Abstract
Background
The aim of this study was to evaluate the safety and efficacy of low-dose everolimus treatment in patients with tuberous sclerosis complex (TSC)-associated angiomyolipoma (AML) with renal dysfunction or low body weight.
Methods
We investigated a total of 50 adult patients underwent everolimus treatment for AML associated with TSC. For patients with renal dysfunction (serum creatinine level ≥ 1.5 mg/dl) or low body weight (body weight < 35 kg), 5 mg of everolimus was administered daily (low-dose group). For patients without renal dysfunction or low body weight, 10 mg of everolimus was administered daily (conventional-dose group). The treatment effects and adverse events were compared between the two groups.
Results
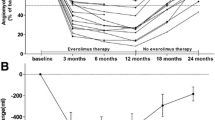
There were 20 patients in the low-dose group, and 30 in the conventional-dose group. The average reduction rate of the AML volume in the low-dose group was 52%, whereas it was 60% in the conventional-dose group. No significant differences were found in the average reduction rate between the groups (P = 0.24). The average blood everolimus trough levels were 7.7 ± 3.1 ng/mL in the low-dose group and 12.2 ± 5.7 ng/mL in the conventional-dose group. The level was significantly higher in the conventional-dose group than in the low-dose group (P = 0.004). The incidences of stomatitis and irregular menstruation were significantly lower in the low-dose group than in the conventional-dose group (P = 0.009, P = 0.045, respectively).
Conclusions
The present study demonstrates that low-dose everolimus treatment is safe and effective for TSC-associated AML. This treatment was well tolerated and adverse events were mild in all cases.

Similar content being viewed by others
Abbreviations
- TSC:
-
Tuberous sclerosis complex
- AML:
-
Angiomyolipoma
- ITSCCC:
-
International Tuberous Sclerosis Complex Consensus Conference
- mTOR:
-
Mammalian target of rapamycin
- TAE:
-
Transcatheter arterial embolization
- ILD:
-
Interstitial lung disease
- eGFR:
-
Estimated glomerular filtration rate
References
Curatolo P, Bombardieri R, Jozwiak S (2008) Tuberous sclerosis. Lancet 372:657–658
van Slegtenhorst M, de Hoogt R, Hermans C et al (1997) Identification of the tuberous sclerosis gene TSC1 on chromosome 9q34. Science 277:805–808
The European Chromosome 16 Tuberous Sclerosis Consortium (1993) Identification and characterization of the tuberous sclerosis gene on chromosome 16. Cell 75:1305–1315
Castagnetti M, Vezzu B, Laverda A et al (2007) Urological counseling and follow up in pediatric tuberous sclerosis complex. J Urol 178:2155–2159
Kaneda MW, Tanaka M, Hamasaki T, Katayama I (2013) Trends in the prevalence of tuberous sclerosis complex manifestations: an epidemiological study of 166 Japanese patients. PLoS ONE 8:e63910
Rouviere O, Nivet H, Grenier N et al (2013) Kidney damage due to tuberous sclerosis complex: management recommendations. Diagn Interv Imaging 94:225–237
Flum AS, Hamoui N, Said MA et al (2016) Update on the diagnosis and management of renal angiomyolipoma. J Urol 195:834–846
Rakowski SK, Winterkorn EB, Paul E et al (2006) Renal manifestations of tuberous sclerosis complex: incidence, prognosis, and predictive factors. Kidney Int 70:1777–1782
Krueger DA, Northrup H (2013) International Tuberous Sclerosis Complex Consensus Group. Tuberous sclerosis complex surveillance and management: recommendation of the 2012 International Tuberous Sclerosis Complex Consensus Conference. Pediat Neurol 48:255–265
Murata M, Ikeda Y, Hasegawa G et al (2019) Low-dose axitinib rechallenge with positive outcomes in a patient with metastatic renal cell carcinoma refractory to interferon a, sunitinib, and nivolumab therapies: a case report. J Med Case Rep 13:98
Wong AL, Sunder R, Wang TT et al (2016) Phase lb/ll randomized, open-label study of doxorubicin and cyclophosphamide with or without low-dose, short-course subitinib in the pre-operative treatment of breast cancer. Oncotarget 27:64089–64099
Chang JS, Chiou PY, Yao SH, Chou IC, Lin CY (2017) Regression of neonatal cardiac rhabdmyoma in two months through low-dose everolimus therapy: a report of three cases. Pediar Cardiol 38:1478–1484
Wei CC, Tsai JD, Sheu JN et al (2019) Continuous Low-dose everolimus shrinkage tuberous sclerosis complex-associated renal angiomyolipoma: a 48-month follow-up study. J investing Med 67:686–690
Bissler JJ, Kingswood JC, Radzikowska E et al (2013) Everolimus for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis (EXIST-2): a multicenter, randomized, double-blind, placebo-controlled trial. Lancet 381:817–824
Bissler JJ, Kingswood JC, Radzikowska E et al (2016) Everolimus for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis: extension of a randomized, controlled trial. Nephrol Dial Transpl 31:111–119
Motzer RJ, Escudier B, Oudard S et al (2010) Phase 3 trial of everolimus for metastatic renal cell carcinoma: final results and analysis of prognostic factors. Cancer 116:4256–4265
Yao JC, Shah MH, Ito T et al (2011) Everolimus for advanced pancreatic neuroendocrine tumors. N Eng J Med 364:514–523
Jones VE, McIntyre KJ, Paul D et al (2019) Evaluation of miracle mouthwash plus hydrocortisone versus prednisolone mouth rinses as prophylaxis for everolimus-associated stomatitis: a randomized phase II study. Oncologist 24:1153–1158
Huyghe E, Zairi A, Nohra J et al (2007) Gonadal impact of target of rapamycin inhibitors (sirolimus and everolimus) in male patients: an overview. Transpl Int 20:305–311
Fritsche L, Budde K, Dragun D et al (2004) Testosterone concentrations and sirolimus in male renal transplant patients. Am J Transplant 4:130–131
Braun M, Young J, Reiner CS et al (2012) Low-dose oral sirolimus and the risk of menstrual-cycle disturbances and ovarian cysts: analysis of the randomized controlled SUISSE ADPKD trial. PLoS ONE 7:e45868
Salgado JV, Neves FA, Bastos MG et al (2010) Monitoring renal function: measured and estimated glomerular filtration rates – a review. Braz J Med Biol Res 43:528–536
Acknowledgements
We thank the patients and their families for participating.
Author information
Authors and Affiliations
Contributions
TH participated in the design of the study and drafted the manuscript. KE participated in data acquisition. MT participated in the design of the study and helped to write the paper. All authors read and approved the final manuscript for submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors of this article declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Hatano, T., Endo, K. & Tamari, M. Efficacy and safety of low-dose everolimus treatment for renal angiomyolipoma associated with tuberous sclerosis complex. Int J Clin Oncol 26, 163–168 (2021). https://doi.org/10.1007/s10147-020-01792-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-020-01792-w