Abstract
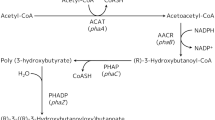
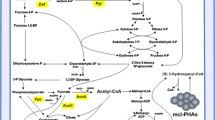
Polyhydroxyalkanoates (PHAs) are biodegradable bioplastics that are synthesized by diverse bacteria. In this study, the synthesis of PHAs by the model aromatic-degrading strain Burkholderia xenovorans LB400 was analyzed. Twelve pha genes including three copies of phaC and five copies of the phasin-coding phaP genes are distributed among the three LB400 replicons. The phaC1ABR gene cluster that encodes the enzymes of the PHA anabolic pathway is located at chromosome 1 of strain LB400. During the growth of strain LB400 on glucose under nitrogen limitation, the expression of the phaC1, phaA, phaP1, phaR, and phaZ genes was induced. Under nitrogen limitation, PHA accumulation in LB400 cells was observed by fluorescence microscopy after Nile Red staining. GC-MS analyses revealed that the PHA accumulated under nitrogen limitation was poly(3-hydroxybutyrate) (PHB). LB400 cells grown on glucose as the sole carbon source under nitrogen limitation accumulated 40 ± 0.96% PHB of the cell dry weight, whereas no PHA was observed in cells grown in control medium. The functionality of the phaC1 gene from strain LB400 was further studied using heterologous expression in a Pseudomonas putida KT40C1ZC2 mutant strain derived from P. putida KT2440 that is unable to synthesize PHAs. Interestingly, KT40C1ZC2[pVNC1] cells that express the phaC1 gene from strain LB400 were able to synthesize PHB (33.5% dry weight). This study indicates that B. xenovorans LB400 possesses a functional PHA synthetic pathway that is encoded by the pha genes and is capable of synthesizing PHB.






Similar content being viewed by others
References
Acevedo F, Villegas P, Urtuvia V, Hermosilla J, Navia R, Seeger M (2018) Bacterial polyhydroxybutyrate for electrospun fiber production. Int J Biol Macromol 106:692–697
Agulló L, Romero-Silva MJ, Domenech M, Seeger M (2017) p-Cymene promotes its catabolism through the p-cymene and the p-cumate pathways, activates a stress response and reduces the biofilm formation in Burkholderia xenovorans LB400. PLoS ONE 12:e0169544
Aldor LS, Keasling JD (2003) Process design for microbial plastic factories: metabolic engineering of polyhydroxyalkanoates. Curr Opin Biotechnol 14:475−483
Anderson A, Dawes EA (1990) Occurrence, metabolism, metabolic role and industrial uses of bacterial polyhydroxyalkanoates. Microbiol Rev 54:450–472
Ayub ND, Pettinari MJ, Méndez BS, López NI (2006) Impaired polyhydroxybutyrate biosynthesis from glucose in Pseudomonas sp. 14−3 is due to a defective β−ketothiolase gene. FEMS Microbiol Lett 264:125−131
Cai L, Yuan MQ, Liu F, Jian J, Chen GQ (2009) Enhanced production of medium-chain-length polyhydroxyalkanoates (PHA) by PHA depolymerase knockout mutant of Pseudomonas putida KT2442. Bioresour Technol 100:2265–2270
Chain PSG, Denef VJ, Konstantinidis KT, Vergez LM, Agulló L, Reyes VL, Hauser L, Córdova M, Gómez L, González M, Land M, Lao V, Larimer F, LiPuma JJ, Mahenthiralingam E, Malfatti SA, Marx CJ, Parnell JJ, Ramette A, Richardson P, Seeger M, Smith D, Spilker T, Sul WJ, Tsoi TV, Ulrich LE, Zhulin IB, Tiedje JM (2006) Burkholderia xenovorans LB400 harbors a multi−replicon 9.73−Mbp genome shaped for versatility. Proc Natl Acad Sci 103:15280–15287
