Abstract
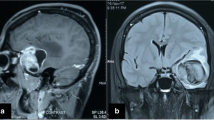
The case of a 7-year-old boy suffering from a supratentorial primitive neuroectodermal tumour (sPNET) at the age of 5 is presented. The tumour has been characterized by astrocytic areas within the sPNET revealing malignant transformation up to a multiform glioblastoma during the course of the disease. The clonal origin of both tumour components was established by loss of heterozygosity (LOH) analysis. Clinically, the tumour showed an aggressive biological behaviour with two recurrences. We discuss this very rare case and the first description of the clonal origin of distinct and distinguishable tumour components taking into consideration published literature.




Similar content being viewed by others
References
Albright AL, Wisoff JH, Zeltner P, Boyett J, Rorke LB, Stanley P, Geyer JR, Milstein JM (1995) Prognostic factors in children with supratentorial (nonpineal) primitive neuroectodermal tumors. A neurosurgical perspective from the Children’s Cancer Group. Pediatr Neurosurg 22:1–7
Barnard RO, Pambakian H (1980) Astrocytic differentiation in medulloblastoma. J Neurol Neurosurg Psychiatry 43(11):1041–1044
Beetz C, Hartmann A, Kiehntopf M, Wolfl S, Kalff R, Deufel T, Patt S (2004) Rapid generation of detailed loss of heterozygosity profiles for routine diagnosis of gliomas. Clin Chem Lab Med 42(6):595–601
Bennett JP, Rubinstein LJ (1984) The biological behaviour of primary cerebral neuroblastoma: a reappraisal of the clinical course in a series of 70 cases. Ann Neurol 16(1):21–27
Burger PC, Yu IT, Tihan T, Friedman HS, Strother DR, Kepner JL, Duffner PK, Kun LE, Perlman EJ (1998) Atypical teratoid/rhabdoid tumor of the central nervous system: a highly malignant tumor of infancy and childhood frequently mistaken for medulloblastoma: a Pediatric Oncology Group study. Am J Surg Pathol 22(9):1083–1092
Burger PC, Scheithauer BW, Vogel FS (2002) Surgical pathology of the nervous system and its coverings, 4th edn. Churchill Livingstone, New York, pp 298–315, 336–338
Cohen BH, Zeltzer PM, Boyett JM, Geyer JR, Allen JC, Finlay JL, McGuire-Cullen P, Milstein JM, Rorke LB, Stanley P et al (1995) Prognostic factors and treatment results for supratentorial primitive neuroectodermal tumors in children using radiation and chemotherapy: a Children’s Cancer Group randomized trial. J Clin Oncol 13:1687–1696
Dehner LP (1986) Peripheral and central primitive neuroectodermal tumors. A nosologic concept seeking a consensus. Arch Pathol Lab Med 110(11):997–1005
Dirks PB, Harris L, Hoffman HJ, Humphreys RP, Drake JM, Rutka JT (1996) Supratentorial primitive neuroectodermal tumors in children. J Neurooncol 29:75–84
Di Rocco C, Lanelli A, Ceddia A (1991) Intracranial tumors of the first year of life. A cooperative survey of the 1986–1987 Education Committee of the ISPN. Childs Nerv Syst 7:150–153
Furuta T, Sugiu K, Tamiya T, Matsumoto K, Ohmoto T (1998) Malignant cerebellar astrocytoma developing 15 years after radiation therapy for a medulloblastoma. Clin Neurol Neurosurg 100(1):56–59
Gaffney CC, Sloane JP, Bradley NJ, Bloom HJ (1985) Primitive neuroectodermal tumours on the cerebrum (pathology and treatment). J Neurooncol 3(1):23–33
Hart MN, Earle KM (1973) Primitive neuroectodermal tumors of the brain in children. Cancer 32:890–897
Janss AJ, Yachnis AT, Silber JH, Trojanowski JQ, Lee VM, Sutton LN, Perilongo G, Rorke LB, Phillips PC (1996) Glial differentiation predicts poor clinical outcome in primitive neuroectodermal brain tumors. Ann Neurol 39(4):481–489
Kleriga E, Sher JH, Nallainathan SK, Stein SC, Sacher M (1978) Development of cerebellar malignant astrocytoma at site of a medulloblastoma treated 11 years earlier. Case report. J Neurosurg 49(3):445–449
Mahapatra AK, Sinha AK, Sharma MC (1998) Medullomyoblastoma: a rare cerebellar tumour in children. Childs Nerv Syst 14(7):312–316
Mannoji H, Takeshita I, Fukui M, Ohta M, Kitamura K (1981) Glial fibrillary acidic protein in medulloblastoma. Acta Neuropathol 55(1):63–69
Maria BL, Steck PA, Yung WK, Milici A, Bruner JM, Pathak S, Becker FF (1989) The modulation of astrocytic differentiation in cells derived from a medulloblastoma surgical specimen. J Neurooncol 7(4):329–338
Maria BL, Wong D, Kalnins VI (1990) Dibutyryl cyclic AMP induces vimentin and GFAP expression in cultured medulloblastoma cells. Can J Neurol Sci 17(1):15–20
McLean TW (2003) Medulloblastomas and central nervous system primitive neuroectodermal tumors. Curr Treat Options Oncol 4(6):499–508
McNeil DE, Cote TR, Clegg L, Rorke LB (2002) Incidence and trends in pediatric malignancies medulloblastoma/primitive neuroectodermal tumor: a SEER update. Surveillance Epidemiology and End Results. Med Pediatr Oncol 39(3):190–194
Mena H, Rushing EJ, Ribas JL, Delahunt B, McCarthy WF (1995) Tumors of pineal parenchymal cells: a correlation of histological features, including nucleolar organizer regions, with survival in 35 cases. Hum Pathol 26(1):20–30
