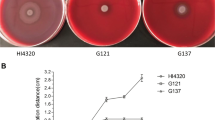
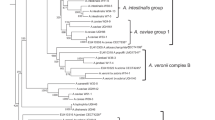
Abstract
In current times, the opportunistic pathogen Morganella morganii is increasingly becoming a cause of urinary tract infections. The condition has been further complicated by the multiple drug resistance of most isolates. Swimming motility plays an important role in the development of urinary tract infections, allowing bacteria to colonize the upper urinary tract. We determined the differences between the growth, swimming motility, and biofilm formation of two M. morganii strains MM 1 and MM 190 isolated from the urine of patients who had community-acquired urinary tract infections. MM 190 showed a lower growth rate but better-formed biofilms in comparison to MM 1. In addition, MM 190 possessed autoaggregation abilities. It was found that a high temperature (37 °C) inhibits the flagellation of strains and makes MM 190 less motile. At the same time, the MM 1 strain maintained its rate of motility at this temperature. We demonstrated that urea at a concentration of 1.5% suppresses the growth and swimming motility of both strains. Genome analysis showed that MM 1 has a 17.7-kb-long insertion in flagellar regulon between fliE and glycosyltransferase genes, which was not identified in corresponding loci of MM 190 and 9 other M. morganii strains with whole genomes. Both strains carry two genes encoding flagellin, which may indicate flagellar antigen phase variation. However, the fliC2 genes have only 91% identity to each other and exhibit some variability in the regulatory region. We assume that all these differences influence the swimming motility of the strains.







Similar content being viewed by others
Data Availability
The datasets analyzed during the current study are available in the DDBJ/EMBL/GenBank repositories, QUOO00000000 and QMKL00000000.
Code availability
Not applicable.
References
Abdalla J, Saad M, Samnani I, Lee P, Moorman J (2006) Central nervous system infection caused by Morganella morganii. Am J Med Sci 331:44–47. https://doi.org/10.1097/00000441-200601000-00013
Akerley BJ, Miller JF (1993) Flagellin gene transcription in Bordetella bronchiseptica is regulated by the BvgAS virulence control system. J Bacteriol 175:3468–3479. https://doi.org/10.1128/jb.175.11.3468-3479.1993
Alcántar-Curiel MD, Blackburn D, Saldaña Z, Gayosso-Vázquez C, Iovine NM, De la Cruz MA, Girón JA (2013) Multi-functional analysis of Klebsiella pneumoniae fimbrial types in adherence and biofilm formation. Virulence 4:129–138. https://doi.org/10.4161/viru.22974
Appelt S, Heuner K (2017) The flagellar regulon of Legionella – a review. Front Cell Infect Microbiol 7:454. https://doi.org/10.3389/fcimb.2017.00454
Armbruster CE, Prenovost K, Mobley HL, Mody L (2017) How often do clinically diagnosed catheter-associated urinary tract infections in nursing homes meet standardized criteria? J Am Geriatr Soc 65:395–401. https://doi.org/10.1111/jgs.14533
Atalay H, Güney I, Solak Y, Almaz E (2010) First case of CAPD-related peritonitis caused by Morganella morganii. Perit Dial Int 30:119–121. https://doi.org/10.3747/pdi.2008.00265
Bandy A (2020) Ringing bells: Morganella morganii fights for recognition. Public Health 182:45–50. https://doi.org/10.1016/j.puhe.2020.01.016
Bertelli C, Laird MR, Williams KP, Lau BY, Hoad G, Winsor GL, Brinkman FSL (2017) Islandviewer 4: expanded prediction of genomic islands for larger-scale datasets. Nucleic Acids Res 45:30–35. https://doi.org/10.1093/nar/gkx343