Chirino B, Strahsburger E, Agulló L, González M, Seeger M (2013) Genomic and functional analyses of the 2−aminophenol catabolic pathway and partial conversion of its substrate into picolinic acid in Burkholderia xenovorans LB400. PLoS ONE 8:e75746
De Eugenio LI, Escapa IF, Morales V, Dinjaski N, Galán B, García JL, Prieto MA (2010) The turnover of medium-chain-length polyhydroxyalkanoates in Pseudomonas putida KT2442 and the fundamental role of PhaZ depolymerase for the metabolic balance. Environ Microbiol 12:207–221
Díaz-Barrera A, Andler R, Martínes I, Peña C (2016) Poly-3-hydroxybutyrate production by Azotobacter vinelandii strains in batch cultures at different oxygen transfer rates. J Chem Technol Biotechnol 91:1063–1071
Eggers J, Steinbüchel A (2013) Poly(3-hydroxybutyrate) degradation in Ralstonia eutropha H16 is mediated stereoselectively to (S)-3-hydroxybutyryl coenzyme A (CoA) via crotonyl-CoA. J Bacteriol 195:3213–3223
Encarnación S, Vargas M, Dunn MF, Dávalos A, Mendoza G, Mora Y, Mora J (2002) AniA regulates reserve polymer accumulation and global protein expression in Rhizobium etli. J Bacteriol 184:2287–2295
Hyakutake M, Saito Y, Tomizawa S, Mizuno K, Tsuge T (2011) Polyhydroxyalkanoate (PHA) synthesis by class IV PHA synthases employing Ralstonia eutropha PHB-4 as host strain. Biosci Biotechnol Biochem 75:1615–1617
Livak K, Schmittgen T (2001) Analysis of relative gene expression data using real−time quantitative PCR and 2-ΔΔCt method. Methods 25:402−408
Lopes M, Gosset G, Rocha R, Gomez J, Ferreira da Silva L (2011) PHB biosynthesis in catabolite repression mutant of Burkholderia sacchari. Curr Microbiol 63:319–326
Madison LL, Huisman GW (1999) Metabolic engineering of poly (3-hydroxyalkanoates): from DNA to plastic. Microbiol Mol Biol Rev 63:21–53
Maehara A, Taguchi S, Nishiyama T, Yamane T, Doi Y (2002) A repressor protein, PhaR, regulates polyhydroxyalkanoate (PHA) synthesis via its direct interaction with PHA. J Bacteriol 184:3992–4002
Méndez V, Agulló L, González M, Seeger M (2011) The homogentisate and homoprotocatechuate central pathways are involved in 3− and 4−hydroxyphenylacetate degradation by Burkholderia xenovorans LB400. PLoS ONE 6:e17583
Mezzina MP, Pettinari MJ (2016) Phasins, multifaceted polyhydroxyalkanoate granule-associated proteins. Appl Environ Microbiol 82:5060–5067
Mozejko J, Ciesielski S (2013) Saponified waste palm oil as an attractive renewable resource for mcl-polyhydroxyalkanoate synthesis. J Biosci Bioeng 116:485−492
Nikodinovic-Runic J, Guzik M, Kenny ST, Babu R, Werker A, O’Connor KE (2013) Carbon-rich wastes as feedstocks for biodegradable polymer (polyhydroxyalkanoate) production using bacteria. Adv Appl Microbiol 84:139–200
Overwin H, Standfuß-Gabisch C, González M, Méndez V, Seeger M, Reichelt J, Wray V, Hofer B (2015) Permissivity of the biphenyl-specific aerobic bacterial metabolic pathway towards analogues with various steric requirements. Microbiology 161(9):1844–1856
Peoples OP, Sinskey AJ (1989) Poly−β−hydroxybutirate (PHB) biosynthesis in Alcaligenes eutrophus H16. J Biol Chem 264:15298–15303
Pfeiffer D, Jendrossek D (2012) Localization of poly (3-hydroxybutyrate)(PHB) granule-associated proteins during PHB granule formation and identification of two new phasins, PhaP6 and PhaP7, in Ralstonia eutropha H16. J Bacteriol 194:5909–5921
Poltronieri P, Kumar P (2017) Polyhydroxyalkanoates (PHAs) in Industrial Applications. In: Torres Martínez L., Kharissova O., Kharisov B. (Eds) Handbook of Ecomaterials. Springer, Cham, pp 1–30. https://doi.org/10.1007/978-3-319-48281-1_70-2
