Abstract
Background
External auditory canal squamous cell carcinoma (EACSCC) is a rare form of malignant tumor. Due to the extremely limited understanding of the genomic landscape in EACSCC, the association between gene mutations and clinicopathologic features remains unclear. This study aimed to explore somatic gene mutations associated with the clinicopathological features in patients with EACSCC, and to identify the candidate gene mutations for predicting survival outcome in EACSCC.
Methods
Twenty-two tissue samples obtained from patients with EACSCC were analyzed for genetic mutations based on targeted next-generation sequencing and genetic expression based on IHC staining to investigate the driver of tumorigenesis and/or the candidates of genes for predicting clinical outcome in EACSCC.
Results
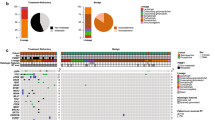
Gene alterations were most frequently observed in TP53 (59.1%), followed by CREBBP (9.1%). TP53 mutations showed significant correlation with T classification (P = 0.027) and p53 expression phenotype (P < 0.001). The 5-year overall survival (OS) rates for EACSCC patients with TP53 mutations and wild-type TP53 were 45.0% and 75.0%, respectively. Multivariable analysis using the Cox proportional hazards model demonstrated that TP53 mutations were independent predictors of OS rates for EACSCC patients (P = 0.007).
Conclusion
This study has suggested that TP53 mutations have potential for use as a biomarker for identifying individuals at high risk of developing tumors and for predicting survival outcome in EACSCC. IHC staining for p53 might play a useful role as screening tool for detecting TP53 mutations in patients with EACSCC.





Similar content being viewed by others
References
Yin M, Ishikawa K, Honda K et al (2006) Analysis of 95 cases of squamous cell carcinoma of the external and middle ear. Auris Nasus Larynx 33:251–257
Moody SA, Hirsch BE, Myers EN (2000) Squamous cell carcinoma of the external auditory canal: an evaluation of a staging system. Am J Otol 21:582–588
Morita S, Homma A, Nakamaru Y et al (2016) Outcomes of surgery and chemoradiotherapy for temporal bone cancer. Otol Neurotol 37:1174–1182
Stransky N, Egloff AM, Tward AD et al (2011) The mutational landscape of head and neck squamous cell carcinoma. Science 333:1157–1160
Mountzios G, Rampias T, Psyrri A (2014) The mutational spectrum of squamous-cell carcinoma of the head and neck: targetable genetic events and clinical impact. Ann Oncol 25:1889–1900
Li YY, Hanna GJ, Laga AC et al (2015) Genomic analysis of metastatic cutaneous squamous cell carcinoma. Clin Cancer Res 21:1447–1456
Zilberg C, Lee MW, Yu B et al (2018) Analysis of clinically relevant somatic mutations in high-risk head and neck cutaneous squamous cell carcinoma. Mod Pathol 31:275–287
Morita S, Nakamaru Y, Homma A et al (2017) Expression of p53, p16, cyclin D1, epidermal growth factor receptor and Notch1 in patients with temporal bone squamous cell carcinoma. Int J Clin Oncol 22:181–189
Zambetti GP (2007) The p53 mutation “gradient effect” and its clinical implications. J Cell Physiol 213:370–373
Sato K, Komune N, Hongo T et al (2020) Genetic landscape of external auditory canal squamous cell carcinoma. Cancer Sci 111:3010–3019
Boyle DP, McArt DG, Irwin G et al (2014) The prognostic significance of the aberrant extremes of p53 immunophenotypes in breast cancer. Histopathology 65:340–352
Takenaka Y, Cho H, Nakahara S et al (2015) Chemoradiation therapy for squamous cell carcinoma of the external auditory canal: A meta-analysis. Head Neck 37:1073–1080
Lawrence MS, Sougnez C, Lichtenstein L et al (2015) Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature 517:576–582
Watt SA, Purdie KJ, den Breems NY et al (2016) CREBBP mutation in human cutaneous squamous cell carcinoma. Exp Dermatol 25:650–651
Tsunoda A, Sumi T, Terasaki O et al (2017) Right dominance in the incidence of external auditory canal squamous cell carcinoma in the Japanese population: Does handedness affect carcinogenesis? Laryngoscope Investig Otolaryngol 2:19–22
Ichise T, Yoshida N, Ichise H (2019) CBP/p300 antagonises EGFR-Ras-Erk signalling and suppresses increased Ras-Erk signalling-induced tumour formation in mice. J Pathol 249:39–51
Levine AJ, Momand J, Finlay CA (1991) The p53 tumor suppressor gene. Nature 351:453–456
Marchetti P, Cannita K, Ricevuto E et al (2003) Prognostic value of p53 molecular status in high-risk primary breast cancer. Ann Oncol 14:704–708
Karpathiou G, Monaya A, Forest F et al (2016) p16 and p53 expression status in head and neck squamous cell carcinoma: a correlation with histological, histoprognostic and clinical parameters. Pathology 48:341–348
Kap M, Smedts F, Oosterhuis W et al (2011) Histological assessment of PAXgene tissue fixation and stabilization reagents. PLoS ONE 6:e27704
Acknowledgements
This study received financial support from the Japan Society for the Promotion of Science, KAKENHI (Grant Number 17K1688307).
Funding
Japan Society for the Promotion of Science, KAKENHI (Grant Number 17K1688307).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Morita, S., Kano, S., Hatanaka, K.C. et al. Association of gene mutations with clinicopathologic features in patients with external auditory canal squamous cell carcinoma. Int J Clin Oncol 27, 1394–1403 (2022). https://doi.org/10.1007/s10147-022-02191-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-022-02191-z