Abstract
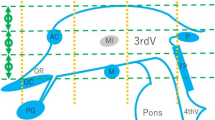
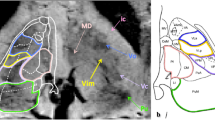
Surgical targeting of the ventral intermediate nucleus of the thalamus (VIM) has been historically done using indirect strategies. Here we depict the cerebello-thalamo-cortical tract (CTCT) through 3 T proton density (PD) in a cohort of patients who underwent high-intensity focus ultrasound (HIFUS) thalamotomy. Forty-seven patients treated in our institution with MR-guided HIFUS VIM thalamotomy were included in this study. PD weighted 3 T MRI used for presurgical planning was compared with postoperative MRI obtained 1 month after surgery. Images were processed with ISTX software (Brain lab, Munich, Germany). The coordinates of the VIM lesion concerning the inter-commissural line (ICL) were annotated. Deterministic tractographies using three ROIs were used to verify the different tracts. The triangle seen in the 3 T PD sequence at the level of the mesencephalic-diencephalic junction was systematically recognized. The posterior angle of this triangle at the junction of the CTCT and the ZI was denominated as “point P.” The area of this triangle corresponds to the posterior subthalamic area (PSA) harboring the Raprl fibers. The CTCT was visible from 1 to 2.5 mm below the ICL. The average center of the final HIFUS lesion (point F) was 11 mm from the medial thalamic border of the thalamus (14.9 mm from the midline), 6.4 mm anterior to PC, and 0.6 mm above the ICL. The FUS point was consistently 1–2 mm directly above point P. The anterior border of the external angle of this triangle (point P) can be used as an intraparenchymal point for targeting the ventral border of the VIM. Three ROIs placed in a single slice around this triangle are a fast way to originate tractography of the CTCT, lemniscus medialis, and pyramidal tract.






Similar content being viewed by others
Data availability
See supplementary contents to view the data for each patient and the imaging protocol employed in this study.
Code availability
Not applicable.
References
Bhatia KP, Bain P, Bajaj N, Elble RJ, Hallett M, Louis ED, Raethjen J, Stamelou M, Testa CM, Deuschl G (2018) Consensus statement on the classification of tremors. From the task force on tremor of the International Parkinson and Movement Disorder Society. Mov Disord 33:75–87. https://doi.org/10.1002/MDS.27121
Louis ED, Machado DG (2015) Tremor-related quality of life: a comparison of essential tremor vs. Parkinson’s disease patients Parkinsonism Relat Disord 21:729–735. https://doi.org/10.1016/J.PARKRELDIS.2015.04.019
Spiegelmann R, Friedman WA (1991) Rapid determination of thalamic CT-stereotactic coordinates: a method. Acta Neurochir (Wien) 110:77–81. https://doi.org/10.1007/BF01402051
Pineda-Pardo JA, Urso D, Martínez-Fernández R, Rodríguez-Rojas R, del-Alamo M, Vernetti PM, Máñez-Miró JU, Hernández-Fernández F, de Luis-Pastor E, Vela-Desojo L, Obeso JA (2020) Transcranial magnetic resonance-guided focused ultrasound thalamotomy in essential tremor: a comprehensive lesion characterization. Neurosurgery 87:256–265. https://doi.org/10.1093/neuros/nyz395
King NKK, Krishna V, Sammartino F, Bari A, Reddy GD, Hodaie M, Kalia SK, Fasano A, Munhoz RP, Lozano AM, Hamani C (2017) Anatomic targeting of the optimal location for thalamic deep brain stimulation in patients with essential tremor. World Neurosurg 107:168–174. https://doi.org/10.1016/j.wneu.2017.07.136