Brahmi S, Touati A, Dunyach-Remy C, Sotto A, Pantel A, Lavigne JP (2018) High prevalence of extended-spectrum β-lactamase-producing Enterobacteriaceae in wild fish from the Mediterranean Sea in Algeria. Microb Drug Resist 24:290–298. https://doi.org/10.1089/mdr.2017.0149
Brízio A, Vasco S, Conceição T, Lito L, Melo-Cristino J, Salgado MJ, Duarte A (2006) First report of Morganella morganii producing CTX-M-15 beta-lactamase. Int J Antimicrob Agents 28:79–80. https://doi.org/10.1016/j.ijantimicag.2006.03.004
Chaban B, Hughes HV, Beeby M (2015) The flagellum in bacterial pathogens: for motility and a whole lot more. Semin Cell Dev Biol 46:91–103. https://doi.org/10.1016/j.semcdb.2015.10.032
Chen YT, Peng HL, Shia WC, Hsu FR, Ken CF, Tsao YM, Chen CH, Liu CE, Hsieh MF, Chen HC (2012) Whole-genome sequencing and identification of Morganella morganii KT pathogenicity-related genes. BMC Genomics. https://doi.org/10.1186/1471-2164-13-S7-S4
Chen Y, Lei C, Zuo L, Kong L, Kang Z, Zeng J, Zhang X, Wang H (2019) A novel cfr-carrying Tn7 transposon derivative characterized in Morganella morganii of swine origin in China. J Antimicrob Chemother 74:603–606. https://doi.org/10.1093/jac/dky494
Chilcott GS, Hughes KT (2000) Coupling of flagellar gene expression to flagellar assembly in Salmonella enterica serovar Typhimurium and Escherichia coli. Microbiol Mol Biol Rev 64:694–708. https://doi.org/10.1128/MMBR.64.4.694-708.2000
Coetzee JN, Deklerk HC (1964) Effect of temperature on flagellation, motility and swarming of Proteus. Nature 202:211–212. https://doi.org/10.1038/202211b0
da Silva Meira QG, de Medeiros BI, Alves Aguiar Athayde AJ, de Siqueira-Júnior JP, de Souza EL (2012) Influence of temperature and surface kind on biofilm formation by Staphylococcus aureus from food-contact surfaces and sensitivity to sanitizers. Food Control 25:469–475. https://doi.org/10.1016/j.foodcont.2011.11.030
Dattelbaum JD, Lockatell CV, Johnson DE, Mobley HL (2003) UreR, the transcriptional activator of the Proteus mirabilis urease gene cluster, is required for urease activity and virulence in experimental urinary tract infections. Infect Immun 71:1026–1030. https://doi.org/10.1128/IAI.71.2.1026-1030.2003
de Kerchove AJ, Elimelech M (2008) Bacterial swimming motility enhances cell deposition and surface coverage. Environ Sci Technol 42:4371–4377. https://doi.org/10.1021/es703028u
De Silva PM, Chong P, Fernando DM, Westmacott G, Kumar A (2018) Effect of incubation temperature on antibiotic resistance and virulence factors of Acinetobacter baumannii ATCC 17978. Antimicrob Agents Chemother. https://doi.org/10.1128/AAC.01514-17
De A, Raj HJ, Maiti PK (2016) Biofilm in osteomyelitis caused by a rare pathogen, Morganella morganii: a case report. J Clin Diagn Res 10:DD06–DD08. https://doi.org/10.7860/JCDR/2016/18666.7990
De A, Raj HJ, Haldar J, Mukherjee P, Maiti PK (2017) Biofilm colonization in chronic treatment refractory infections presenting with discharging sinuses: a study in a tertiary care hospital of Eastern India. J Lab Physicians 9:125–131. https://doi.org/10.4103/0974-2727.199637
Di Bonaventura G, Piccolomini R, Paludi D, D’Orio V, Vergara A, Conter M, Ianieri A (2008) Influence of temperature on biofilm formation by Listeria monocytogenes on various food-contact surfaces: relationship with motility and cell surface hydrophobicity. J Appl Microbiol 104:1552–1561. https://doi.org/10.1111/j.1365-2672.2007.03688.x
