Abstract
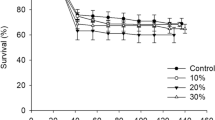
We investigated the effect of two commercial nucleotide products (NT1 and NT2), administered through live feed, on growth and stress tolerance of Atlantic cod larvae. Expression of genes related to muscle growth (igf-1, igf1r, igf-2, fst, fgf6, myod, and myhc) and nucleotide metabolism (uox, hprt, ndk, and uck) was evaluated during larval development. In addition, the expression of genes related to stress (hif-1α, hif-2α, hif-3α, and mb) was studied after an air exposure stress test. The enrichment of rotifers with nucleotides did not reveal any difference in nucleotide profiles, the exception being the RNA level of the NT1-enriched group that was significantly higher than the unenriched rotifer. Unenriched Artemia showed poor nucleotide profiles compared to enriched Artemia since 5′ UMP, 5′ GMP, and 5′ AMP were observed only in the nucleotide groups. At 38 days post-hatch (dph), NT1 group had significantly higher dry weight (3.1 ± 0.1 mg) than the control (CON; 2.3 ± 0.1 mg). The treatments did not produce any significant differences in the expression of the key myogenic genes. Among the genes associated with nucleotide metabolism, ndk was down-regulated in NT1 at 38 dph. In the air exposure test, survival was significantly higher in the CON (77 ± 6 %) than in NT1 (48 ± 3 %) and NT2 (50 ± 3 %). After air exposure, mb was expressed at lower levels in NT2 group, hif-2α was induced in NT1 group, and hif-3α was upregulated in all groups. Our findings indicate that the improvement in the nucleotide profile of Artemia upon nucleotide enrichment could eventuate in the rapid growth of larvae.





Similar content being viewed by others
References
Alami-Durante H, Médale F, Cluzeaud M, Kaushik SJ (2010a) Skeletal muscle growth dynamics and expression of related genes in white and red muscles of rainbow trout fed diets with graded levels of a mixture of plant protein sources as substitutes for fishmeal. Aquaculture 303:50–58
Alami-Durante H, Wrutniak-Cabello C, Kaushik SJ, Médale F (2010b) Skeletal muscle cellularity and expression of myogenic regulatory factors and myosin heavy chains in rainbow trout (Oncorhynchus mykiss): effects of changes in dietary plant protein sources and amino acid profiles. Comp Biochem Physiol A 156:561–568
Andersen Ø, Aas TS, Skugor S, Takle H, van Nes S, Grisdale-Helland B, Helland SJ, Terjesen BF (2006) Purine-induced expression of urate oxidase and enzyme activity in Atlantic salmon (Salmo salar). FEBS J 273:2839–2850
Ayson FG, de Jesus EG, Moriyama S, Hyodo S, Funkenstein B, Gertler A, Kawauchi H (2002) Differential expression of insulin-like growth factor I and II mRNAs during embryogenesis and early larval development in rabbitfish, Siganus guttatus. Gen Comp Endocrinol 126:165–174
Baskerville-Bridges B, Kling LJ (2000) Early weaning of Atlantic cod (Gadus morhua) larvae onto a microparticulate diet. Aquaculture 189:109–117
Burrells C, Williams PD, Southgate PJ, Wadsworth SL (2001) Dietary nucleotides: a novel supplement in fish feeds: 2. Effects on vaccination, salt water transfer, growth rates and physiology of Atlantic salmon (Salmo salar L.). Aquaculture 199:171–184
Busch KET, Falk-Petersen IB, Peruzzi S, Rist NA, Hamre K (2010) Natural zooplankton as larval feed in intensive rearing systems for juvenile production of Atlantic cod (Gadus morhua L.). Aquac Res 41:1727–1740
Campos C, Valente LMP, Borges P, Bizuayehu T, Fernandes JMO (2010) Dietary lipid levels have a remarkable impact on the expression of growth-related genes in Senegalese sole (Solea senegalensis Kaup). J Exp Biol 213:200–209
