Abstract
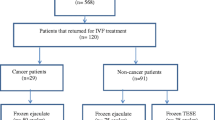
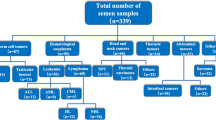
Chemotherapy-induced gonadal dysfunction resulting in transient or persistent infertility depends on the type of drugs and cumulative dose, and it is an important long-term complication, especially for adolescent and young adult (AYA) cancer patients. Due to its importance, a clinical practice guideline for fertility preservation in childhood and AYA cancer patients was published by the Japan Society of Clinical Oncology (JSCO) in 2017. Although the precise mechanisms remain unclear, several studies reported that the cancer itself, not the cancer treatment, adversely affected semen quality. It is reported that that poor pretreatment semen quality is commonly seen in various cancer types including testicular cancer, leukemia, brain tumor, and sarcoma. Fortunately, however, even men with poor sperm quality can be candidates for sperm cryopreservation due to recent advances in assisted reproductive technology (ART) and sperm banking techniques. Therefore, the JSCO guideline and others recommend that sperm cryopreservation should be considered as early as possible when patients are planning to undergo treatment that may render them infertile. The previous studies showed that testicular cancer and hematological tumors are the two leading types of cancer among patients who requested sperm cryopreservation. This is followed by bone and soft-tissue tumors and central nervous system tumors and others. Although the efficacy of postchemotherapy testicular sperm extraction (TESE)/intracytoplasmic sperm injection (ICSI) was reported recently, it is quite important to inform patients of the potential risk of treatment-induced infertility and the possibility of fertility preservation by sperm cryopreservation before chemotherapy.

Modified from the Japan Society of Clinical Oncology (JSCO) guideline

Similar content being viewed by others
References
NCCN Guideline (2018) Adolescent and young adult oncology, ver.2.2018, October 11, 2017, MS12–MS15. https://www.nccn.org/professionals/physician_gls/pdf/aya.pdf
Lee SJ, Schover LR, Partridge AH et al (2006) American Society of Clinical Oncology recommendations on fertility preservation in cancer patients. J Clin Oncol 24:2917–2931
Loren AW, Mangu PB, Beck LN et al (2013) Fertility preservation for patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol 31:2500–2510
Tournaye H, Dohle GR, Barratt CL (2014) Fertility preservation in men with cancer. Lancet 384:1295–1301
Japanese Society of Clinical Oncology (2017) JSCO Clinical practice guidelines 2017 for fertility preservation in childhood, adolescent, and young adults cancer patients. Kanehara, Tokyo
Williams DH 4th, Karpman E, Sander JC (2009) Pretreatment semen parameters in men with cancer. J Urol 181:736–740
Auger J, Sermondade N, Eustache F (2016) Semen quality of 4480 young cancer and systemic disease patients: baseline data and clinical considerations. Basic Clin Androl 26:3. https://doi.org/10.1186/s12610-016-0031-x
van der Kaaij MA, Heutte N, van Echten-Arends J (2009) Sperm quality before treatment in patients with early stage Hodgkin’s lymphoma enrolled in EORTC-GELA Lymphoma Group trials. Haematologica 94:1691–1697
Suzuki K, Shin T, Shimomura Y (2009) Spermatogenesis in tumor-bearing testes in germ cell testicular cancer patients. Hum Reprod 30:2853–2858
Meistrich ML (2013) Effects of chemotherapy and radiotherapy on spermatogenesis in humans. Fertil Steril 100:1180–1186
Lorenzi E, Simonelli M, Santoro A (2016) Infertility risk and teratogenicity of molecularly targeted anticancer therapy: A challenging issue. Crit Rev Oncol Hematol 107:1–13
Daudin M, Rives N, Walschaerts M et al (2015) Sperm cryopreservation in adolescents and young adults with cancer: results of the French national sperm banking network (CECOS). Fertil Steril. 103:478–486
Yumura Y, Tsujimura A, Okada H et al (2018) Current status of sperm banking for young cancer patients in Japanese nationwide survey. Asian J Androl. https://doi.org/10.4103/aja.aja_74_17 (in press)
Kobayashi H, Tamura K, Tai T et al (2017) Semen cryopreservation as an oncofertility treatment in 122 Japanese men with cancer: A decade-long study. Reprod Med Biol 16:320–324
