Abstract
Background
Recently, the approval of some molecularly targeted drugs has been questioned, due to differing opinions on their risks and benefits. The approval process remains a challenge in regulatory science.
Methods
We analyzed the molecularly targeted drugs listed in the 2013 Medical Formulary. For the 21 identified drugs, 32 published Pharmaceuticals and Medical Devices Agency (PMDA) reports were open to the public. Data regarding clinical trials were extracted from these reports and assessed in order to clarify the characteristic examinations required for the approval of molecularly targeted drugs.
Results
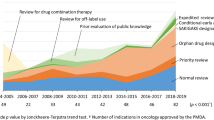
There was no correlation between the application year and the time between application and approval (p = 0.139). The median number of clinical trials in these reports was 5 (range 1–22). Phase III studies were not included in the assessment materials for 11 reports. A survival benefit was demonstrated for six of the 32 drugs. The PMDA issued approval terms, including all-case surveillance and additional clinical trials, for 24 of these 32 drugs.
Conclusion
Molecularly targeted drugs were approved by the PMDA using a flexible process based on drug safety and efficacy. Doctors and patients who are administering or receiving these drugs should be fully informed about the lack of Japanese data assessment during these approval processes.




Similar content being viewed by others
References
O’Brien SG, Guilhot F, Larson RA et al (2003) Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med 348:994–1004
Miksad RA, Schnipper L, Goldstein AM (2007) Does a statistically significant survival benefit of erlotinib plus gemcitabine for advanced pancreatic cancer translate into clinical significance and value? J Clin Oncol 25:4506–4507
Stegmeier F, Warmuth M, Sellers WR et al (2010) Targeted cancer therapies in the twenty-first century: lessons from imatinib. Clin Pharmacol Ther 87:543–552
Segaert S, Van Cutsem E (2005) Clinical signs, pathophysiology and management of skin toxicity during therapy with epidermal growth factor receptor inhibitors. Ann Oncol 16:1425–1433
Seidman A, Hudis C, Pierri MK et al (2002) Cardiac dysfunction in the trastuzumab clinical trials experience. J Clin Oncol 20:1215–1221
Moore MJ, Goldstein D, Hamm J et al (2007) Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 25:1960–1966
Narimatsu H, Oiso G, Ono S et al (2009) Critical review of the determination process by the Japanese reviewing authority in approving the additional efficacy of fludarabine phosphate. J Clin Oncol 27:e236–e238
Ito Y, Narimatsu H, Fukui T et al (2013) Critical review of ‘Public domain application’: a flexible drug approval system in Japan. Ann Oncol 24:1297–1305
Antitumor agents, Medical Formulary of Japan 2013, Tokyo, Japan, Pharmaceutical Information Center, 2013, p 87–91
Grever MR (2013) Accelerating safe drug development: an ideal approach to approval. Hematol Am Soc Hematol Educ Progr 2013:24–29
Narimatsu H, Hori A, Matsumura T et al (2008) Cooperative relationship between pharmaceutical companies, academia, and media explains sharp decrease in frequency of pulmonary complications after bortezomib in Japan. J Clin Oncol 26:5820–5823
Cancer information service: information of domestic unapproved drugs that have been addressed in public meeting: 04/2014 update. http://ganjoho.jp/professional/med_info/drug/index.html. Accessed Jan 2014
Kudoh S, Kato H, Nishiwaki Y et al (2008) Interstitial lung disease in Japanese patients with lung cancer: a cohort and nested case-control study. Am J Respir Crit Care 177:1348–1357
Cohen MH, Williams GA, Sridhara R et al (2004) United States Food and Drug Administration Drug Approval summary: Gefitinib (ZD1839; Iressa) tablets. Clin Cancer Res 10:1212–1218
Wilson MK, Karakasis K, Oza AM (2015) Outcomes and endpoints in trials of cancer treatment: the past, present, and future. Lancet Oncol 16:e32–e42
Qureshi ZP, Seoane-Vazquez E, Rodriguez-Monguio R (2011) Market withdrawal of new molecular entities approved in the United States from 1980 to 2009. Pharmacoepidemiol Drug Saf 20:772–777
Acknowledgments
This work was supported by JSPS KAKENHI Grant Number 25670240.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
About this article
Cite this article
Sasahara, Y., Narimatsu, H., Fukao, A. et al. Approving molecularly targeted drugs: different approval processes for cytotoxic agents. Int J Clin Oncol 21, 1004–1013 (2016). https://doi.org/10.1007/s10147-016-0980-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-016-0980-2