Carver JD (1999) Dietary nucleotides: effects on the immune and gastrointestinal systems. Acta Paediatr Suppl 88:83–88
Chapalamadugu KC, Robison BD, Drew RE, Powell MS, Hill RA, Amberg JJ, Rodnick KJ, Hardy RW, Hill ML, Murdoch GK (2009) Dietary carbohydrate level affects transcription factor expression that regulates skeletal muscle myogenesis in rainbow trout. Comp Biochem Physiol B 153:66–72
Cheng Z, Buentello A, Gatlin DM III (2011) Dietary nucleotides influence immune responses and intestinal morphology of red drum Sciaenops ocellatus. Fish Shellfish Immunol 30:143–147
Conceição LEC, Yúfera M, Makridis P, Morais S, Dinis MT (2010) Live feeds for early stages of fish rearing. Aquac Res 41:613–640
Cosgrove M (1998) Nucleotides. Nutrition 14:748–751
De-Santis C, Jerry DR (2007) Candidate growth genes in finfish—where should we be looking? Aquaculture 272:22–38
Eliès G, Duval H, Bonnec G, Wolff J, Boeuf G, Boujard D (1999) Insulin and insulin-like growth factor-1 receptors in an evoluted fish, the turbot: cDNA cloning and mRNA expression. Mol Cell Endocrinol 158:173–185
Falk-Petersen IB (2005) Comparative organ differentiation during early life stages of marine fish. Fish Shellfish Immunol 19:397–412
Fernandes JMO, Kinghorn JR, Johnston IA (2007) Differential regulation of multiple alternatively spliced transcripts of MyoD. Gene 391:178–185
Fernandes JMO, Mommens M, Hagen Ø, Babiak I, Solberg C (2008) Selection of suitable reference genes for real-time PCR studies of Atlantic halibut development. Comp Biochem Physiol B 150:23–32
Florini JR, Ewton DZ, Coolican SA (1996) Growth hormone and the insulin-like growth factor system in myogenesis. Endocr Rev 17:481–517
Funes V, Asensio E, Ponce M, Infante C, Canavate JP, Manchado M (2006) Insulin-like growth factors I and II in the sole Solea senegalensis: cDNA cloning and quantitation of gene expression in tissues and during larval development. Gen Comp Endocrinol 149:166–172
Funkenstein B, Rebhan Y, Skopal T (2009) Molecular cloning and characterization of follistatin in the gilthead sea bream, Sparus aurata. Mol Biol Rep 36:501–511
Galloway TF, Kjørsvik E, Kryvi H (1999) Muscle growth and development in Atlantic cod larvae (Gadus morhua L.), related to different somatic growth rates. J Exp Biol 202:2111–2120
Gauvry L, Fauconneau B (1996) Cloning of a trout fast skeletal myosin heavy chain expressed both in embryo and adult muscles and in myotubes neoformed in vitro. Comp Biochem Physiol B 115:183–190
Greene MW, Chen TT (1999) Quantitation of IGF-I, IGF-II, and multiple insulin receptor family member messenger RNAs during embryonic development in rainbow trout. Mol Reprod Dev 54:348–361
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hall TE, Cole NJ, Johnston IA (2003) Temperature and the expression of seven muscle-specific protein genes during embryogenesis in the Atlantic cod Gadus morhua L. J Exp Biol 206:3187–3200
Heidbreder M, Fröhlich F, Jöhren O, Dendorfer A, Qadri F, Dominiak P (2003) Hypoxia rapidly activates HIF-3alpha mRNA expression. FASEB J 17:1541–1543
Hevrøy EM, Jordal A-EO, Hordvik I, Espe M, Hemre G-I, Olsvik PA (2006) Myosin heavy chain mRNA expression correlates higher with muscle protein accretion than growth in Atlantic salmon, Salmo salar. Aquaculture 252:453–461
Hill JJ, Davies MV, Pearson AA, Wang JH, Hewick RM, Wolfman NM, Qiu Y (2002) The myostatin propeptide and the follistatin-related gene are inhibitory binding proteins of myostatin in normal serum. J Biol Chem 277:40735–40741
Hiroi J, Sakakura Y, Tagawa M, Seikai T, Tanaka M (1997) Developmental changes in low-salinity tolerance and responses of prolactin, cortisol and thyroid hormones to low-salinity environment in larvae and juveniles of Japanese flounder, Paralichthys olivaceus. Zool Sci 14:987–992
