Abstract
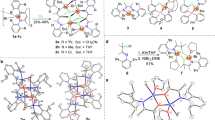
The highly efficient method has been developed for the synthesis of NHC-VOCl3 containing symmetrical or unsymmetrical N-heterocyclic carbene (NHC) ligands by the transmetallation reaction of NHG·AgCl with VOCl3. The total isolated yield of VOCl3[1,3-(2,4,6-Me3C6H2)2(NCH=)2C:] (V4′) reached 86% by transmetallation reaction, which is much higher than that (48%) by direct coordination method. This methodology has also been used to synthesize the novel vanadium complexes containing unsymmetrical NHC ligands of VOCl3[PhCH2NCH=CHNR)C:] (V5′, R = 2,4,6-Me3C6H2; V6′, R = 2,4-Me2-6-Ph-C6H2; V7′, R = 2,6-iPr2-C6H3) with high yield, which could not be obtained by direct coordination method. The catalytic activity and copolymerization ability would be improved by introducing unsymmetrical NHC ligands due to their less steric bulky effect. The vanadium complex V5′ containing unsymmetrical NHC ligand exhibits higher catalytic activity (3.7×105 gcopolymer·mol−1 of V·h−1) than that of V4′ containing symmetrical NHC ligand. Moreover, the higher propylene incorporation ratio (45.6 mol%) in the copolymers of ethylene with propylene could be obtained by using V5′ than that (39.9%) by using V4′. The results would provide a highly efficient strategy for the synthesis of early transition metal complexes containing versitile NHC ligands, affording the catalyst with both high catalytic activity and copolymerization ability for the synthesis of high performance polyolefin elastomers.
Similar content being viewed by others
References
Gibson, V. C.; Spitzmesser, S. K. Advances in non-metallocene olefin polymerization catalysis. Chem. Rev. 2003, 103, 283–315.
Nomura, K.; Zhang, S. Design of vanadium complex catalysts for precise olefin polymerization. Chem. Rev. 2011, 111, 2342–2362.
Makio, H.; Terao, H.; Iwashita, A.; Fujita, T. FI Catalysts for olefin polymerization—a comprehensive treatment. Chem. Rev. 2011, 111, 2363–2449.
Baier, M. C.; Zuideveld, M. A.; Mecking, S. Post-metallocenes in the industrial production of polyolefins. Angew. Chem. Int. Ed. 2014, 53, 9722–9744.
Zhang, S.; Zhang, Z. Q.; Wu, Y. X. Progress in advanced catalyst and their use in ethylene/propylene copolymerization. Chin. Sci. Bull. 2018, 63, 3530–3545.
Zhang, S.; Wu, Y. X. Controlled olefin polymerization and its macromolecular engineering. Sci. Sin. Chim. 2018, 48, 590–600.
Phillips, A. M. F.; Suo, H. Y.; da Silva, M. D. C. G.; Pombeiro, A. J. L.; Sun, W. H. Recent developments in vanadium-catalyzed olefin coordination polymerization. Coord. Chem. Rev. 2020, 416, e213332.
Yuan, S. F.; Yan, Y.; Solan, G. A.; Ma, Y. P.; Sun, W. H. Recent advancements in N-ligated group 4 molecular catalysts for the (co)polymerization of ethylene. Coord. Chem. Rev. 2020, 411, e213254.
Chen, J. Z.; Gao, Y. S.; Marks, T. J. Early transition metal catalysis for olefin-polar monomer copolymerization. Angew. Chem. Int. Ed. 2020, 59, 14726–14735.
Zhang, S.; Zhang, X. Y.; Wang, Y. C. Wu, Y. X. Progress in vanadium complex catalysts and their catalytic behavior toward copolymerization of ethylene with α-olefins. Chin. Sci. Bull 2021, 66, 3849–3865.
Bravaya, N. M.; Faingol’d, E. E.; Sanginov E. A.; Badamshina E. R. Homogeneous group IVB catalysts of new generations for synthesis of ethylene-propylene-diene rubbers: a mini-review. Catalysts 2022, 12, e704.
