Abstract
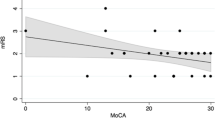
Predicting recovery potential of patients with aneurysmal subarachnoid hemorrhage (aSAH) is challenging. We investigated whether the gray-to-white matter ratio (GWR) predicts recovery of cognitive function (CF) and quality of life (QOL) of these patients. We analyzed data of 69 patients with aSAH. Patients’ demographics, comorbidities, and neurological status were recorded. One year after aSAH, Montreal Cognitive Assessment (MoCA) and Short Form-36 (SF-36) tests were administered to the patients, and brain volumes of patients were examined using MRI. Three years after aSAH, MoCA and SF-36 tests were conducted again. Differences between the test scores 1 and 3 years after aSAH were evaluated (ΔMoCA and ΔSF-36). Patients with ΔMoCA ≥ 4 points and those with ΔSF-36 ≥ 8 points were referred to as good MoCA and SF-36 recovery, respectively. ΔMoCA correlated with GWR in male and female patients (females: p < 0.001, R2 = 0.581; males: p < 0.001, R2 = 0.481). In female patients, GWR > 1.34 predicted good MoCA recovery with 82.3% sensitivity and 80% specificity, and in male patients, GWR > 1.36 predicted good MoCA recovery with 80% sensitivity and 95% specificity. ΔSF-36 correlated with GWR in male and female patients (females: p < 0.001, R2 = 0.479; males: p < 0.001, R2 = 0.627). In female patients, GWR > 1.35 predicted good SF-36 recovery with 74% sensitivity and 84% specificity, and in male patients, GWR > 1.38 predicted good SF-36 recovery with 72% sensitivity and 92% specificity. GWR is a good predictor of the recovery of CF and QOL in patients with aSAH and, thus, can help physicians to better organize rehabilitation of patients.



Similar content being viewed by others
References
Ali A, Tanirgan G, Sabanci PA, Sivrikoz N, Abdullah T, Sencer A, Sencer S, Orhan-Sungur M, Akinci IO (2018) Relation of gray-white matter ratio with long-term cognitive functions and quality of life in patients with mild to moderate aneurysmal subarachnoid hemorrhage: a prospective observational study. Acta Neurochir 160:181–189
Al-Khindi T, Macdonald RL, Schweizer TA (2010) Cognitive and functional outcome after aneurysmal subarachnoid hemorrhage. Stroke 41:519–536
Bendel P, Koivisto T, Aikiä M et al (2010) Atrophic enlargement of CSF volume after subarachnoid hemorrhage: correlation with neuropsychological outcome. Am J Neuroradiol 31:370–376
Bendel P, Koivisto T, Niskanen E, Könönen M, Äikiä M, Hänninen T, Koskenkorva P, Vanninen R (2009) Brain atrophy and neuropsychological outcome after treatment of ruptured anterior cerebral artery aneurysms: a voxel-based morphometric study. Neuroradiology 51:711–722
Bendel P, Koivisto T, Könönen M, Hänninen T, Hurskainen H, Saari T, Vapalahti M, Hernesniemi J, Vanninen R (2008) MR imaging of the brain 1 year after aneurysmal subarachnoid hemorrhage: randomized study comparing surgical with endovascular treatment. Radiology 246:543–552
Boerboom W, Heijenbrok-Kal MH, Khajeh L, van Kooten F, Ribbers GM (2016) Long-term functioning of patients with aneurysmal subarachnoid hemorrhage: a 4-yr follow-up study. Am J Phys Med Rehabil 95:112–120
Cahill J, Zhang JH (2009) Subarachnoid hemorrhage: is it time for a new direction? Stroke 40:86–87
Carne RP, Vogrin S, Litewka L, Cook MJ (2006) Cerebral cortex: an MRI-based study of volume and variance with age and sex. J Clin Neurosci 13:60–72
Choi SP, Park HK, Park KN, Kim YM, Ahn KJ, Choi KH, Lee WJ, Jeong SK (2008) The density ratio of grey to white matter on computed tomography as an early predictor of vegetative state or death after cardiac arrest. Emerg Med J 25:666–669
Connolly ES Jr, Rabinstein AA, Carhuapoma JR et al (2012) Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 43:1711–1737
De Bresser J, Schaafsma JD, Luitse MJ, Viergever MA, Rinkel GJ, Biessels GJ (2015) Quantification of structural cerebral abnormalities on MRI 18 months after aneurysmal subarachnoid hemorrhage in patients who received endovascular treatment. Neuroradiology 57:269–274
De Bresser J, Vincken KL, Kaspers AJ, Rinkel GJ, Viergever MA, Biessels GJ (2012) Quantification of cerebral volumes on MRI 6 months after aneurysmal subarachnoid hemorrhage. Stroke 43:2782–2784
de Rooij NK, Linn FH, van der Plas JA, Algra A, Rinkel GJ (2007) Incidence of subarachnoid haemorrhage: a systematic review with emphasis on region, age, gender and time trends. J Neurol Neurosurg Psychiatry 78:1365–1372
Frangou S, Chitins X, Williams SC (2004) Mapping IQ and gray matter density in healthy young people. NeuroImage 23:800–805