Pötter M, Steinbüchel A (2005) Poly (3-hydroxybutyrate) granule-associated proteins: impacts on poly (3-hydroxybutyrate) synthesis and degradation. Biomacromolecules 6:552–560
Pötter M, Madkour MH, Mayer F, Steinbüchel A (2002) Regulation of phasin expression and polyhydroxyalkanoate (PHA) granule formation in Ralstonia eutropha H16. Microbiology 148:2413− 2426
Pötter M, Müller H, Reinecke F, Wieczorek R, Fricke F, Bowien B, Friedrich B, Steinbüchel A (2004) The complex structure of polyhydroxybutyrate (PHB) granules: four orthologous and paralogous phasins occur in Ralstonia eutropha. Microbiology 150:2301–2311
Pötter M, Müller H, Steinbüchel A (2005) Influence of homologous phasins (PhaP) on PHA accumulation and regulation of their expression by the transcriptional repressor PhaR in Ralstonia eutropha H16. Microbiology 151:825–833
Prieto MA, Bühler B, Jung K, Witholt B, Kessler B (1999) PhaF, a polyhydroxyalkanoate-granule-associated protein of Pseudomonas oleovorans GPo1 involved in the regulatory expression system for pha genes. J Bacteriol 181:858–868
Ramsay JA, Hassan MCA, Ramsay BA (1995) Hemicellulose as a potential substrate for production of poly (β-hydroxyalkanoates). Can J Microbiol 41:262–266
Reddy CSK, Ghai R, Kalia V (2003) Polyhydroxyalkanoates: an overview. Bioresour Technol 87:137–146
Rehm BHA (2003) Polyester synthases: natural catalysts for plastics. Biochem J 376:15–33
Rehm BHA (2007) Biogenesis of microbial polyhydroxyalkanoate granules: a platform technology for the production of tailor−made bioparticles. Curr Issues Mol Biol 9:41–62
Rehm CSK, Steinbüchel A (1999) Biochemical and genetic analysis of PHA synthases and other proteins required for PHA synthesis. Int J Biol Macromol 25:3–19
Riis V, Mai W (1988) Gas chromatographic determination of poly−β−hydroxybutyric acid in microbial biomass after hydrochloric acid propanolysis. J Chromatogr 445:285–289
Romero−Silva MJ, Méndez V, Agulló L, Seeger M (2013) Genomic and functional analyses of the gentisate and protocatechuate ring−cleavage pathways and related 3−hydroxybenzoate and 4−hydroxybenzoate peripheral pathways in Burkholderia xenovorans LB400. PLoS ONE 8:e56038
Roy I, Visakh PM (2014) Polyhydroxyalkanoate (PHA) based blends, composites and nanocomposites. The Royal Society of Chemistry, Cambridge. https://doi.org/10.1039/9781782622314
Sabbagh F, Muhamad II (2017) Production of poly-hydroxyalkanoate as secondary metabolite with main focus on sustainable energy. Renew Sust Energ Rev 72:95–104
Savenkova L, Gercberga Z, Nikolaeva V, Dzene A, Bibers I, Kalnin M (2000) Mechanical properties and biodegradation characteristics of PHB-based films. Process Biochem 35:573–579
Sawana A, Adeolu M, Gupta RS (2014) Molecular signatures and phylogenomic analysis of the genus Burkholderia: proposal for division of this genus into the emended genus Burkholderia containing pathogenic organisms and a new genus Paraburkholderia gen. nov. harboring environmental species. Front Genet 5:429
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108
Schwartz E, Henne A, Cramm R, Eitinger T, Friedrich B, Gottschalk G (2003) Complete nucleotide sequence of pHG1: a Ralstonia eutropha H16 megaplasmid encoding key enzymes of H 2-based lithoautotrophy and anaerobiosis. J Mol Biol 332:369–383
