Abstract
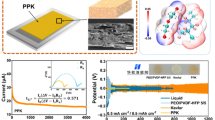
Lithium-ion batteries (LIBs) benefit from an effective electrolyte system design in both terms of their safety and energy storage capability. Herein, a series of precursor membranes with high porosity were produced using electrospinning technology by mixing PVDF and triblock copolymer (PS-PEO-PS), resulting in a porous structure with good interconnections, which facilitates the absorbency of a large amount of electrolyte and further increases the ionic conductivity of gel polymer electrolytes (GPEs). It has been demonstrated that post-cross-linking of the precursor membranes increases the rigidity of the nanofibers, which allows the polymer film to be dimensionally stable up to 260 °C while maintaining superior electrochemical properties. The obtained cross-linked GPEs (CGPEs) showed high ionic conductivity up to 4.53×10−3 S·cm−1. With the CGPE-25, the assembled Li/LiFePO4 half cells exhibited good rate capability and maintained a capacity of 99.4% and a coulombic efficiency of 99.3% at 0.1 C. These results suggest that the combination of electrospinning technique and post-cross-linking is an effective method to construct polymer electrolytes with high thermal stability and steadily decent electrochemical performance, particularly useful for Lithium-ion battery applications that require high-temperature usage.
Similar content being viewed by others
Data Availability Statement
The related data (DOI: https://doi.org/10.57760/sciencedb.j00189.00003) for this paper is available in the Data Repository of China Association for Science and Technology (https://www.scidb.cn/c/cjps).
References
Zhang, J. G.; Xu, W.; Xiao, J.; Cao, X.; Liu, J. Lithium metal anodes with nonaqueous electrolytes. Chem. Rev. 2020, 120, 13312–13348.
Zhang, X.; Xu, C.; Yosef, N. Simulating multiple faceted variability in single cell RNA sequencing. Nat. Commun. 2019, 10, 1–16.
Zhang, Y.; Sun, J.; Liu, W.; Niu, Z.; Yan, Y.; Qiao, L.; Wu, N. A low-surface-tension strategy to construct ultrathin reduced graphene oxide as lithiophilic matrix for lithium metal anode. Adv. Mater. Interfaces 2022, 9, 2200752.
Chombo, P. V.; Laoonual, Y. A review of safety strategies of a Li-ion battery. J. Power Sources 2020, 478, 228649.
Feng, X.; Ouyang, M.; Liu, X.; Lu, L.; Xia, Y.; He, X. Thermal runaway mechanism of lithium ion battery for electric vehicles: a review. Energy Storage Mater. 2018, 10, 246–267.
Yuan, S.; Kong, T.; Zhang, Y.; Dong, P.; Zhang, Y.; Dong, X.; Wang, Y.; Xia, Y. Advanced electrolyte design for high-energy-density Li-metal batteries under practical conditions. Angew. Chem. Int. Ed. 2021, 133, 25828–25842.
Zampardi, G.; La Mantia, F. Solid-electrolyte interphase at positive electrodes in high-energy Li-ion batteries: current understanding and analytical tools. Batteries Supercaps 2020, 3, 672–697.
Zhang, S. S. A review on the separators of liquid electrolyte Li-ion batteries. J. Power Sources 2007, 164, 351–364.
Liu, F.; Bin, F.; Xue, J.; Wang, L.; Yang, Y.; Huo, H.; Zhou, J.; Li, L. Polymer electrolyte membrane with high ionic conductivity and enhanced interfacial stability for lithium metal battery. ACS Appl. Mater. Interfaces 2020, 12, 22710–22720.
Pei, X.; Li, Y.; Ou, T.; Liang, X.; Yang, Y.; Jia, E.; Tan, Y.; Guo, S. Li-N interaction induced deep eutectic gel polymer electrolyte for high performance lithium-metal batteries. Angew. Chem. Int. Ed. 2022, 61, e202205075.
Wang, Q.; Xu, X.; Hong, B.; Bai, M.; Li, J.; Zhang, Z.; Lai, Y. Molecular engineering of a gel polymer electrolyte via in-situ polymerization for high performance lithium metal batteries. Chem. Eng. J. 2022, 428, 131331.
Huo, S.; He, Y.; Hu, Z.; Bao, W.; Chen, W.; Wang, Y.; Zeng, D.; Cheng, H.; Zhang, Y. New insights into designation of single-ion conducting gel polymer electrolyte for high-performance lithium metal batteries. J. Membr. Sci. 2022, 647, 120287.
Wang, J.; Wang, Z.; Ni, J.; Li, L. Electrospun materials for batteries moving beyond lithium-ion technologies. Electrochem. Energy Rev. 2022, 5, 211–241.
Bicy, K.; Gueye, A. B.; Rouxel, D.; Kalarikkal, N.; Thomas, S. Lithiumion battery separators based on electrospun PVDF: a review. Surf. Interfaces 2022, 31, 101977.