Eberspächer B, Hugo F, Pohl M, Bhakdi S (1990) Functional similarity between the haemolysins of Escherichia coli and Morganella morganii. J Med Microbiol 33:165–170. https://doi.org/10.1099/00222615-33-3-165
Emborg J, Dalgaard P, Ahrens P (2006) Morganella psychrotolerans sp. nov., a histamine-producing bacterium isolated from various seafoods. Int J Syst Evol Microbiol 56:2473–2479. https://doi.org/10.1099/ijs.0.64357-0
Erlanger D, Assous MV, Wiener-Well Y, Yinnon AM, Ben-Chetrit E (2019) Clinical manifestations, risk factors and prognosis of patients with Morganella morganii sepsis. J Microbiol Immunol Infect 52:443–448. https://doi.org/10.1016/j.jmii.2017.08.010
Fitzgerald DM, Bonocora RP, Wade JT (2014) Comprehensive mapping of the Escherichia coli flagellar regulatory network. PLoS Genet. https://doi.org/10.1371/journal.pgen.1004649
Fuerst JA, Hayward AC (1980) The effect of temperature on the formation of sheathed flagella by Pseudomonas stizolobii. J Gen Microbiol 117:111–117. https://doi.org/10.1099/00221287-117-1-111
Gal-Mor O, Finlay BB (2006) Pathogenicity islands: a molecular toolbox for bacterial virulence. Cell Microbiol 8:1707–1719. https://doi.org/10.1111/j.1462-5822.2006.00794.x
Gendlina I, Gutman DM, Thomas V, Collins CM (2002) Urea-dependent signal transduction by the virulence regulator UreR. J Biol Chem 277:37349–37358. https://doi.org/10.1074/jbc.M203462200
Glinskaya EV, Al-Bayati BM, Nechaeva OV, Luneva IO (2015) Antibiotic susceptibility of pathogens in urinary tract infections in community. Izv Sarat un-ta 15:63–66. https://doi.org/10.18500/1816-9775-2015-15-4-63-66
Gründling A, Burrack LS, Bouwer HG, Higgins DE (2004) Listeria monocytogenes regulates flagellar motility gene expression through MogR, a transcriptional repressor required for virulence. Proc Natl Acad Sci U S A 101:12318–12323. https://doi.org/10.1073/pnas.0404924101
Guzman JPMD, De Las Alas TPL, Lucban MC, Sevilla CEC (2020) Green tea (Camellia sinensis) extract inhibits biofilm formation in acyl homoserine lactone-producing, antibiotic-resistant Morganella morganii isolated from Pasig River, Philippines. Heliyon. https://doi.org/10.1016/j.heliyon.2020.e05284
Ha DG, Kuchma SL, O’Toole GA (2014) Plate-based assay for swimming motility in Pseudomonas aeruginosa. Methods Mol Biol 1149:59–65. https://doi.org/10.1007/978-1-4939-0473-0_7
Hacker J, Kaper JB (2000) Pathogenicity islands and the evolution of microbes. Annu Rev Microbiol 54:641–679. https://doi.org/10.1146/annurev.micro.54.1.641
Hakyemez IN, Sit M, Aktas G, Tas T, Mengeloglu FZ, Kucukbayrak A (2012) A case of giant hepatic hydatid cyst infected with Morganella morganii and the literature review. Case Rep Gastrointest Med. https://doi.org/10.1155/2012/591561
Hannan TJ, Mysorekar IU, Chen SL, Walker JN, Jones JM, Pinkner JS, Hultgren SJ, Seed PC (2008) LeuX tRNA-dependent and -independent mechanisms of Escherichia coli pathogenesis in acute cystitis. Mol Microbiol 67:116–128. https://doi.org/10.1111/j.1365-2958.2007.06025.x
Harshey RM, Estepa G, Yanagi H (1989) Cloning and nucleotide sequence of a flagellin-coding gene (hag) from Serratia marcescens 274. Gene 79:1–8. https://doi.org/10.1016/0378-1119(89)90087-5
Hochhut B, Wilde C, Balling G, Middendorf B, Dobrindt U, Brzuszkiewicz E, Gottschalk G, Carniel E, Hacker J (2006) Role of pathogenicity island-associated integrases in the genome plasticity of uropathogenic Escherichia coli strain 536. Mol Microbiol 61:584–595. https://doi.org/10.1111/j.1365-2958.2006.05255.x
