Abstract
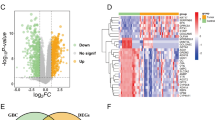
Congenital diaphragmatic hernia (CDH) is an anomaly characterized by a defect in the diaphragm, leading to the passage of intra-abdominal organs into the thoracic cavity. Herein, the presented work analyzes the global gene expression profiles in nine CDH and one healthy newborn. All of the patients had left posterolateral (Bochdalek) diaphragmatic hernia, operated via an abdominal approach, and stomach and bowels in the thorax cavity. Some patients also had additional anomalies. A total of 560 differentially regulated genes were measured. Among them, 11 genes showed significant changes in expression associated with lung tissue, vascular structure development, and vitamin A metabolism, which are typical ontologies related to CDH etiology. Among them, SLC25A24 and RAB3IL1 are involved in angiogenesis, HIF1A and FOXC2-AS1 are related with the alveolus, MAGI2-AS3 is associated with the diaphragm, LHX4 and DHH are linked with the lung, and BRINP1, FZD9, WNT4, and BLOC1S1-RDH5 are involved in retinol. Besides, the expression levels of some previously claimed genes with CDH etiology also showed diverse expression patterns in different patients. All these indicated that CDH is a complex, multigenic anomaly, requiring holistic approaches for its elucidation.




Similar content being viewed by others
Data availability
The generated RNA-seq datasets were uploaded into the NCBI SRA database under the Bioproject ID: PRJNA768307.
Change history
14 June 2022
A Correction to this paper has been published: https://doi.org/10.1007/s10142-022-00872-6
References
Ackerman KG, Herron BJ, Vargas SO, Huang H, Tevosian SG, Kochilas L, Rao C, Pober BR, Babiuk RP, Epstein JA, Greer JJ, Beier DR (2005) Fog2 is required for normal diaphragm and lung development in mice and humans. Plos Genet 1:58–65. https://doi.org/10.1371/journal.pgen.0010010
Amann R, Wyder S, Slavotinek AM, Trueb B (2014) The FgfrL1 receptor is required for development of slow muscle fibers. Dev Biol 394(2):228–241. https://doi.org/10.1016/j.ydbio.2014.08.016
Ameis D, Khoshgoo N, Keijzer R (2017) Abnormal lung development in congenital diaphragmatic hernia. Semin Pediatr Surg 26(3):123–128. https://doi.org/10.1053/j.sempedsurg.2017.04.011
Beck TF, Campeau PM, Jhangiani SN, Gambin T, Li AH, Abo-Zahrah R, Jordan VK, Hernandez-Garcia A, Wiszniewski WK, Muzny D, Gibbs RA, Boerwinkle E, Lupski JR, Lee B, Reardon W, Scott DA (2015) FBN1 contributing to familial congenital diaphragmatic hernia. Am J Med Genet A 167(4):831–836. https://doi.org/10.1002/ajmg.a.36960
Brady PD, Van Houdt J, Callewaert B, Deprest J, Devriendt K, Vermeesch JR (2014) Exome sequencing identifies ZFPM2 as a cause of familial isolated congenital diaphragmatic hernia and possibly cardiovascular malformations. Eur J Med Genet 57(6):247–252. https://doi.org/10.1016/j.ejmg.2014.04.006
Cannata G, Caporilli C, Grassi F, Perrone S, Esposito S (2021) Management of congenital diaphragmatic hernia (CDH): role of molecular genetics. Int J Mol Sci 22(12):6353. https://doi.org/10.3390/ijms22126353
Caprioli A, Vilasenor A, Wylie LA, Braitsch C, Marty-Santos L, Barry D, Karner CM, Fu S, Meadows SM, Carrol TJ, Cleaver O (2015) WNT4 is essential to normal mammalian lung development. Dev Biol 406(2):222–234. https://doi.org/10.1016/j.ydbio.2015.08.017