Ravishankar, P. S. Treatise on EPDM. Rubber Chem. Technol. 2012, 85, 327–349.
Redshaw, C.; Warford, L.; Elsegood, M. R. J.; Dale, S. H. Vanadyl complexes bearing bi- and triphenolate chelate ligands: highly active ethylene polymerisation procatalysts. Chem. Commun. 2004, 1954–1955.
Redshaw, C.; Rowan, M. A.; Homden, D. M.; Dale, S. H.; Elesgood, M. R. J.; Matsui, S.; Matsuura, S. Vanadyl C and N-capped tris (phenolate) complexes: influence of pro-catalyst geometry on catalytic activity. Chem. Commun. 2006, 3329–3331.
Redshaw, C.; Walton, M. J.; Elsegood, M. R. J.; Prior, T. J.; Michiue, K. Vanadium(V) tetra-phenolate complexes: synthesis, structural studies and ethylene homo-(co-) polymerization capability. RSC Adv. 2015, 5, 89783–89796.
Redshaw, C.; Rowan, M. A.; Warford, L.; Homden, D. M.; Arbaoui, A.; Elsegood, M. R. J.; Dale, S. H.; Yamato, T.; Casas, C. P.; Matsui, S.; Matsuura, S. Oxo- and imidovanadium complexes incorporating methylene- and dimethyleneoxa-bridged calix[3]- and -[4]arenes: synthesis, structures and ethylene polymerisation catalysis. Chem. Eur. J. 2007, 13, 1090–1107.
Redshaw, C.; Walton, M.; Michiue, K.; Chao, Y.; Walton, A.; Elo, P.; Sumerin, V.; Jiang, C.; Elsegood, M. Vanadyl calix[6]arene complexes: synthesis, structural studies and ethylene homo-(co-) polymerization capability. Dalton Trans. 2015, 44, 12292–12303.
Redshaw, C.; Walton, M. J.; Lee, D. S.; Jiang, C.; Elscood, M. R. J.; Michiue, K. Vanadium(V) oxo and imido calix[8] arene complexes: synthesis, structural studies, and ethylene homo/copolymerisation capability. Chem. Eur. J. 2015, 21, 5199–5210.
Zanchin, G.; Bertini, F.; Vendier, L.; Ricci, G.; Lorber C.; Leone, G. Copolymerization of ethylene with propylene and higher α-olefins catalyzed by (imido)vanadium(IV) dichloride complexes. Polym. Chem. 2019, 10, 6200–6216.
Desmangles, N.; Gambarotta, S.; Bensimon, C.; Davis, S.; Zahalka, H. Preparation and characterization of (R2N)2VCl2 [R= Cy, i-Pr] and its activity as olefin polymerization catalyst. J. Organomet. Chem. 1998, 562, 53–60.
Cuomo, C.; Milione, S.; Grassi, A. Olefin polymerization catalyzed by amide vanadium(IV) complexes: the stereo- and regiochemistry of propylene insertion. J. Polym. Sci., Part A: Polym. Chem. 2006, 44, 3279–3289.
Hagen, H.; Boersma, J.; Lutz, M.; Spek, A. L.; Koten, G. V. Vanadium (III) and -(IV) complexes with O, N-chelating aminophenolate ligands: synthesis, characterization and activity in ethene/propene copolymerization. Eur. J. Inorg. Chem. 2001, 117–123.
Mu, J. S.; Li, Y. S. Measurement and control of the microstructure of ethylene-propylene copolymers. Acta Polymerica Sinica 2013, 1492–1500.
Hao, X.; Zhang, C.; Li, L.; Zhang, H.; Hu, Y.; Hao, D.; Zhang, X. Use of vanadium complexes bearing naphthalene-bridged nitrogen-sulfonate ligands as catalysts for copolymerization of ethylene and propylene. Polymers 2017, 9, e325.