Ito WE, Maia de Almeida SH (2019) Purple urine-bag syndrome. Int Urogynecol J 30:157–158. https://doi.org/10.1007/s00192-018-3747-7
Iwalokun BA, Olukosi YA, Adejoro A, Olaye JA, Fashade O (2004) Comparative biochemical and molecular evaluation of swarming of Proteus and effects of anti-swarm agents. Afr J Biotechnol 3:99–104. https://doi.org/10.5897/AJB2004.000-2018
Javadi M, Bouzari S, Oloomi M (2017) Horizontal gene transfer and the diversity of Escherichia coli. Escherichia coli – recent advances on physiology, pathogenesis and biotechnological applications. IntechOpen, London, pp 317–331
Joo HS, Otto M (2012) Molecular basis of in vivo biofilm formation by bacterial pathogens. Chem Biol 19:1503–1513. https://doi.org/10.1016/j.chembiol.2012.10.022
Joseph J, Sharma S, Dave VP (2018) Filamentous gram-negative bacteria masquerading as actinomycetes in infectious endophthalmitis: a review of three cases. J Ophthalmic Inflamm Infect. https://doi.org/10.1186/s12348-018-0157-4
Kamp HD, Higgins DE (2011) A protein thermometer controls temperature-dependent transcription of flagellar motility genes in Listeria monocytogenes. PLoS Pathog. https://doi.org/10.1371/journal.ppat.1002153
Kapatral V, Minnich SA (1995) Co-ordinate, temperature-sensitive regulation of the three Yersinia enterocolitica flagellin genes. Mol Microbiol 17:49–56. https://doi.org/10.1111/j.1365-2958.1995.mmi_17010049.x
Kaye D (1968) Antibacterial activity of human urine. J Clin Invest 47:2374–2390. https://doi.org/10.1172/JCI105921
Ko M, Park C (2000) H-NS-dependent regulation of flagellar synthesis is mediated by a LysR family protein. J Bacteriol 182:4670–4672. https://doi.org/10.1128/JB.182.16.4670-4672.2000
Kühn MJ, Schmidt FK, Farthing NE, Rossmann FM, Helm B, Wilson LG, Eckhardt B, Thormann KM (2018) Spatial arrangement of several flagellins within bacterial flagella improves motility in different environments. Nat Commun. https://doi.org/10.1038/s41467-018-07802-w
Kukla R, Chudejova K, Papagiannitsis CC, Medvecky M, Habalova K, Hobzova L, Bolehovska R, Pliskova L, Hrabak J, Zemlickova H (2018) Characterization of KPC-encoding plasmids from Enterobacteriaceae isolated in a Czech Hospital. Antimicrob Agents Chemother. https://doi.org/10.1128/AAC.02152-17
Kuo J (2007) Electron microscopy: methods and protocols, 2nd edn. Humana Press Inc, New Jersey
Lam O, Wheeler J, Tang CM (2014) Thermal control of virulence factors in bacteria: a hot topic. Virulence 5:852–862. https://doi.org/10.4161/21505594.2014.970949
Lebeaux D, Chauhan A, Rendueles O, Beloin C (2013) From in vitro to in vivo models of bacterial biofilm-related infections. Pathogens 2:288–356. https://doi.org/10.3390/pathogens2020288
Leylabadlo HE, Kafil HS, Yousefi M, Aghazadeh M, Asgharzadeh M (2016) Persistent infection with metallo-beta-lactamase and extended spectrum β-lactamase producer Morganella morganii in a patient with urinary tract infection after kidney transplantation. J Nat Sci Biol Med 7:179–181. https://doi.org/10.4103/0976-9668.184707
Lin TY, Chan MC, Yang YS, Lee Y, Yeh KM, Lin JC, Chang FY (2015) Clinical manifestations and prognostic factors of Morganella morganii bacteremia. Eur J Clin Microbiol Infect Dis 34:231–236. https://doi.org/10.1007/s10096-014-2222-8