Chen M, Mac Gowan A, Ward S, Bavik C, Greer JJ (2018) Activation of the retinoid response element is inhibited in an animal model of congenital diaphragmatic hernia. Biol Neonate 83:157–161. https://doi.org/10.1159/000068932
Clugston RD, Klattig J, Engkert C, Clagett-Dame M, Martinovic J, Benachi A, Greer JJ (2006) Teratogen-induced, dietary and genetic models of congenital diaphragmatic hernia share a common mechanism of pathogenesis. Am J Pathol 169(5):1541–1549. https://doi.org/10.2353/ajpath.2006.060445
Dalmer TRA, Clugston RD (2019) Gene ontology enrichment analysis of congenital diaphragmatic hernia-associated genes. Pediatr Res 85(1):13–19. https://doi.org/10.1038/s41390-018-0192-8
Dirami G, Massaro GD, Clerch LB, Ryan US, Reczek PR, Massaro D (2004) Lung retinol storing cells synthesize and secrete retinoic acid, and inducer of alveolus formation. Am J Physiol Lung Cell Mol Physiol 286:249–256. https://doi.org/10.1152/ajplung.00140.2003
Dong X, Xie X, Guo L, Xu J, Xu M, Liang G, Gan L (2019) Generation and characterization of LHX4 (tdT) reporter knock-in and LHX4 (loxP) conditional knocout mice. Genesis 57(10):23328. https://doi.org/10.1002/dvg.23328
Duester G, Mic FA, Molotokov A (2003) Cytosolic retinoid dehydrogenases govern ubiquitous metabolism to retinoic acid. Chem Biol Interact 144:201–210. https://doi.org/10.1016/s0009-2797(02)00204-1
Eckle T, Brodsky K, Bonney M, Packard T, Han J, Borchers CH, Mariani TJ, Kominsky DJ, Mittelbronn M, Eltzschig HK (2013) HIF1A reduces acute lung injury by optimizing carbonhydrate metabolism in the alveolar epithelium. PloS Biol 11(9):1001665. https://doi.org/10.1371/journal.pbio.1001665
Gallot D, Marceau G, Coste K, Hadden H, Robert-Gnansia E, LaurichesseH DPJ, Labbe A, Dastugue B, Lemery D, Sapin V (2005) Congenital diaphragmatic hernia: a retinoid-signaling pathway disruption during lung development? Brith Defects Res A Clin Mol Teratol 73(8):523–531. https://doi.org/10.1002/bdra.20151
Greer JJ, Allan DW, Babiuk RP, Lemke RP (2000) Recent advances in understanding the pathogenesis of nitrofen-induced congenital diaphragmatic hernia. Pediatr Pulmonol 29(5):394–399. https://doi.org/10.1002/(sici)1099-0496(200005)29:5%3c394::aid-ppul9%3e3.0.co;2-2
Greer JJ, Babiuk RP, Thebaud B (2003) Etiology of congenital diaphragmatic hernia: the retinoid hypothesis. Pediatr Res 53(5):726–730. https://doi.org/10.1203/01.PDR.0000062660.12769
Hao XZ, Yang K (2019) LncRNA MAGI2-AS3 suppresses the proliferation and invasion of non-small cell lung carcinoma through miRNA-23A-3P/PTEN axis. Eur Rev Med Pharmacol Sci 23(17):7399–7407. https://doi.org/10.26355/eurrev_201909_18848
Harrison EH (2005) Mechanisms of digestion and absorption of dietary vitamin A. Annu Rev Nutr 25:87–103. https://doi.org/10.1146/annurev.nutr.25.050304.092614
Hind M, Corcoran J, Maden M (2002) Alveolar proliferation, retinoid synthesizing enzymes, and endogenous retinoids in the postnatal Mouse lung. Different roles for Aldh-1, and Aldh-2. Am J Respir Cell Mol Biol 26:67–73. https://doi.org/10.1165/ajrcmb.26.1.4575
Kammoun M, Brady P, De Catte L, Deprest J, Devriendt K, Vermeesch JR (2018) Congenital diaphragmatic hernia as a part of Nance- Horan syndrome? Eur J Hum Genet 26(3):359–366. https://doi.org/10.1038/s41431-017-0032-z
