Abstract
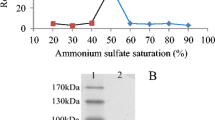
A hyaluronate lyase (BniHL) was purified to homogeneity from a culture of a deep-sea Bacillus niacin strain JAM F8. The molecular mass of purified BniHL was approximately 120 kDa. The purified enzyme degraded hyaluronan as well as chondroitin sulfates A and C by a β-elimination mechanism. The optimal pH and temperature were around pH 6 and 45 °C for hyaluronan degradation. The enzyme required optimally 2, 50, and 100 mM calcium ions for degradation of hyaluronan, chondroitin sulfate C, and chondroitin sulfate A, respectively. Calcium ions slightly increased the thermal stability of the enzyme. In a genome analysis of strain JAM F8, a BniHL coding gene was identified on the bases of the molecular mass and N-terminal and internal amino acid sequences. The gene consisted of 3411 nucleotides and coded 1136 amino acids. The deduced amino acid sequence showed the highest similarity to the hyaluronate lyase of a Bacillus sp. A50 with 89 % identity.







Similar content being viewed by others
References
Bellamy WR (1990) A novel Bacillus sp. capable of degrading sulfated glycosaminoglycans. In: Horikoshi K, Grant WD (eds) Superbugs: microorganisms in extreme environments. Japan Scientific Societies Press, Tokyo, pp 143–157
Cássaro CMF, Dietrich CP (1977) Distribution of sulfated mucopolysaccharides in invertebrates. J Biol Chem 252:2254–2261
Girish KS, Kemparaju K (2007) The magic glue hyaluronan and its eraser hyaluronidase: a biological overview. Life Sci 80:1921–1943
Guo X, Shi Y, Sheng J, Wang F (2014) A novel hyaluronidase produced by Bacillus sp. A50. PLoS One 9:e94156
Hamai A, Hashimoto N, Mochizuki H, Kato F, Makiguchi Y, Horie K, Suzuki S (1997) Two distinct chondroitin sulfate ABC lyases. J Biol Chem 272:9123–9130
Hiyama K, Okada S (1975) Crystallization and some properties of chondroitinase from Arthrobacter aurescens. J Biol Chem 250:1824–1828
Hong SW, Kim BT, Shin HY, Kim WS, Lee KS, Kim YS, Kim DH (2002) Purification and characterization of novel chondroitin ABC and AC lyases from Bacteroides stercoris HJ-15, a human intestinal anaerobic bacterium. Eur J Biochem 269:2934–2940
Ingham E, Holland KT, Gowland G, Cunliffe WJ (1979) Purification and partial characterization of hyaluronate lyase (EC 4.2.2.1) from Propionibacterium acnes. J Gen Microbiol 115:411–418
Jedrzejas MJ (2000) Structural and functional comparison of polysaccharide-degrading enzymes. Crit Rev Biochem Mol Biol 35:221–251
Jedrzejas MJ, Mello LV, de Groot BL, Li S (2002) Mechanism of hyaluronan degradation by Streptococcus pneumoniae hyaluronate lyase. J Biol Chem 277:28287–28297
Kitamikado M, Lee YZ (1975) Chondroitinase-producing bacteria in natural habitats. Appl Environ Microbiol 29:414–421
Kurata A, Nishimura M, Kishimoto N, Kobayashi T (2014) Draft genome sequence of a deep-sea bacterium, Bacillus niacini strain JAM F8, involved in the degradation of glycosaminoglycans. Genome Announc 2:e00983–14. doi:10.1128/genomeA.00983-14
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Li S, Jedrzejas MJ (2001) Hyaluronan binding and degradation by Streptococcus agalactiae hyaluronate lyase. J Biol Chem 276:41407–41416
Lin B, Hollingshead SK, Coligan JE, Egan ML, Baker JR, Pritchard DG (1994) Cloning and expression of the gene for group B streptococcal hyaluronate lyase. J Biol Chem 269:30113–30116
Linn S, Chan T, Lipeski L, Salyers AA (1983) Isolation and characterization of two chondroitin lyases from Bacteroides thetaiotaomicron. J Bateriol 156:859–866
Nukui M, Taylor KB, McPherson DT, Shigenaga MK, Jedrzejas MJ (2003) The function of hydrophobic residues in the catalytic cleft of Streptococcus pneumoniae hyaluronate lyase. J Biol Chem 278:3079–3088
Petit E, Delattre C, Papy-Garcia D, Michaud P (2006) Chondroitin sulfate lyases: applications in analysis and glycobiology. Adv Pharmacol 53:167–186
Pritchard DG, Trent JO, Zhang P, Egan ML, Baker JR (2000) Characterization of the active site of group B streptococcal hyaluronate lyase. Proteins 40:126–134
Senni K, Pereira J, Gueniche F, Delbarre-Ladrat C, Sinquin C, Ratiskol J, Godeau G, Fischer AM, Helley D, Colliec-Jouault S (2011) Marine polysaccharides: a source of bioactive molecules for cell therapy and tissue engineering. Mar Drugs 9:1664–1681
Smith RF, Willett NP (1968) Rapid plate method for screening hyaluronidase and chondroitin sulfatase-producing microorganisms. Appl Environ Microbiol 16:1434–1436
Tam YC, Chan ESC (1985) Purification and characterization of hyaluronidase from oral Peptostreptococcus species. Infect Immun 47:508–513
Volpi N, Maccari F (2003) Purification and characterization of hyaluronic acid from the mollusk bivalve Mytilus galloprovincialis. Biochimie 85:619–625
Winter WT, Arnott S (1977) Hyaluronic acid : the role of divalent cations in conformation and packing. J Mol Biol 117:761–784
Yasuda S, Sugahara K, Özbek S (2011) Evolution of glycosaminoglycans: comparative biochemical study. Commun Integr Biol 4:150–158
Author information
Authors and Affiliations
Corresponding authors
Additional information
Atsushi Kurata and Tohru Kobayashi contributed equally to this work.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Fig. S1
ESI-MS/MS spectrograms of degradation products from HA and ChS-C. A, spectrogram of degradation products from HA; B, that of authentic ∆Di-HA; C, that of degradation products from ChS-C; D that of authentic ∆Di-6S. (PPTX 134 kb)
Fig. S2
Diagram of domain structure of BniHL. Amino acid numbers are shown above diagram. Each domain is shown as follows:, CBM_4_9 superfamily domain; tandem Big_2 domains (bacterial Ig-like domain); glycosaminoglycan (GAG)-lyase superfamily domain; PL family 8N-terminal alpha helical domain; PL family 8 super- sandwich domain (PPTX 49 kb)
ESM 1
CBM_4_9 superfamily domain (GIF 905 bytes)
ESM 2
tandem Big_2 domains (bacterial Ig-like domain) (GIF 1088 bytes)
ESM 3
glycosaminoglycan (GAG)-lyase superfamily domain (GIF 905 bytes)
ESM 4
PL family 8N-terminal alpha helical domain (GIF 1.32 kb)
ESM 5
PL family 8 super- sandwich domain (GIF 997 bytes)
Table S1
(PPTX 51 kb)
Table S2
(PPTX 51 kb)
Rights and permissions
About this article
Cite this article
Kurata, A., Matsumoto, M., Kobayashi, T. et al. Hyaluronate Lyase of a Deep-Sea Bacillus niacini . Mar Biotechnol 17, 277–284 (2015). https://doi.org/10.1007/s10126-015-9618-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-015-9618-z