Song, X. Y.; Wang, Z.; Zhao, F. Z.; Sun, Y. Z.; Cheng, B. W.; Xing, J. F. A separator with a novel thermal crosslinking structure based on electrospun PI/A-POSS for lithium-ion battery with high safety and outstanding electrochemical performance. Adv. Mater. Interfaces 2021, 8, 2100458.
Tang, W.; Liu, Q. Q.; Luo, N.; Chen, F.; Fu, Q. High safety and electrochemical performance electrospun para-aramid nanofiber composite separator for lithium-ion battery. Compos. Sci. Technol. 2022, 225, 109479.
Lai, W.; Shan, L.; Bai, J.; Xiao, L.; Liu, L.; Wang, S.; Gong, L.; Jiao, Y.; Xie, W.; Liu, H. Highly permeable and acid-resistant nanofiltration membrane fabricated by in-situ interlaced stacking of COF and polysulfonamide films. Chem. Eng. J. 2022, 450, 137965.
Bandyopadhyay, S.; Gupta, A.; Srivastava, R.; Nandan, B. Bio-inspired design of electrospun poly(acrylonitrile) and novel ionene based nanofibrous mats as highly flexible solid state polymer electrolyte for lithium batteries. Chem. Eng. J. 2022, 440, 135926.
Seidl, L.; Grissa, R.; Zhang, L.; Trabesinger, S.; Battaglia, C. Unraveling the voltage-dependent oxidation mechanisms of poly(ethylene oxide)-based solid electrolytes for solid-state batteries. Adv. Mater. Interfaces 2022, 9, 2100704.
Sharon, D.; Deng, C.; Bennington, P.; Webb, M. A.; Patel, S. N.; de Pablo, J. J.; Nealey, P. F. Critical percolation threshold for solvation-site connectivity in polymer electrolyte mixtures. Macromolecules 2022, 55, 7212–7221.
Ma, X.; Zuo, X.; Wu, J.; Deng, X.; Xiao, X.; Liu, J. S.; Nan, J. M. Polyethylene-supported ultra-thin polyvinylidene fluoride/hydroxyethyl cellulose blended polymer electrolyte for 5 V high voltage lithium ion batteries. J. Mater. Chem. A 2018, 6, 1496–1503.
Silva, F. C. A.; Ortega, P. F. R.; dos Reis, R. A.; Lavall, R. L.; Costa, L. T. Polymer-ion interactions in PVDF@ionic liquid polymer electrolytes: a combined experimental and computational study. Electrochim. Acta 2022, 427, 140831.
Chiu, L. L.; Chung, S. H. Composite gel-polymer electrolyte for high-loading polysulfide cathodes. J. Mater. Chem. A 2022, 10, 13719–13726.
Yi, L. G.; Zou, C. F.; Chen, X. Y.; Liu, J. L.; Cao, S.; Tao, X. Y.; Zang, Z. H.; Liu, L.; Chang, B. B.; Shen, Y. Q.; Wang, X. Y. One-step synthesis of PVDF-HFP/PMMA-ZrO2 gel polymer electrolyte to boost the performance of a lithium metal battery. ACS Appl. Energy Mater. 2022, 5, 7317–7327.
Shen, Z.; Zhong, J.; Jiang, S.; Xie, W.; Zhan, S.; Lin, K.; Zeng, L.; Hu, H.; Lin, G.; Lin, Y.; Sun, S.; Shi, Z. Polyacrylonitrile porous membrane-based gel polymer electrolyte by in situ free-radical polymerization for stable Li metal batteries. ACS Appl. Mater. Interfaces 2022, 14, 41022–41036.
He, C. F.; Liu, J. Q.; Li, J.; Zhu, F. F.; Zhao, H. J. Blending based polyacrylonitrile/poly(vinyl alcohol) membrane for rechargeable lithium ion batteries. J. Membr. Sci. 2018, 560, 30–37.
Long, L.; Wang, S.; Xiao, M.; Meng, Y. Polymer electrolytes for lithium polymer batteries. J. Mater. Chem. A 2016, 4, 10038–10069.
Xiao, W.; Wang, Z.; Zhang, Y.; Fang, R.; Yuan, Z.; Miao, C.; Yan, X.; Jiang, Y. Enhanced performance of P(VDF-HFP)-based composite polymer electrolytes doped with organic-inorganic hybrid particles PMMA-ZrO2 for lithium ion batteries. J. Power Sources 2018, 382, 128–134.
Nakajima, H.; Ohno, H. Preparation of thermally stable polymer electrolytes from imidazolium-type ionic liquid derivatives. Polym. 2005, 46, 11499–11504.
Xu, D.; Wang, B.; Wang, Q.; Gu, S.; Li, W.; Jin, J.; Chen, C.; Wen, Z. High-strength internal cross-linking bacterial cellulose-network-based gel polymer electrolyte for dendrite-suppressing and high-rate lithium batteries. ACS Appl. Mater. Interfaces 2018, 10, 17809–17819.
Lu, Q.; He, Y.; Yu, Q.; Li, B.; Kaneti, Y. V.; Yao, Y.; Kang, F.; Yang, Q. Dendrite-free, high-rate, long-life lithium metal batteries with a 3D cross-linked network polymer electrolyte. Adv. Mater. 2017, 29, 1604460.