Liu R, Ochman H (2007) Stepwise formation of the bacterial flagellar system. Proc Natl Acad Sci U S A 104:7116–7121. https://doi.org/10.1073/pnas.0700266104
Lüthje P, Brauner A (2014) Virulence factors of uropathogenic Escherichia coli and their interaction with the host. Adv Microb Physiol 65:337–372. https://doi.org/10.1016/bs.ampbs.2014.08.006
Mahrouki S, Belhadj O, Chihi H, Mohamed BM, Celenza G, Amicosante G, Perilli M (2012) Chromosomal blaCTX-M-15 associated with ISEcp1 in Proteus mirabilis and Morganella morganii isolated at the military hospital of Tunis, Tunisia. J Med Microbiol 61:1286–1289. https://doi.org/10.1099/jmm.0.039487-0
McGann P, Snesrud E, Ong AC, Appalla L, Koren M, Kwak YI, Waterman PE, Lesho EP (2015) War wound treatment complications due to transfer of an IncN plasmid harboring bla(OXA-181) from Morganella morganii to CTX-M-27-producing sequence type 131 Escherichia coli. Antimicrob Agents Chemother 59:3556–3562. https://doi.org/10.1128/AAC.04442-14
Merritt JH, Kadouri DE, O'Toole GA (2005) Growing and analyzing static biofilms. Curr Protoc Microbiol. Chapter 1:Unit 1B.1. https://doi.org/10.1002/9780471729259.mc01b01s00
Minnullina L, Pudova D, Shagimardanova E, Shigapova L, Sharipova M, Mardanova A (2019) Comparative genome analysis of uropathogenic Morganella morganii strains. Front Cell Infect Microbiol. https://doi.org/10.3389/fcimb.2019.00167
Nakao T, Yoshida M, Kanashima H, Yamane T (2013) Morganella morganii pericarditis in a patient with multiple myeloma. Case Rep Hematol. https://doi.org/10.1155/2013/452730
Nedwell DB (1999) Effect of low temperature on microbial growth: lowered affinity for substrates limits growth at low temperature. FEMS Microbiol Ecol 30:101–111. https://doi.org/10.1111/j.1574-6941.1999.tb00639.x
Noor R, Islam Z, Kishore Munshi S, Rahman F (2013) Influence of temperature on Escherichia coli growth in different culture media. J Pure Appl Microbiol 7:899–904
O’Hara CM, Brenner FW, Miller JM (2000) Classification, identification, and clinical significance of Proteus, Providencia, and Morganella. Clin Microbiol Rev 13:534–546. https://doi.org/10.1128/cmr.13.4.534-546.2000
Olaitan AO, Diene SM, Gupta SK, Adler A, Assous MV, Rolain JM (2014) Genome analysis of NDM-1 producing Morganella morganii clinical isolate. Expert Rev Anti Infect Ther 12:1297–1305. https://doi.org/10.1586/14787210.2014.944504
Omoyibo EE, Oladele AO, Ibrahim MH, Adekunle OT (2018) Antibiotic susceptibility of wound swab isolates in a tertiary hospital in Southwest Nigeria. Ann Afr Med 17:110–116. https://doi.org/10.4103/aam.aam_22_17
Ott M, Messner P, Heesemann J, Marre R, Hacker J (1991) Temperature-dependent expression of flagella in Legionella. J Gen Microbiol 137:1955–1961. https://doi.org/10.1099/00221287-137-8-1955
Patil AB, Nadagir SD, Lakshminarayana S, Syeda FM (2012) Morganella morganii, subspecies morganii, biogroup A: an unusual causative pathogen of brain abscess. J Neurosci Rural Pract 3:370–372. https://doi.org/10.4103/0976-3147.102631
Peel M, Donachie W, Shaw A (1988) Temperature-dependent expression of flagella of Listeria monocytogenes studied by electron microscopy, SDS-PAGE and western blotting. J Gen Microbiol 134:2171–2178. https://doi.org/10.1099/00221287-134-8-2171
Ponte A, Costa C (2015) Multiple liver abscesses due to Morganella morganii. Acta Med Port. 28:539
Prieto Amorin J, Lopez M, Rando K, Castelli J, Medina Presentado J (2018) Early bacterial pneumonia after hepatic transplantation: epidemiologic profile. Transplant Proc 50:503–508. https://doi.org/10.1016/j.transproceed.2017.11.047