Depalo R, Falagario D, Masciandaro P et al (2016) Fertility preservation in males with cancer: 16-year monocentric experience of sperm banking and post-thaw reproductive outcomes. Ther Adv Med Oncol 8:412–420
Petersen PM, Hansen SW, Giwercman A et al (1994) Dose-dependent impairment of testicular function in patients treated with cisplatin-based chemotherapy for germ cell cancer. Ann Oncol 5:355–358
Stephenson WT, Poirier SM, Rubin L et al (1995) Evaluation of reproductive capacity in germ cell tumor patients following treatment with cisplatin, etoposide, and bleomycin. J Clin Oncol 13:2278–2280
Lampe H, Horwich A, Norman A et al (1997) Fertility after chemotherapy for testicular germ cell cancers. J Clin Oncol 15:239–245
Pectasides D, Pectasides M, Farmakis D et al (2004) Testicular function in patients with testicular cancer treated with bleomycin-etoposide-carboplatin (BEC(90)) combination chemotherapy. Eur Urol 45:187–193
Gandini L, Sgrò P, Lombardo F et al (2006) Effect of chemo- or radiotherapy on sperm parameters of testicular cancer patients. Hum Reprod 21:2882–2889
Brydøy M, Fosså SD, Klepp O et al (2005) Paternity following treatment for testicular cancer. J Natl Cancer Inst 97:1580–1588
Kojima T, Kawai K, Tsuchiya K (2015) Identification of a subgroup with worse prognosis among patients with poor-risk testicular germ cell tumor. Int J Urol 22:923–927
Viviani S, Santoro A, Ragni G (1985) Gonadal toxicity after combination chemotherapy for Hodgkin’s disease. Comparative results of MOPP vs ABVD. Eur J Cancer Clin Oncol 21:601–605
Borgmann-Staudt A, Rendtorff R, Reinmuth S et al (2012) Fertility after allogeneic haematopoietic stem cell transplantation in childhood and adolescence. Bone Marrow Transplant 47:271–276
Rovó A, Aljurf M, Chiodi S et al (2013) Ongoing graft-versus-host disease is a risk factor for azoospermia after allogeneic hematopoietic stem cell transplantation: a survey of the late effects working party of the European group for blood and marrow transplantation. Haematologica 98:339–345
van Casteren NJ, van Santbrink EJ, van Inzen W et al (2008) Use rate and assisted reproduction technologies outcome of cryopreserved semen from 629 cancer patients. Fertil Steril 90:2245–2250
Agarwal A, Ranganathan P, Kattal N et al (2004) Fertility after cancer: a prospective review of assisted reproductive outcome with banked semen specimens. Fertil Steril 81:342–348
Ferrari S, Paffoni A, Filippi F et al (2016) Sperm cryopreservation and reproductive outcome in male cancer patients: a systematic review. Reprod Biomed Online 33:29–38
Hamano I, Hatakeyama S, Ohyama C (2017) Fertility preservation of patients with testicular cancer. Reprod Med Biol 16:240–251
Soda T, Okubo K, Ichioka K et al (2009) Sperm cryopreservation for cancer patients: 5-year experience in a private hospital in Japan. Hinyokika Kiyo 55:9–13
Suzuki K, Matuzaki J, Hattori Y (2007) Semen cryopreservation for patients with malignant or non-malignant disease: our 14-year experience. Hinyokika Kiyo 53:539–544
Meseguer M, Garrido N, Remohí J (2003) Testicular sperm extraction (TESE) and ICSI in patients with permanent azoospermia after chemotherapy. Hum Reprod 18:1281–1285
Hsiao W, Stahl PJ, Osterberg EC (2011) Successful treatment of postchemotherapy azoospermia with microsurgical testicular sperm extraction: the Weill Cornell experience.J Clin Oncol. 29:1607–1611
Shiraishi K, Matsuyama H (2014) Microdissection testicular sperm extraction and salvage hormonal treatment in patients with postchemotherapy azoospermia. Urology 83:100–106
Shin T, Kobayashi T, Shimomura Y (2016) Microdissection testicular sperm extraction in Japanese patients with persistent azoospermia after chemotherapy. Int J Clin Oncol 21:1167–1171
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No author has any conflict of interest.
About this article
Cite this article
Kawai, K., Nishiyama, H. Preservation of fertility of adult male cancer patients treated with chemotherapy. Int J Clin Oncol 24, 34–40 (2019). https://doi.org/10.1007/s10147-018-1333-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-018-1333-0