Abstract
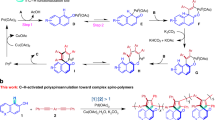
Spiropolymers have gained a great deal of interest from both academic and industrial fields by virtue of their unique geometric structures and physical properties. Herein, we prepared a series of spirocopolymers through the catalyst-free four-component spiropolymerization of diisocyanides, activated alkynes, and two different kinds of monomers with reactive carbonyl groups. It is found that the polymerization reactivity of monomers, feeding modes, and feed ratios play significant roles in spirocopolymerization. Monomers with high reactivity and feeding reactive monomers first contribute to improving the molecular weights and yields of the polymers. The constructed copolymers have two different kinds of spiro structures, which is confirmed by the nuclear magnetic resonance. In addition, the spirocopolymers display the unique cluster-triggered emission and aggregation-induced emission properties, and their emission properties can be well-modulated by altering the ratio of comonomers. It is highly anticipated that this line of research will enrich the methodology of multi-component spiropolymerization, and provide a new insight into developing spiropolymers with various spiro structures and tunable properties.
Similar content being viewed by others
References
Liaw, D. J.; Wang, K. L.; Huang, Y. C.; Lee, K. R.; Lai, J. Y.; Ha, C. S. Advanced polyimide materials: syntheses, physical properties and applications. Prog. Polym. Sci. 2012, 37, 907–974.
Pham, T. H.; Olsson, J. S.; Jannasch, P. N-spirocyclic quaternary ammonium ionenes for anion-exchange membranes. J. Am. Chem. Soc. 2017, 139, 2888–2891.
Han, T.; Yao, Z.; Qiu, Z.; Zhao, Z.; Wu, K.; Wang, J.; Poon, A. W.; Lam, J. W. Y.; Tang, B. Z. Photoresponsive spiro-polymers generated in situ by C-H-activated polyspiroannulation. Nat. Commun. 2019, 10, 5483.
Thompson, K. A.; Mathias, R.; Kim, D.; Kim, J.; Rangnekar, N.; Johnson, J. R.; Hoy, S. J.; Bechis, I.; Tarzia, A.; Jelfs, K. E.; McCool, B. A.; Livingston, A. G.; Lively, R. P.; Finn, M. G. N-aryl-linked spirocyclic polymers for membrane separations of complex hydrocarbon mixtures. Science 2020, 369, 310–315.
Cai, Z.; Ren, Y.; Li, X.; Shi, J.; Tong, B.; Dong, Y. Functional isocyanide-based polymers. Acc. Chem. Res. 2020, 53, 2879–2891.
Wang, M.; Li, C.; Lv, A.; Wang, Z.; Bo, Z. Spirobifluorene-based conjugated polymers for polymer solar cells with high open-circuit voltage. Macromolecules 2012, 45, 3017–3022.
Wang, X.; Zhao, L.; Shao, S.; Ding, J.; Wang, L.; Jing, X.; Wang, F. Poly(spirobifluorene)s containing nonconjugated diphenylsulfone moiety: toward blue emission through a weak charge transfer effect. Macromolecules 2014, 47, 2907–2914.
Valero, S.; Collavini, S.; Völker, S. F.; Saliba, M.; Tress, W. R.; Zakeeruddin, S. M.; Grätzel, M.; Delgado, J. L. Dopant-free hole-transporting polymers for efficient and stable perovskite solar cells. Macromolecules 2019, 52, 2243–2254.
Ma, J.; Tian, J.; Liu, Z.; Shi, D.; Zhang, X.; Zhang, G.; Zhang, D. Multi-stimuli-responsive field-effect transistor with conjugated polymer entailing spiropyran in the side chains. CCS Chem. 2019, 1, 632–641.
Vidavsky, Y.; Yang, S. J.; Abel, B. A.; Agami, I.; Diesendruck, C. E.; Coates, G. W.; Silberstein, M. N. Enabling room-temperature mechanochromic activation in a glassy polymer: synthesis and characterization of spiropyran polycarbonate. J. Am. Chem. Soc. 2019, 141, 10060–10067.
Wu, Y.; Zhang, J.; Fei, Z.; Bo, Z. Spiro-bridged ladder-type poly(p-phenylene)s: towards structurally perfect light-emitting materials. J. Am. Chem. Soc. 2008, 130, 7192–7193.