Brierley JB, Beck E (1959) The significance in human stereotactic brain surgery of individual variation in the diencephalon and globus pallidus. J Neurol Neurosurg Psychiatry 22:287–298. https://doi.org/10.1136/jnnp.22.4.287
Spiegelmann R, Nissim O, Daniels D, Ocherashvilli A, Mardor Y (2006) Stereotactic targeting of the ventrointermediate nucleus of the thalamus by direct visualization with high-field MRI. Stereotact Funct Neurosurg 84:19–23. https://doi.org/10.1159/000092683
Schaltenbrand G, Bailey P (1959) Introduction to stereotaxis with an atlas of the human. Georg Thieme Verlag, Brain Stuttgart
Morel A (2007) Stereotactic atlas of the human thalamus and basal ganglia. CRC Press
Low HL, Ismail MN bin M, Taqvi A, Deeb J, Fuller C, Misbahuddin A (2019) Comparison of posterior subthalamic area deep brain stimulation for tremor using conventional landmarks versus directly targeting the dentatorubrothalamic tract with tractography. Clin Neurol Neurosurg 185:105466. https://doi.org/10.1016/j.clineuro.2019.105466
Ramirez-Zamora A, Smith H, Kumar V, Prusik J, Phookan S, Pilitsis JG (2016) Evolving concepts in posterior subthalamic area deep brain stimulation for treatment of tremor: surgical neuroanatomy and practical considerations. Stereotact Funct Neurosurg 94:283–297. https://doi.org/10.1159/000449007
Wong JK, Hess CW, Almeida L, Middlebrooks EH, Christou EA, Patrick EE, Shukla AW, Foote KD, Okun MS (2020) Deep brain stimulation in essential tremor: targets, technology, and a comprehensive review of clinical outcomes. Expert Rev Neurother 20:319–331
Gallay MN, Moser D, Rossi F, Pourtehrani P, Magara AE, Kowalski M, Arnold A, Jeanmonod D (2016) Incisionless transcranial MR-guided focused ultrasound in essential tremor: cerebellothalamic tractotomy. J Ther Ultrasound 4:5. https://doi.org/10.1186/s40349-016-0049-8
Xie T, Bernard J, Warnke P (2012) Post subthalamic area deep brain stimulation for tremors: a mini-review. Transl Neurodegener 1:20. https://doi.org/10.1186/2047-9158-1-20
Tomsak RL (2005) Encyclopedia of the neurological sciences. J Neuro-Ophthalmology 25:252–253. https://doi.org/10.1097/01.wno.0000176626.56739.d4
Elias WJ, Lipsman N, Ondo WG, Ghanouni P, Kim YG, Lee W, Schwartz M, Hynynen K, Lozano AM, Shah BB, Huss D, Dallapiazza RF, Gwinn R, Witt J, Ro S, Eisenberg HM, Fishman PS, Gandhi D, Halpern CH, Chuang R, Butts Pauly K, Tierney TS, Hayes MT, Cosgrove GR, Yamaguchi T, Abe K, Taira T, Chang JW (2016) A randomized trial of focused ultrasound thalamotomy for essential tremor. N Engl J Med 375:730–739. https://doi.org/10.1056/nejmoa1600159
Fahn S, Tolosa E, Marin C (1988) Clinical rating scale for tremor. In: Parkinson’s Disease and Movement Disorders. pp 225–34
Yamada K, Akazawa K, Yuen S, Goto M, Matsushima S, Takahata A, Nakagawa M, Mineura K, Nishimura T (2010) MR imaging of ventral thalamic nuclei. Am J Neuroradiol 31:732–735. https://doi.org/10.3174/ajnr.A1870
Louis ED (2010) Essential tremor: evolving clinicopathological concepts in an era of intensive post-mortem enquiry. Lancet Neurol 9:613–622
Louis ED (2016) Linking essential tremor to the cerebellum: neuropathological evidence. Cerebellum 15:235–242. https://doi.org/10.1007/s12311-015-0692-6
Lehman VT, Lee KH, Klassen BT, Blezek DJ, Goyal A, Shah BR, Gorny KR, Huston J, Kaufmann TJ MRI and tractography techniques to localize the ventral intermediate nucleus and dentatorubrothalamic tract for deep brain stimulation and MR-guided focused ultrasound: a narrative review and update. Neurosurg Focus FOC 49:E8. https://doi.org/10.3171/2020.4.FOCUS20170