Ge Y, Grossman RI, Babb JS, Rabin ML, Mannon LJ, Kolson DL (2002) Age-related total gray matter and white matter changes in normal adult brain. Part I: volumetric MR imaging analysis. Am J Neuroradiol 23:1327–1333
Ikawa F, Abiko M, Ishii D, Ohshita J, Matsushige T, Okazaki T, Sakamoto S, Hida E, Kobayashi S, Kurisu K (2018) Analysis of outcome at discharge after aneurysmal subarachnoid hemorrhage in Japan according to the Japanese stroke databank. Neurosurg Rev 41:567–574
Krajewski K, Dombek S, Martens T, Köppen J, Westphal M, Regelsberger (2014) Neuropsychological assessments in patients with aneurysmal subarachnoid hemorrhage, perimesencephalic SAH, and incidental aneurysms. J Neurosurg Rev 37:55–62
Martinaud O, Perin B, Gérardin E, Proust F, Bioux S, Gars DL, Hannequin D, Godefroy O (2009) Anatomy of executive deficit following ruptured anterior communicating artery aneurysm. Eur J Neurol 16:595–601
Molyneux AJ, Kerr RS, Yu LM, Clarke M, Sneade M, Yarnold JA, Sandercock P, International Subarachnoid Aneurysm Trial (ISAT) Collaborative Group (2005) International subarachnoid aneurysm trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised comparison of effects on survival, dependency, seizures, rebleeding, subgroups, and aneurysm occlusion. Lancet 366:809–817
Ozdilek B, Kenangil G (2014) Validation of the Turkish version of the Montreal cognitive assessment scale (MoCA-TR) in patients with Parkinson's disease. Clin Neuropsychol 28:333–343
Powell J, Kitchen N, Heslin J, Greenwood R (2002) Psychosocial outcomes at three and nine months after good neurological recovery from aneurysmal subarachnoid haemorrhage: predictors and prognosis. J Neurol Neurosurg Psychiatry 72:772–781
Pyysalo LM, Keski-Nisula LH, Niskakangas TT, Kahara VJ, Ohman JE (2011) Long-term MRI findings of patients with embolized cerebral aneurysms. Acta Radiol 52:204–210
Samra SK, Giordani B, Caveney AF, Clarke WR, Scott PA, Anderson S, Thompson BG, Todd MM, for the CFAAST Investigators (2007) Recovery of cognitive function after surgery for aneurysmal subarachnoid hemorrhage. Stroke 38:1864–1872
Stehouwer BL, van der Kleij LA, Hendrikse J, Rinkel GJ, De Vis JB (2018) Magnetic resonance imaging and brain injury in the chronic phase after aneurysmal subarachnoid hemorrhage: a systematic review. Int J Stroke 13:24–34
Terada T, Miyata J, Obi T, Kubota M, Yoshizumi M, Murai T (2018) Reduced gray matter volume is correlated with frontal cognitive and behavioral impairments in Parkinson’s disease. J Neurol Sci 390:231–238
Torbey MT, Selim M, Knorr J, Bigelow C, Recht L (2000) Quantitative analysis of the loss of distinction between gray and white matter in comatose patients after cardiac arrest. Stroke 31:2163–2167
Wallmark S, Ronne-Engström E, Lundström E (2016) Predicting return to work after subarachnoid hemorrhage using the Montreal Cognitive Assessment (MoCA). Acta Neurochir 158:233–239
Ware JE, Sherbourne CD (1992) The MOS 36-item short-form health survey. (SF-36). Med Care 30:473–483
Zheng L, Bin G, Zeng H, Zou D, Gao J, Zhang J, Huang B. Meta-analysis of voxel-based morphometry studies of gray matter abnormalities in patients with mesial temporal lobe epilepsy and unilateral hippocampal sclerosis. Brain Imaging Behav. 2018 Jan 4
Acknowledgments
Preparation for publication of this article was partly supported by the Turkish Neurosurgical Society.
Funding
Support was provided solely by institutional and/or departmental sources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
The present study was approved by the Ethics Committee of the Istanbul University, Istanbul Medical Faculty, Istanbul, Turkey (Ethics committee no. 2018/276).
Informed consent
Written informed consent was obtained from all patients and/or surrogates.
Electronic supplementary material
Supplementary image 1A
Belong to female 48 years aged patient. MRI was performed 3 years after aSAH. Basilar artery aneurysm treated by coiling. GWR value is 1.32 (good GWR value). ΔMoCA is 9 and ΔSF-36 is 10 1B: Belong to male 57 years aged patient. MRI was performed 2 years and 11 months after aSAH. Anterior communicating artery aneurysm treated by coiling. GWR value is 1.22 (low GWR). ΔMoCA is 3 and ΔSF-36 is 6 (PNG 347 kb)
Rights and permissions
About this article
Cite this article
Ali, A., Bitir, B., Abdullah, T. et al. Gray-to-white matter ratio predicts long-term recovery potential of patients with aneurysmal subarachnoid hemorrhage. Neurosurg Rev 43, 195–202 (2020). https://doi.org/10.1007/s10143-018-1029-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-018-1029-y