Jiang, J.-X.; Laybourn, A.; Clowes, R.; Khimyak, Y. Z.; Bacsa, J.; Higgins, S. J.; Adams, D. J.; Cooper, A. I. High surface area contorted conjugated microporous polymers based on spiro-bipropylenedioxythiophene. Macromolesules 2010, 43, 7577–7582.
Bezzu, C. G.; Carta, M.; Tonkins, A.; Jansen, J. C.; Bernardo, P.; Bazzarelli, F.; McKeown, N. B. A spirobifluorene-based polymer of intrinsic microporosity with improved performance for gas separation. Adv. Mater. 2012, 24, 5930–5933.
Ma, X.; Salinas, O.; Litwiller, E.; Pinnau, I. Novel spirobifluorene- and dibromospirobifluorene-based polyimides of intrinsic microporosity for gas separation applications. Macromolecules 2013, 46, 9618–9624.
Zhao, Y. C.; Zhang, L. M.; Wang, T.; Han, B. H. Microporous organic polymers with acetal linkages: synthesis, characterization, and gas sorption properties. Polym. Chem. 2014, 5, 614–621.
Shamsipur, H.; Dawood, B. A.; Budd, P. M.; Bernardo, P.; Clarizia, G.; Jansen, J. C. Thermally rearrangeable PIM-polyimides for gas separation membranes. Macromolecules 2014, 47, 5595–5606.
McDowell, J. J.; Gao, D.; Seferos, D. S.; Ozin, G. Synthesis of poly(spirosilabifluorene) copolymers and their improved stability in blue emitting polymer LEDs over non-spiro analogs. Polym. Chem. 2015, 6, 3781–3789.
Olsson, J. S.; Pham, T. H.; Jannasch, P. Poly(N,N-diallylazacycloalkane)s for anion-exchange membranes functionalized with N-spirocyclic quaternary ammonium cations. Macromolecules 2017, 50, 2784–2793.
Nagatsu, G.; Sakanoue, T.; Tane, S.; Yonekawa, F.; Takenobu, T. An ester-substituted polyfluorene derivative for light-emitting electrochemical cells: bright blue emission and its application in a host-guest system. Mater. Chem. Front. 2018, 2, 952–958.
Xu, X.; Li, X.; Wang, S.; Ding, J.; Wang, L. Deep-blue emitting poly[spiro(dibenzoazasiline-10′,9-silafluorene)] for power-efficient PLEDs. J. Mater. Chem. C 2018, 6, 9599–9606.
Foster, A. B.; Tamaddondar, M.; Luque-Alled, J. M.; Harrison, W. J.; Li, Z.; Gorgojo, P.; Budd, P. M. Understanding the topology of the polymer of intrinsic microporosity PIM-1: cyclics, tadpoles, and network structures and their impact on membrane performance. Macromolecules 2020, 53, 569–583.
Pemba, A. G.; Rostagno, M.; Lee, T. A.; Miller, S. A. Cyclic and spirocyclic polyacetal ethers from lignin-based aromatics. Polym. Chem. 2014, 5, 3214–3221.
Okuda, H.; Koyama, Y.; Uchida, S.; Michinobu, T.; Sogawa, H.; Takata, T. Reversible transformation of a one-handed helical foldamer utilizing a planarity-switchable spacer and C2-chiral spirobifluorene units. ACS Macro Lett. 2015, 4, 462–466.
K, S. N.; Azechi, M.; Endo, T. Synthesis and properties of spiro-centered benzoxazines. Macromolecules 2015, 48, 7466–7472.
Zhao, Y. C.; Wang, T.; Zhang, L. M.; Cui, Y.; Han, B. H. Microporous spiro-centered poly(benzimidazole) networks: preparation, characterization, and gas sorption properties. Polym. Chem. 2015, 6, 748–753.
Modak, A.; Maegawa, Y.; Goto, Y.; Inagaki, S. Synthesis of 9,9′-spirobifluorene-based conjugated microporous polymers by FeCl3-mediated polymerization. Polym. Chem. 2016, 7, 1290–1296.
Sycks, D. G.; Safranski, D. L.; Reddy, N. B.; Sun, E.; Gall, K. Tough semicrystalline thiol-ene photopolymers incorporating spiroacetal alkenes. Macromolecules 2017, 50, 4281–4291.