Ratiner YA (1999) Temperature-dependent flagellar antigen phase variation in Escherichia coli. Res Microbiol 150:457–463. https://doi.org/10.1016/S0923-2508(99)00111-4
Reitzer L, Zimmern P (2019) Rapid growth and metabolism of uropathogenic Escherichia coli in relation to urine composition. Clin Microbiol Rev. https://doi.org/10.1128/CMR.00101-19
Rozales FP, Ribeiro VB, Magagnin CM, Pagano M, Lutz L, Falci DR, Machado A, Barth AL, Zavascki AP (2014) Emergence of NDM-1-producing Enterobacteriaceae in Porto Alegre, Brazil. Int J Infect Dis 25:79–81. https://doi.org/10.1016/j.ijid.2014.01.005
Rudenko I, Ni B, Glatter T, Sourjik V (2019) Inefficient secretion of anti-sigma factor FlgM inhibits bacterial motility at high temperature. iScience 16:145–154. https://doi.org/10.1016/j.isci.2019.05.022
Rütschlin S, Böttcher T (2020) Inhibitors of bacterial swarming behavior. Chemistry 26:964–979. https://doi.org/10.1002/chem.201901961
Sanawar H, Pinel I, Farhat NM, Bucs SS, Zlopasa J, Kruithof JC, Witkamp GJ, van Loosdrecht MCM, Vrouwenvelder JS (2018) Enhanced biofilm solubilization by urea in reverse osmosis membrane systems. Water Res X. https://doi.org/10.1016/j.wroa.2018.10.001
Sarigul N, Korkmaz F, Kurultak İ (2019) A new artificial urine protocol to better imitate human urine. Sci Rep. https://doi.org/10.1038/s41598-019-56693-4
Schmidt H, Hensel M (2004) Pathogenicity islands in bacterial pathogenesis. Clin Microbiol Rev 17:14–56. https://doi.org/10.1128/CMR.17.1.14-56.2004
Senior BW, Hughes C (1988) Production and properties of haemolysins from clinical isolates of the Proteeae. J Med Microbiol 25:17–25. https://doi.org/10.1099/00222615-25-1-17
Sica S, Di Mario A, Salutari P, d’Onofrio G, Antinori A, Chiusolo P, Leone G (1995) Morganella morganii pericarditis after resolvent splenectomy for immune pancytopenia following allogeneic bone marrow transplantation for acute lymphoblastic leukemia. Clin Infect Dis 21:1052–1053. https://doi.org/10.1093/clinids/21.4.1052
Simms AN, Mobley HL (2008) Multiple genes repress motility in uropathogenic Escherichia coli constitutively expressing type 1 fimbriae. J Bacteriol 190:3747–3756. https://doi.org/10.1128/JB.01870-07
Singh P, Mosci R, Rudrik JT, Manning SD (2015) Draft genome sequence of a diarrheagenic Morganella morganii isolate. Genome Announc. https://doi.org/10.1128/genomeA.01165-15
Soutourina OA, Bertin PN (2003) Regulation cascade of flagellar expression in Gram-negative bacteria. FEMS Microbiol Rev 27:505–523. https://doi.org/10.1016/S0168-6445(03)00064-0
Soutourina O, Kolb A, Krin E, Laurent-Winter C, Rimsky S, Danchin A, Bertin P (1999) Multiple control of flagellum biosynthesis in Escherichia coli: role of H-NS protein and the cyclic AMP-catabolite activator protein complex in transcription of the flhDC master operon. J Bacteriol 181:7500–7508. https://doi.org/10.1128/JB.181.24.7500-7508.1999
Soutourina OA, Krin E, Laurent-Winter C, Hommais F, Danchin A, Bertin PN (2002) Regulation of bacterial motility in response to low pH in Escherichia coli: the role of H-NS protein. Microbiology (reading) 148:1543–1551. https://doi.org/10.1099/00221287-148-5-1543
Stickler DJ (2014) Clinical complications of urinary catheters caused by crystalline biofilms: something needs to be done. J Intern Med 276:120–129. https://doi.org/10.1111/joim.12220