Schwartz E, Voigt B, Zühlke D, Pohlmann A, Lenz O, Albrecht D, Schwarze A, Kohlmann Y, Krause C, Heker M, Friedrich B (2009) A proteomic view of the facultatively chemolithoautotrophic lifestyle of Rasltonia eutropha H16. Proteomics 9:5132−5142
Seeger M, Cámara B, Hofer B (2001) Dehalogenation, denitration, dehydroxylation, and angular attack on substituted biphenyls and related compounds by a biphenyl dioxygenase. J Bacteriol 183:3548−3555
Silva-Rocha R, Martínez-García E, Calles B, Chavarría M, Arce-Rodríguez A, de Las Heras A, Paéz-Espina D, Durante-Rodriguez G, Kim J, Nikel P I, Platero R, de Lorenzo V (2012) The Standard European Vector Architecture (SEVA): a coherent platform for the analysis and deployment of complex prokaryotic phenotypes. Nucleic Acids Res, 41(D1):D666−D675
Steinbüchel A (2001) Perspectives for biotechnological production and utilization of biopolymers: metabolic engineering of polyhydroxyalkanoate biosynthesis pathways as a successful example. Macromol Biosci 1(1):1–24
Steinbüchel A, Lütke-Eversloh T (2003) Metabolic engineering and pathway construction for biotechnological production of relevant polyhydroxyalkanoates in microorganisms. Biochem Eng J 16:81−96
Urtuvia V, Villegas P, González M, Seeger M (2014) Bacterial production of the biodegradable plastics polyhydroxyalkanoates. Int J Biol Macromol 70:208−213
Ushimaru K, Motoda Y, Numata K, Tsuge T (2014) Phasin proteins activate Aeromonas caviae polyhydroxyalkanoate (PHA) synthase but not Ralstonia eutropha PHA synthase. Appl Environ Microbiol 80:2867–2873
Valappil SP, Boccaccini AR, Bucke C, Roy I (2007) Polyhydroxyalkanoates in Gram-positive bacteria: insights from the genera Bacillus and Streptomyces. Antonie Van Leeuwenhoek 91:1–17
Wahl A, Schuth N, Pfeiffer D, Nussberger S, Jendrossek D (2012) PHB granules are attached to the nucleoid via PhaM in Ralstonia eutropha. BMC Microbiol 12:262–273
Ward PG, O’Connor KE (2005) Bacterial synthesis of polyhydroxyalkanoates containing aromatic and aliphatic monomers by Pseudomonas putida CA-3. Int J Biol Macromol 35:127−133
Yagi K, Miyawaki I, Kayashita A, Kondo M, Kitano Y, Murakami Y, Mizoguchi T (1996) Biosynthesis of poly (3-hydroxyalkanoic acid) copolymer from CO (inf2) in Pseudomonas acidophila through introduction of the DNA fragment responsible for chemolithoautotrophic growth of Alcaligenes hydrogenophilus. Appl Environ Microbiol 62:1004–1007
Zou H, Shi M, Zhang T, Li L, Li L, Xian M (2017) Natural and engineered polyhydroxyalkanoate (PHA) synthase: key enzyme in biopolyester production. Appl Microbiol Biotechnol 101:7417–7426
Acknowledgments
The authors thank Maria Auxiliadora Prieto for the generous support in heterologous expression assays and Vincent Collins for critical reading of the manuscript.
Funding
The present work was supported by PhD scholarship CONICYT 21100268 (VU), Programa de Investigación Asociativa (PIA) Anillo ACT172128 GAMBIO (MS), FONDECYT 1151174 and 1110992 (MS), USM 131342 and 131562 (MS), USM Pie>A (PV), USM CN&SB (MS), and CYTED-PRIBOP grants (MS).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Urtuvia, V., Villegas, P., Fuentes, S. et al. Burkholderia xenovorans LB400 possesses a functional polyhydroxyalkanoate anabolic pathway encoded by the pha genes and synthesizes poly(3-hydroxybutyrate) under nitrogen-limiting conditions. Int Microbiol 21, 47–57 (2018). https://doi.org/10.1007/s10123-018-0004-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-018-0004-3