Mikaeloff Y, Raquin MA, Lellouch-Tubiana A, Terrier-Lacombe MJ, Zerah M, Bulteau C, Habrand JL, Kalifa C (1998) Primitive cerebral neuroectodermal tumors excluding medulloblastomas: a retrospective study of 30 cases. Pediatr Neurosurg 29:170–177
Molenaar WM, Jansson DS, Gould VE, Rorke LB, Franke WW, Lee VM, Packer RJ, Trojanowski JQ (1989) Molecular markers of primitive neuroectodermal tumors and other pediatric central nervous system tumors. Lab Invest 61(6):635–643
Mørk SJ, Rubinstein LJ (1985) Ependymoblastoma: a reappraisal of a rare embryonal tumor. Cancer 55(7):1536–1542
Nakamizo A, Nishio S, Inamura T, Koga H, Yamabe K, Kuba H, Matsushima T, Fukui M (2001) Evolution of malignant cerebellar astrocytoma at the site of a treated medulloblastoma: report of two cases. Acta Neurochir (Wien) 143:697–700
Nishio S, Morioka T, Fukui M (1998) Primitive neuroectodermal tumors. Crit Rev Neurosurg 8:216–268
Packer RJ, Sutton LN, Rorke LB, Littman PA, Sposto R, Rosenstock JG, Bruce DA, Schut L (1984) Prognostic impotrance of cellular differentiation in medulloblastoma of childhood. J Neurosurg 61(2):296–301
Packer RJ, Cogen P, Vezina G, Rorke LB (1999) Medulloblastoma: clinical and biological aspects. Neuro-oncol 1(3):232–250
Paulino AC, Melian E (1999) Medulloblastoma and supratentorial primitive neuroectodermal tumors. Cancer 86:142–148
Paulino AC, Cha DT, Barker JL, Lo S, Manera RB (2004) Patterns of failure in relation to radiotherapy fields in supratentorial primitive neuroectodermal tumor. Int J Radiat Oncol Biol Phys 58(4):1171–1176
Rickert CH, Probst-Cousin S, Gullotta F (1997) Primary intracranial neoplasms of infancy and early childhood. Childs Nerv Syst 13:507–513
Rorke LB (1983) The cerebellar medulloblastoma and its relationship to primitive neuroectodermal tumors. J Neuropathol Exp Neurol 42(1):1–15
Rorke LB, Packer RJ, Biegel JA (1996) Central nervous system atypical teratoid/rhabdoid tumors of infancy and childhood: definition of an entity. J Neurosurg 85(1):56–65
Rorke LB, Hart MN, McLendon RE (2000) Supratentorial primitive neuroectodermal tumour (PNET). In: Kleihues P, Cavenee WK (eds) Pathology and genetics of tumours of the nervous system. IARC Press, Lyon, pp 141–144
Sawa H, Takeshita I, Kuramitsu M, Mannoji H, Machi T, Fukui M, Kitamura K (1986) Neuronal and glial proteins in medulloblastomas. Immunohistochemical study. Anticancer Res 6(5):905–909
Schild SE, Scheithauer BW, Haddock MG, Wong WW, Lyons MK, Marks LB, Norman MG, Burger PC (1996) Histologically confirmed pineal tumors and other germ cell tumors of the brain. Cancer 78(12):2564–2571
Smits A, van Grieken D, Hartman M, Lendahl U, Funa K, Nistér N (1996) Coexpression of platelet-derived growth factor alpha and beta receptors on medulloblastomas and other primitive neuroectodermal tumors is consistent with an immature stem cell and neuronal derivation. Lab Invest 74:188–198
Van Calenbergh F, D’Haen B, Dom R, Menten J, Plets C (1999) Secondary supratentorial anaplastic astrocytoma following treatment of medulloblastoma. Eur J Peadiatr Neurol 3(4):177–180
Acknowledgements
This work was supported by LUBOM Thuringia of the Ministry for Science, Research, and Art to SAK.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Axel Pagenstecher, Marburg, Germany
This is an interesting case report demonstrating the histopathological course of a supratentorial PNET (sPNET) in a five year old boy. The tumor was surgically removed and treated by irradiation and chemotherapy. Three months later, a low grade astrocytoma developed and in the histological specimens an astrocytic tumor with no detectable neuronal differentiation was observed. Thirteen months later, a glioblastoma was diagnosed. Analysis of LOH of the PNET and the low grade astrocytoma suggested a common clonal origin of both tumors. This case represents an unusual course of a PNET. Supratentorial PNETs often have a grave prognosis, with only about a third of the patients surviving 5 years. The tumors show a strong tendency to seed via the CSF. In the patient presented here, the occurrence of low grade astrocytoma with no PNET residues three months after first surgery is intriguing and raises the question whether there were more malignant portions of the tumor that evaded histological examination. The clinical course of the disease with extremely proliferating glioblastoma 13 months later supports the notion that highly malignant tumor cells remained in situ after the second surgical intervention.
Rights and permissions
About this article
Cite this article
Kuhn, S.A., Hanisch, UK., Ebmeier, K. et al. A paediatric supratentorial primitive neuroectodermal tumour associated with malignant astrocytic transformation and a clonal origin of both components. Neurosurg Rev 30, 143–149 (2007). https://doi.org/10.1007/s10143-007-0069-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-007-0069-5