Kardon G, Ackerman KG, McCulley DJ, Shen Y, Wynn J, Shang L, Bogenschutz E, Sun X, Chung WK (2017) Congenital diaphragmatic hernias: from genes to mechanism to therapies. Dis Mode Mech 10(8):955–970. https://doi.org/10.1242/dmm.028365
Katoh M (2008) WNT signaling in stem cell biology and regenerative medicine. Curr Drug Targets 9(7):565–570. https://doi.org/10.2174/138945008784911750
Kling DE, Schnitzer JJ (2007) Vitamin A deficiency (VAD), teratogenic, and surgical models of congenital diaphragmatic hernia (CDH). Am J Med Genet C Semin Med Genet 145C(2):139–157. https://doi.org/10.1002/ajmg.c.30129
Lida K, Hidaka K, Takeuchi M, Nakayama M, Yutani C, Mukai T, Morisaki T (1999) Expression of MEF2 genes during hman cardiac development. Tohoku J Exp Med 187(1):15–23. https://doi.org/10.1620/tjem.187.15
Longoni M, High FA, Russell MK, Kashani A, Tracy AA, Coletti CM, Hila R, Shamia A, Wells J, Ackerman KG, Wilson JM, Bult CJ, Lee C, Lage K, Pober BR, Donahoe PK (2014) Molecular pathogenesis of congenital diaphragmatic hernia revealed by exome sequencing, developmental data, and bioinformatics. Proc Natl Acad Sci U S A 111(34):12450–12455. https://doi.org/10.1073/pnas.1412509111.Sdfgsdg
Mey J, Babiuk RP, Clugston R, Zhang W, Greer JJ (2003) Retinal dehydrogenase-2 is inhibited by compounds that induce congenital diaphragmatic hernias in rodents. Am J Pathol 162(3):673–679. https://doi.org/10.1016/S0002-9440(10)63861-8
Montalva L, Zani A (2019) Assessment of the nitrofen model of congenital diaphragmatic hernia and of the dysregulated factors involved in pulmonary hypoplasia. Pediatr Surg Int 35(1):41–61. https://doi.org/10.1007/s00383-018-4375-5
Ni FB, Lin Z, Fan XH, Shi KQ, Ao JY, Wang XD, Chen RC (2020) A novel genomic-clinicopathologic nomogram to improve prognosis prediction of hepatocellular carcinoma. Clin Chim Acta 504:88–97. https://doi.org/10.1016/j.cca.2020.02.001
Noble BR, Babiuk RP, Clugston RD, Underhill TM, Sun H, Kawaguchi R, Walfish PG, Blomhoff R, Gundersen TE, Greer JJ (2007) Mechanism of action of the congenital diaphragmatic hernia-induced teratogen nitrofen. Am J Physiol Lung Cell Mol Physiol 293(4):1079–1087. https://doi.org/10.1152/ajplung.00286.2007
Paris ND, Coles GL, Ackerman KG (2015) Wt1 and beta-catenin cooperatively regulate diaphragm development in the Mouse. Dev Biol 407(1):40–56. https://doi.org/10.1016/j.ydbio.2015.08.009
Perron M, Boy S, Amato MA, Viczian A, Koebernick K, Pieler T, Harris WA (2003) A novel function for Hedgehog signalling in retinal pigment epithelium differatiation. Dev 130(8):1565–1577. https://doi.org/10.1242/dev.00391
Stolar CJH, Dillon PW (2006) Congenital diaphragmatic hernia and eventration. In Grosfeld JL, O’Neill JA, Fonkalsrud EW, Coran AG editors. Textbook Pediatr Surg Ed Mosby Phila 1(Chap.60):931–954
Takashi T, Friedmacher F, Zimmer J, Puri P (2016) Decreased desmin expression in the developing diaphragm of the nitrofen-induced congenital diaphragmatic hernia rat model. Pediatr Surg Int 32(12):1127–1132. https://doi.org/10.1007/s00383-016-3968-0