Jiralerspong S, Patel PI (1996) Regulation of the hypoxanthine phosphoribosyltransferase gene: in vitro and in vivo approaches. Proc Soc Exp Biol Med 212:116–127
Johansen KA, Overturf K (2005) Quantitative expression analysis of genes affecting muscle growth during development of rainbow trout (Oncorhynchus mykiss). Mar Biotechnol 7:576–587
Keyvanshokooh S, Tahmasebi-Kohyani A (2012) Proteome modifications of fingerling rainbow trout (Oncorhynchus mykiss) muscle as an effect of dietary nucleotides. Aquaculture 324–325:79–84
King VW, Berlinsky DL (2006) Whole-body corticosteroid and plasma cortisol concentrations in larval and juvenile Atlantic cod Gadus morhua L. following acute stress. Aquac Res 37:1282–1289
Kortner TM, Overrein I, Øie G, Kjørsvik E, Arukwe A (2011) The influence of dietary constituents on the molecular ontogeny of digestive capability and effects on growth and appetite in Atlantic cod larvae (Gadus morhua). Aquaculture 315:114–120
Law SHW, Wu RSS, Ng PKS, Yu RMK, Kong RYC (2006) Cloning and expression analysis of two distinct HIF-alpha isoforms—gcHIF-1alpha and gcHIF-4alpha—from the hypoxia-tolerant grass carp. Ctenopharyngodon idellus. BMC Mol Biol 7:15
Li P, Gatlin DM III (2006) Nucleotide nutrition in fish: current knowledge and future applications. Aquaculture 251:141–152
Li P, Burr GS, Goff J, Whiteman KW, Davis KB, Vega RR, Neill WH, Gatlin DM III (2005) A preliminary study on the effects of dietary supplementation of brewers yeast and nucleotides, singularly or in combination, on juvenile red drum (Sciaenops ocellatus). Aquac Res 36:1120–1127
Lin YH, Wang H, Shiau SY (2009) Dietary nucleotide supplementation enhances growth and immune responses of grouper, Epinephelus malabaricus. Aquac Nutr 15:117–122
Lopez-Navarro AT, Ortega MA, Peragón J, Bueno JD, Gil A, Sánchez-Pozo A (1996) Deprivation of dietary nucleotides decreases protein synthesis in the liver and small intestine in rats. Gastroenterology 110:1760–1769
Maloney D (1998) Yeasts. In: Kroschwitz JI, Howe-Grant M (eds) Kirk–Othmer encyclopedia of chemical technology, 4th edn. Wiley, New York, pp 761–788
McCollum A, Geubtner J, von Herbing IH (2006) Metabolic cost of feeding in Atlantic cod (Gadus morhua) larvae using microcalorimetry. ICES J Mar Sci 63:335–339
Peterson BC, Bosworth BG, Bilodeau AL (2005) Differential gene expression of IGF-I, IGF-II, and toll-like receptors 3 and 5 during embryogenesis in hybrid (channel × blue) and channel catfish. Comp Biochem Physiol A 141:42–47
Ponce M, Infante C, Funes V, Manchado M (2008) Molecular characterization and gene expression analysis of insulin-like growth factors I and II in the redbanded seabream, Pagrus auriga: transcriptional regulation by growth hormone. Comp Biochem Physiol B 150:418–426
Rahman MS, Thomas P (2007) Molecular cloning, characterization and expression of two hypoxia-inducible factor alpha subunits, HIF-1alpha and HIF-2alpha, in a hypoxia-tolerant marine teleost, Atlantic croaker (Micropogonias undulatus). Gene 396:273–282
Rescan PY (1998) Identification of a fibroblast growth factor 6 (FGF6) gene in a non-mammalian vertebrate: continuous expression of FGF6 accompanies muscle fiber hyperplasia. Biochim Biophys Acta 1443:305–314
Rescan PY (2005) Muscle growth patterns and regulation during fish ontogeny. Gen Comp Endocrinol 142:111–116
Ricker WE (1958) Handbook of computations for biological statistics of fish populations. Bull Fish Res Board Can 119:1–30
Sæle Ø, Nordgreen A, Hamre K, Olsvik PA (2009) Evaluation of candidate reference genes in Q-PCR studies of Atlantic cod (Gadus morhua) ontogeny, with emphasis on the gastrointestinal tract. Comp Biochem Physiol B 152:94–101