Lau JC, Xiao Y, Haast RAM, Gilmore G, Uludağ K, MacDougall KW, Menon RS, Parrent AG, Peters TM, Khan AR (2020) Direct visualization and characterization of the human zona incerta and surrounding structures. Hum Brain Mapp 41:4500–4517. https://doi.org/10.1002/hbm.25137
Akram H, Dayal V, Mahlknecht P, Georgiev D, Hyam J, Foltynie T, Limousin P, De Vita E, Jahanshahi M, Ashburner J, Behrens T, Hariz M, Zrinzo L (2018) Connectivity derived thalamic segmentation in deep brain stimulation for tremor. NeuroImage Clin 18:130–142. https://doi.org/10.1016/j.nicl.2018.01.008
Al-Fatly B, Ewert S, Kübler D, Kroneberg D, Horn A, Kühn AA (2019) Connectivity profile of thalamic deep brain stimulation to effectively treat essential tremor. Brain 142:3086–3098. https://doi.org/10.1093/brain/awz236
Fiechter M, Nowacki A, Oertel MF, Fichtner J, Debove I, Lachenmayer ML, Wiest R, Bassetti CL, Raabe A, Kaelin-Lang A, Schüpbach MW, Pollo C (2017) Deep brain stimulation for tremor: is there a common structure? Stereotact Funct Neurosurg 95:243–250. https://doi.org/10.1159/000478270
Herrington TM, Cheng JJ, Eskandar EN (2016) Mechanisms of deep brain stimulation. J Neurophysiol 115:19–38
Horn A, Reich M, Vorwerk J, Li N, Wenzel G, Fang Q, Schmitz-Hübsch T, Nickl R, Kupsch A, Volkmann J, Kühn AA, Fox MD (2017) Connectivity predicts deep brain stimulation outcome in Parkinson disease. Ann Neurol 82:67–78. https://doi.org/10.1002/ana.24974
Gallay MN, Moser D, Jeanmonod D (2021) MR-guided focused ultrasound cerebellothalamic tractotomy for chronic therapy-resistant essential tremor: anatomical target reappraisal and clinical results. J Neurosurg 134:376–385. https://doi.org/10.3171/2019.12.JNS192219
Su JH, Choi EY, Tourdias T, Saranathan M, Halpern CH, Henderson JM, Pauly KB, Ghanouni P, Rutt BK (2020) Improved vim targeting for focused ultrasound ablation treatment of essential tremor: a probabilistic and patient-specific approach. Hum Brain Mapp 41:4769–4788. https://doi.org/10.1002/hbm.25157
Acknowledgements
We are indebted to the Social Medicine and Family Health Department. The University of Cauca, Popayan, Colombia, for its critical support methodological issues.
Author information
Authors and Affiliations
Contributions
Concept and design: Paez and Spiegelmann. Data acquisition: Paez, Korn, and Zibly. Article draft: Paez. Critical review: Spielgelmann, Paez, and Garcia. Epidemiological advice: Daza. Statistical analysis: Illera and Alcazar. Study supervision: Spiegelmann and Garcia.
Corresponding authors
Ethics declarations
Ethics approval
For this retrospective study, an approval was granted by the institutional review board (IRB).
Consent to participate
Not applicable.
Consent for publication
All authors have previously acknowledged for the publication of this article in Neurosurgical Review.
Conflict of interest
The senior author (RS) has served as a consultant to Brainlab. The other authors have no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Páez-Nova, M., Spiegelmann, R., Korn-Israeli, S. et al. Targeting the vim by direct visualization of the cerebello-thalamo-cortical pathway in 3 T proton density MRI: correlation with focused ultrasound lesioning. Neurosurg Rev 45, 2323–2332 (2022). https://doi.org/10.1007/s10143-022-01752-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-022-01752-0