Terashima M, Kobayashi M, Motomiya M, Inoue N, Yoshida T, Okano H, Iwasaki N (2010) Analysis of the expression and function of BRINP family genes during neuronal differentiation in Mouse embryonic stem cell-derived neural stem cells. J Neurosci Res 88(7):1387–1393. https://doi.org/10.1002/jnr.22315
Unger S, Copland I, Tibboel D, Post M (2003) Down-regulation of sonic hedgehog expression in pulmonary hypoplasia is associated with congenital diaphragmatic hernia. Am J Pathol 162(2):547–555. https://doi.org/10.1016/S0002-9440(10)63848-5
Urano T, Shiraki M, Sasaki M, Ouchi Y, Inoue S (2015) SLC25A24 as a novel susceptibility gene for low fat mass in humans and mice. J Clin Endocrinol Metab 100(4):655–663. https://doi.org/10.1210/jc.2014-2829
Van Loenhout RB, Tibboel D, Post M, Keijzer R (2009) Congenital diaphragmatic hernia: comparison of animal models and relevance to the human situation. Neonatol 96(3):137–149. https://doi.org/10.1159/000209850
Wang YQ, Xu ZM, Wang XL, Zheng JK, Du Q, Yang JX, Zhang HC (2020) LncRNAFOXC2-AS1 regulated proliferation and apoptosis of vascular smooth muscle cell through targeting miR-1253/FOXF1 axis in atherosclerosis. Eur Rev Med Pharmacol Sci 24(6):3302–3314. https://doi.org/10.26355/eurrev_202003_20698
You LR, Takamato N, Yu CT, Kodama T, Demayo FJ, Tsai SY, Tsai MJ (2005) Mouse lacking COUP-TFII as an animal model of Bochdaleck-type congenital diaphragmatic hernia. Proc Natl Acad Sci USA 102(45):16351–16356. https://doi.org/10.1073/pnas.0507832102
Yu L, Bennett JT, Wynn J, Carvill GL, Cheung YH, Shen Y, Mychaliska GB, Azarow KS, Crombleholme TM, Chung DH, Potoka D, Warner BW, Bucher B, Lim FY, Pietsch J, Stolar C, Aspelund G, Arkovitz MS, Mefford H, Chung WK (2014) Whole exome sequencing identifies de novo mutations in GATA6 associated with congenital diaphragmatic hernia. J Med Genet. 51(3):197–202. https://doi.org/10.1136/jmedgenet-2013-101989
Yu L, Hernan RR, Wynn J, Chung WK (2020) The influence of genetics in congenital diaphragmatic hernia. Semin Perinatol 44(1):151–169. https://doi.org/10.1053/j.semperi.2019.07.008
Zhaorigetu S, Bair H, Lu J, Jin D, Olson SD, Harting MT (2018) Pertubartions in endothelial dysfunction- associated pathways in the nitrofen induced congenital diaphragmatic hernia model. J Vasc Res 55(1):26–34. https://doi.org/10.1159/000484087
Acknowledgements
We would like to thank Turan YILDIZ for his contribution to this study.
Funding
This work was supported by Inonu University Scientific Research Project Coordination Unit (project code: TSA-2021–2454).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: In the Introduction part, 4th sentence, "It is thought to be caused by a developmental defect of the pleuroperitoneal membrane, one of the four parts of the diaphragm (Ameis et al. 2017)" should be replaced with: "Its cause has not been fully known but genetic defects are thought to play roles in the etiology due to its emergence in the early period (embryologic); lung and heart anomalies frequently accompanied by this disease (Ameis et al. 2017)
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gürünlüoğlu, K., Dündar, M., Unver, T. et al. Global gene expression profiling in congenital diaphragmatic hernia (CDH) patients. Funct Integr Genomics 22, 359–369 (2022). https://doi.org/10.1007/s10142-022-00837-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10142-022-00837-9