Michael, A. R.; Martin, H.; Jörg, S.; Li, X. Novel vanadium-imidoaryl compounds with electron-withdrawing groups on the aryl group are useful together with organo-metallic compounds as olefin polymerization catalysts. EP1284269A, 2001.
Zhang, S.; Zhang, W. C.; Shang, D. D.; Zhang, Z. Q.; Wu, Y. X. Ethylene/propylene copolymerization catalyzed by vanadium complexes containing N-heterocyclic carbenes. Dalton Trans. 2015, 44, 15264–15270.
Zhang, S.; Zhang, W. C.; Shang, D. D.; Wu, Y. X. Synthesis of ultra-high-molecular-weight ethylene-propylene copolymer via quasi-living copolymerization with N-heterocyclic carbene ligated vanadium complexes. J. Polym. Sci., Part A: Polym. Chem. 2019, 57, 553–561.
Zhang, S.; Wu, N.; Wu, Y. X. Highly cis-1,4 selective polymerization of conjugated dienes catalyzed by N-heterocyclic carbene-ligated neodymium complexes. Chinese J. Polym. Sci. 2020, 38, 1305–1312.
Abernethy, C. D.; Codd, G. M.; Spicer, M. D.; Taylor, M. K. A highly stable N-heterocyclic carbene complex of trichloro-oxo-vanadium(V) displaying novel Cl−C carbene bonding interaction. J. Am. Chem. Soc. 2003, 125, 1128–1129.
Lorber, C.; Vendier L. Synthesis and structure of early transition metal NHC complexes. Dalton Trans. 2009, 6972–6984.
Horrer, G.; Krummenacher, I.; Mann, S.; Braunschweig, H.; Radius, U. N-Heterocyclic carbene and cyclic (alkyl)(amino) carbene complexes of vanadium(III) and vanadium(V). Dalton Trans. 2022, 51, 11054–11071.
Arduengo III, A. J.; Harlow, R. L.; Kline, M. A stable crystalline carbene. J. Am. Chem. Soc. 1991, 113, 361–363.
Nelson, D. J. Accessible syntheses of late transition metal (pre)catalysts bearing W-heterocyclic carbene ligands. Eur. J. Inorg. Chem. 2015, 2012–2027.
Wang, H. M. J.; Lin, I. J. B. Facile synthesis of silver(I)-carbene complexes. Useful carbene transfer agents. Organometallics 1998, 17, 972–975.
Furst, M. R. L.; Cazin, C. S. J. Copper N-heterocyclic carbene (NHC) complexes as carbene transfer reagents. Chem. Commun. 2010, 46, 6924–6925.
Lin, J. C. Y.; Huang, R. T. W.; Lee, C. S.; Bhattacharyya, A.; Hwang, W. S.; Lin, I. J. B. Coinage metal-N-heterocyclic carbene complexes. Chem. Rev. 2009, 109, 3561–3598.
Nahra, F.; Gomez-Herrera, A.; Cazin, C. S. J. Copper(I)-NHC complexes as NHC transfer agents. Dalton Trans. 2017, 46, 628–631.
Garrison, J. C.; Youngs, W. J. Ag(I) N -heterocyclic carbene complexes: synthesis, structure, and application. Chem. Rev. 2005, 105, 3978–4008.
Bidal, Y. D.; Santoro, O.; Melaimi, M.; Cordes, D. B.; Slawin, A. M. Z.; Bertrand, G.; Cazin, C. S. J. Generalization of the copper to late-transition-metal transmetallation to carbenes beyond N-heterocyclic carbenes. Chem. Eur. J. 2016, 22, 9404–9409.
Danopoulos, A. A.; Simler, T.; Braunstein, P. N-heterocyclic carbene complexes of copper, nickel, and cobalt. Chem. Rev. 2019, 119, 3730–3961.
Mikhaylov, V. N.; Igor V. K.; Tatiana N. P.; Olesya V. K.; Manfred S.; Alexey Y. T.; Irina A. B. The carbene transfer to strong Lewis acids: copper is better than silver. Dalton Trans. 2021, 50, 2872–2879.