Sánchez-Pozo A, Gil A (2002) Nucleotides as semi-essential nutritional components. Br J Nutr 87:S135–S137
Sauer N, Mosenthin R, Bauer E (2011) The role of dietary nucleotides in single-stomached animals. Nutr Res Rev 24:46–59
Shen R-J, Jiang X-Y, Pu J-W, Zou S-M (2010) HIF-1α and -2α genes in a hypoxia-sensitive teleost species Megalobrama amblycephala: cDNA cloning, expression and different responses to hypoxia. Comp Biochem Physiol B 157:273–280
Soitamo AJ, Råbergh CMI, Gassmann M, Sistonen L, Nikinmaa M (2001) Characterization of a hypoxia-inducible factor (HIF-1α) from rainbow trout. J Biol Chem 276:19699–19705
Tahmasebi-Kohyani A, Keyvanshokooh S, Nematollahi A, Mahmoudi N, Pasha-Zanoosi H (2011) Dietary administration of nucleotides to enhance growth, humoral immune responses, and disease resistance of the rainbow trout (Oncorhynchus mykiss) fingerlings. Fish Shellfish Immunol 30:189–193
Terova G, Rimoldi S, Corà S, Bernadini G, Gornati R, Saroglia M (2008) Acute and chronic hypoxia affects HIF-1α mRNA levels in sea bass (Dicentrarchus labrax). Aquaculture 279:150–159
Uauy R, Quan R, Gil A (1994) Role of nucleotides in intestinal development and repair: implications for infant nutrition. J Nutr 124:1436S–1441S
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:RESEARCH0034
Walsh MJ, Sanchez-Pozo A, Leleiko NS (1990) A regulatory element is characterized by purine-mediated and cell-type-specific gene transcription. Mol Cell Biol 10:4536–4364
Walsh MJ, Tsao K-L, Leleiko NS (1992) Characterization of DNA–protein interactions within a distal regulatory element upstream of a mammalian housekeeping gene promoter. J Biol Chem 267:7026–7035
Welker TL, Lim C, Yildirim-Aksoy M, Klesius PH (2011) Effects of dietary supplementation of a purified nucleotide mixture on immune function and disease and stress resistance in channel catfish, Ictalurus punctatus. Aquac Res 42:1878–1889
Welt C, Sidis Y, Keutmann H, Schneyer A (2002) Activins, inhibins, and follistatins: from endocrinology to signaling. A paradigm for the new millennium. Exp Biol Med 227:724–752
Werner A (1991) Analysis of nucleotides, nucleosides, nucleobases in cells by ion-pair reversed-phase HPLC. Chromatographia 31:401–410
Yousefi M, Abtahi B, Kenari AA (2011) Hematological, serum biochemical parameters, and physiological responses to acute stress of Beluga sturgeon (Huso huso, Linnaeus 1785) juveniles fed dietary nucleotide. Comp Clin Path 1-6. doi:10.1007/00580-011-1225-4
Acknowledgments
The study was funded by the Research Council of Norway, project 182653/V10. The authors appreciate the excellent technical assistance provided by Bjørnar Eggen, Magnus Røkke, and Heidi Ludviksen during live feed production and collection of samples. The support of Rudi Ripman Seim and Odd Elvebø from Biorigin Scandinavia AS, Oslo, Norway, is gratefully acknowledged. Peter Koeppel, Chemoforma Ltd, Augst, Switzerland, is thanked for gifting one of the nucleotide products. The nucleotide analysis of the live feeds was kindly performed by Biorigin Brazil A/S, São Paulo, Brazil. Norwegian Cod Breeding Centre, Tromsø, Norway, supplied the eggs for the study. C.F.C. Lanes is a Ph.D. student supported by funds from Research Council of Norway and University of Nordland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lanes, C.F.C., Bolla, S., Fernandes, J.M.O. et al. Nucleotide Enrichment of Live Feed: A Promising Protocol for Rearing of Atlantic Cod Gadus morhua Larvae. Mar Biotechnol 14, 544–558 (2012). https://doi.org/10.1007/s10126-012-9458-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-012-9458-z