Hsu, C.; Liu, R.; Hsu, C.; Kuo, P. High thermal and electrochemical stability of PVDF-graft-PAN copolymer hybrid PEO membrane for safety reinforced lithium-ion battery. RSC Adv. 2016, 6, 18082–18088.
Tang, Z. H.; Liu, D.; Lyu, X. L.; Liu, Y. D.; Liu, Y.; Yang, W. L.; Shen, Z. H.; Fan, X. H. Ultra-stretchable ion gels based on physically cross-linked polymer networks. J. Mater. Chem. C 2022, 10, 10926–10934.
Tan, L.; Tan, B. Hypercrosslinked porous polymer materials: design, synthesis, and applications. Chem. Soc. Rev. 2017, 46, 3322–3356.
Luo, Y.; Li, B.; Wang, W.; Wu, K.; Tan, B. Hypercrosslinked aromatic heterocyclic microporous polymers: a new class of highly selective CO2 capturing materials. Adv. Mater. 2012, 24, 5703–5707.
Yang, Y.; Tan, B.; Wood, C. D. Solution-processable hypercrosslinked polymers by low cost strategies: a promising platform for gas storage and separation. J. Mater. Chem. A 2016, 4, 15072–15080.
Zhou, B. H.; Yang, M. L.; Zuo, C.; Chen, G.; He, D.; Zhou, X. P.; Liu, C. M.; Xie, X. L.; Xue, Z. G. Flexible, self-healing, and fire-resistant polymer electrolytes fabricated via photopolymerization for all-solid-state lithium metal batteries. ACS Macro Lett. 2020, 9, 525–532.
Xiao, Q.; Deng, C.; Wang, Q.; Zhang, Q.; Yue, Y.; Ren, S. In situ cross-linked gel polymer electrolyte membranes with excellent thermal stability for lithium ion batteries. ACS Omega 2019, 4, 95–103.
Wang, S.; Zhou, L.; Tufail, M. K.; Yang, L.; Zhai, P.; Chen, R.; Yang, W. In-situ synthesized non-flammable gel polymer electrolyte enable highly safe and dendrite-free lithium metal batteries. Chem. Eng. J. 2021, 415, 128846.
Naderi, R.; Gurung, A.; Zhou, Z.; Varnekar, G.; Chen, K.; Zai, J.; Qian, X.; Qiao, Q. Activation of passive nanofillers in composite polymer electrolyte for higher performance lithium-ion batteries. Adv. Sustainable Syst. 2017, 1, 1700043.
Gong, C.; Liu, H.; Zhang, B.; Wang, G.; Cheng, F.; Zheng, G.; Wen, S.; Xue, Z.; Xie, X. High level of solid superacid coated poly(vinylidene fluoride) electrospun nanofiber composite polymer electrolyte membranes. J. Membr. Sci. 2017, 535, 113–121.
Gao, T. N.; Wang, T.; Wu, W.; Liu, Y.; Huo, Q.; Qiao, Z. A.; Dai, S. Solvent-induced self-assembly strategy to synthesize well-defined hierarchically porous polymers. Adv. Mater. 2019, 31, 1806254.
Qi, X. T.; Zhang, Z.; Tu, C. B.; Zhu, C.; Wei, J. C.; Yang, Z. Y. Covalent grafting interface engineering to prepare highly efficient and stable polypropylene/mesoporous SiO2 separator for Li-ion batteries. Appl. Surf. Sci. 2021, 541, 148405.
Carvalho, D. V.; Loeffler, N.; Kim, G. T.; Passerini, S. High temperature stable separator for lithium batteries based on SiO2 and hydroxypropyl guar gum. Membranes 2015, 5, 632–645.
Sheng, L.; Xie, X.; Arbizzani, C.; Bargnesi, L.; Bai, Y. Z.; Liu, G. J.; Dong, H. Y.; Wang, T.; He, J. P. A tailored ceramic composite separator with electron-rich groups for high-performance lithium metal anode. J. Membr. Sci. 2022, 657, 120644.
Quartarone, E.; Mustarelli, P. Electrolytes for solid-state lithium rechargeable batteries: recent advances and perspectives. Chem. Soc. Rev. 2011, 40, 2525–2540.
Wang, J.; Wang, C.; Wang, W.; Li, W.; Lou, J. Carboxymethylated nanocellulose-based gel polymer electrolyte with a high lithium ion transfer number for flexible lithium-ion batteries application. Chem. Eng. J. 2022, 428, 132604.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 21574087 and 51973128) and Science and Technology Department of Sichuan Province (Nos. 2019YJ0128 and 2019YFG0277). LL thanks the supports from the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no interest conflict.
Electronic Supplementary Information
Rights and permissions
About this article
Cite this article
Gong, X., Xiao, Q., Li, QY. et al. Cross-linked Electrospun Gel Polymer Electrolytes for Lithium-Ion Batteries. Chin J Polym Sci (2024). https://doi.org/10.1007/s10118-024-3136-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10118-024-3136-2