Al Thagfi, J.; Lavoie, G. G. Synthesis, characterization, and ethylene polymerization studies of chromium, iron, and cobalt complexes containing 1,3-bis(imino)-N-heterocyclic carbene ligands. Organometallics 2012, 31, 2463–2469.
Al Thagfi, J.; Lavoie, G. G. Preparation and reactivity study of chromium(III), iron(II), and cobalt(II) complexes of 1,3-bis(imino)benzimidazol-2-ylidene and 1,3-bis(imino)pyrimidin-2-yli-dene. Organometallics 2012, 31, 7351–7358.
Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. A.; Nakatsuji, H.; Caricato, M.; Li, X.; Hratchian, H. P.; Izmaylov, A. F.; Bloino, J.; Zheng, G.; Sonnenberg, J. L.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Montgomery, J. A., Jr.; Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd, J. J.; Brothers, E.; Kudin, K. N.; Staroverov, V. N.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Cossi, M.; Rega, N.; Millam, J. M.; Klene, M.; Knox, J. E.; Cross, J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Zakrzewski, V. G.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Dapprich, S.; Daniels, A. D.; Farkas, O.; Foresman, J. B.; Ortiz, J. V.; Cioslowski, J.; Fox, D. J. Gaussian 09, revision D.01; Gaussian, Inc.: Wallingford, CT, 2009.
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652.
Krishnan, R.; Binkley, J. S.; Seeger, R.; Pople, J. A. Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J. Chem. Phys. 1980, 72, 650–654.
Dolg, M.; Wedig, U.; Stoll, H.; Preuss, H. Energy-adjusted ab initio pseudopotentials for the first row transition elements. J. Chem. Phys. 1987, 86, 866–872.
Wang, Z.; Yu, Y.; Zhang, Y. X.; Li, S. Z.; Qian, H.; Lin, Z. Y. A magnetically separable palladium catalyst containing a bulky N-heterocyclic carbene ligand for the Suzuki-Miyaura reaction. Green Chem. 2015, 17, 413–420.
Belger, K.; Krause, N. Smaller, faster, better: modular synthesis of unsymmetrical ammonium salt-tagged NHC-gold(I) complexes and their application as recyclable catalysts in water. Org. Biomol. Chem. 2015, 13, 8556–8560.
Falivene, L.; Cao, Z.; Petta, A.; Serra, L.; Poater, A.; Oliva, R.; Scarano, V.; Cavallo, L. Towards the online computer-aided design of catalytic pockets. Nat. Chem. 2019, 11, 872–879.
Wang, W. J.; Zhu, S. P. Structural analysis of ethylene/propylene copolymers synthesized with a constrained geometry catalyst. Macromolecules 2000, 33, 1157–1162.
Lu, T.; Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592.
Humphrey, W.; Dalke, A.; Schulten, K. VMD: visual molecular dynamics. J. Mol. Graphics 1996, 14, 33–38.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 21774006 and 21634002).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no interest conflict.
Additional information
S.Z. dedicated this work to Prof. Wen-Hua Sun in the occasion of his 60th birthday.
10118_2023_3020_MOESM1_ESM
10118_2023_3020_MOESM1_ESM.pdf
Vanadium(V) Complexes Containing Unsymmetrical N-Heterocyclic Carbene Ligands: Highly Efficient Synthesis and Catalytic Behavior Towards Ethylene/Propylene Copolymerization
Rights and permissions
About this article
Cite this article
Wang, YC., Zha, H., Cheng, PY. et al. Vanadium(V) Complexes Containing Unsymmetrical N-Heterocyclic Carbene Ligands: Highly Efficient Synthesis and Catalytic Behavior towards Ethylene/Propylene Copolymerization. Chin J Polym Sci 42, 32–41 (2024). https://doi.org/10.1007/s10118-023-3020-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-023-3020-5