Sullivan MJ, Petty NK, Beatson SA (2011) Easyfig: a genome comparison visualizer. Bioinformatics 27:1009–1010. https://doi.org/10.1093/bioinformatics/btr039
Szabó O, Gulyás D, Szabó N, Kristóf K, Kocsis B, Szabó D (2018) Plasmid-mediated quinolone resistance determinants in Enterobacteriaceae from urine clinical samples. Acta Microbiol Immunol Hung 65:255–265. https://doi.org/10.1556/030.65.2018.012
Tan PSC, Badiei A, Fitzgerald DB, Kuok YJ, Lee YCG (2019) Pleural empyema in a patient with a perinephric abscess and diaphragmatic defect. Respirol Case Rep. https://doi.org/10.1002/rcr2.400
Tanabe Y, Wada T, Ono K, Abo T, Kutsukake K (2011) The transcript from the σ(28)-dependent promoter is translationally inert in the expression of the σ(28)-encoding gene fliA in the fliAZ operon of Salmonella enterica serovar Typhimurium. J Bacteriol 193:6132–6141. https://doi.org/10.1128/JB.05909-11
Trunk T, Khalil HS, Leo JC (2018) Bacterial Autoaggregation AIMS Microbiol 4:140–164. https://doi.org/10.3934/microbiol.2018.1.140
Tsai MT, Yeh JT, Yang WC, Wu TH (2013) CAPD-related peritonitis caused by Morganella morganii. Perit Dial Int 33:104–105. https://doi.org/10.3747/pdi.2012.00035
Tsuji A, Kaneko Y, Takahashi K, Ogawa M, Goto S (1982) The effects of temperature and pH on the growth of eight enteric and nine glucose non-fermenting species of gram-negative rods. Microbiol Immunol 26:15–24. https://doi.org/10.1111/j.1348-0421.1982.tb00149.x
Ueda S (2017) The effects of temperature on the growth and heat resistance of Cronobacter spp. Biocontrol Sci 22:125–129. https://doi.org/10.4265/bio.22.125
Van Asten FJ, Gaastra W (1999) Urea restrains swarming of Proteus mirabilis. J Clin Microbiol 37:1652. https://doi.org/10.1128/JCM.37.5.1652-1652.1999
van Ditmarsch D, Boyle KE, Sakhtah H, Oyler JE, Nadell CD, Déziel É, Dietrich LE, Xavier JB (2013) Convergent evolution of hyperswarming leads to impaired biofilm formation in pathogenic bacteria. Cell Rep 4:697–708. https://doi.org/10.1016/j.celrep.2013.07.026
Wiebe WJ, Sheldon WM, Pomeroy LR (1992) Bacterial growth in the cold: evidence for an enhanced substrate requirement. Appl Environ Microbiol 58:359–364. https://doi.org/10.1128/aem.58.1.359-364.1992
Wiles TJ, Kulesus RR, Mulvey MA (2008) Origins and virulence mechanisms of uropathogenic Escherichia coli. Exp Mol Pathol 85:11–19. https://doi.org/10.1016/j.yexmp.2008.03.007
Withman B, Gunasekera TS, Beesetty P, Agans R, Paliy O (2013) Transcriptional responses of uropathogenic Escherichia coli to increased environmental osmolality caused by salt or urea. Infect Immun 81:80–89. https://doi.org/10.1128/IAI.01049-12
Yazdi S, Ardekani AM (2012) Bacterial aggregation and biofilm formation in a vortical flow. Biomicrofluidics DOI 10(1063/1):4771407
Funding
This paper was supported by the Kazan Federal University Strategic Academic Leadership Program.
Author information
Authors and Affiliations
Contributions
Not applicable.
Corresponding author
Ethics declarations
Ethics approval
Ethical approval was not required since clinical isolates were collected and stored as part of routine clinical care.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Minnullina, L., Kostennikova, Z., Evtugin, V. et al. Diversity in the swimming motility and flagellar regulon structure of uropathogenic Morganella morganii strains. Int Microbiol 25, 111–122 (2022). https://doi.org/10.1007/s10123-021-00197-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-021-00197-7