviations: eGFRcre/eGFRcys, ratio of Cr and CysC estimated glomerular filtration rates. Figure. 4 Kaplan–Meier curves for overall survival (OS) and recurrence-free survival (RFS) by the classifications of the creatinine and cystatin C (Cr/CysC) ratio in 107 of 190 patients who had skeletal muscle index (SMI) and handgrip strength data. Patients in the low Cr/CysC ratio group (Cr/CysC ≤ 0.848) tended to have poor OS and poor RFS, but there was no significant difference. Abbreviations: Cr/CysC, creatinine and cystatin C. Table 1. Univariate and multivariate analyses of the OS (Cr/CysC cutoff value: 0.839). Table 2. Univariate and multivariate analyses of the RFS (Cr/CysC cutoff value: 0.839). Table 3. Association between Cr/CysC ratios and sarcopenia. (Data for 107 patients measured both SMI and handgrip strength). Table 4. Association between Cr/CysC ratios and SMI.
About this article
Cite this article
Fukushima, R., Harimoto, N., Okuyama, T. et al. Impact of the serum creatinine and cystatin C ratio for prediction of sarcopenia and prognosis in biliary tract cancer. Int J Clin Oncol (2024). https://doi.org/10.1007/s10147-024-02539-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10147-024-02539-7