Schmidt, S. B.; Kempe, F.; Brügner, O.; Walter, M.; Sommer, M. Alkyl-substituted spiropyrans: electronic effects, model compounds and synthesis of aliphatic main-chain copolymers. Polym. Chem. 2017, 8, 5407–5414.
Feng, Q. Y.; Li, B.; Zuo, Z. Y.; Xie, S. L.; Yu, M. N.; Liu, B.; Wei, Y.; Xie, L. H.; Xia, R. D.; Huang, W. A comparison study of physicochemical properties and stabilities of H-shaped molecule and the corresponding polymer. Chinese J. Polym. Sci. 2019, 37, 11–17.
Fang, L.; Zhou, J.; He, C.; Tao, Y.; Wang, C.; Dai, M.; Wang, H.; Sun, J.; Fang, Q. Understanding how intrinsic micro-pores affect the dielectric properties of polymers: an approach to synthesize ultra-low dielectric polymers with bulky tetrahedral units as cores. Polym. Chem. 2020, 11, 2674–2680.
Tan, W. Y.; Jian, L. F.; Chen, W. P.; Zhang, Y. W.; Lu, X. C.; Huang, W. J.; Zhang, J. S.; Wu, J. W.; Feng, J. L.; Liu, Y. D.; Cui, T. T.; Min, Y. G. A facile strategy for intrinsic low-Dk and low-Df polyimides enabled by spirobifluorene groups. Chinese J. Polym. Sci. 2023, 41, 288–296.
Fu, W. Q.; Zhu, G. N.; Shi, J. B.; Tong, B.; Cai, Z. X.; Dong, Y. P. Synthesis and properties of photodegradable poly(furan-amine)s by a catalyst-free multicomponent cyclopolymerization. Chinese J. Polym. Sci. 2019, 37, 981–989.
Liu, P.; Fu, W.; Verwilst, P.; Won, M.; Shin, J.; Cai, Z.; Tong, B.; Shi, J.; Dong, Y.; Kim, J. S. MDM2-associated clusterization-triggered emission and apoptosis induction effectuated by a theranostic spiropolymer. Angew. Chem. Int. Ed. 2020, 59, 8435–8439.
Deng, X. X.; Li, L.; Li, Z. L.; Lv, A.; Du, F. S.; Li, Z. C. Sequence regulated poly(ester-amide)s based on passerini reaction. ACS Macro Lett. 2012, 1, 1300–1303.
Han, T.; Deng, H.; Qiu, Z.; Zhao, Z.; Zhang, H.; Zou, H.; Leung, N. L. C.; Shan, G.; Elsegood, M. R. J.; Lam, J. W. Y.; Tang, B. Z. Facile multicomponent polymerizations toward unconventional luminescent polymers with readily openable small heterocycles. J. Am. Chem. Soc. 2018, 140, 5588–5598.
Zhang, J.; Wu, Y. H.; Wang, J. C.; Du, F. S.; Li, Z. C. Functional poly(ester-amide)s with tertiary ester linkages via the passerini multicomponent polymerization of a dicarboxylic acid and a diisocyanide with different electron-deficient ketones. Macromolecules 2018, 51, 5842–5851.
Wu, X.; Lin, H.; Dai, F.; Hu, R.; Tang, B. Z. Functional polyselenoureas for selective gold recovery prepared from catalyst-free multicomponent polymerizations of elemental selenium. CCS Chem. 2020, 2, 191–202.
Liu, X.; Xiao, M.; Xue, K.; Li, M.; Liu, D.; Wang, Y.; Yang, X.; Hu, Y.; Kwok, R. T. K.; Qin, A.; Zhu, C.; Lam, J. W. Y.; Tang, B. Z. Heteroaromatic hyperbranched polyelectrolytes: multicomponent polyannulation and photodynamic biopatterning. Angew. Chem. Int. Ed. 2021, 60, 19222–19231.
Wu, X.; He, J.; Hu, R.; Tang, B. Z. Room-temperature metal-free multicomponent polymerizations of elemental selenium toward stable alicyclic poly(oxaselenolane)s with high refractive index. J. Am. Chem. Soc. 2021, 143, 15723–15731.
Zhang, J.; Zang, Q.; Yang, F.; Zhang, H.; Sun, J. Z.; Tang, B. Z. Sulfur conversion to multifunctional poly(O-thiocarbamate)s through multicomponent polymerizations of sulfur, diols, and diisocyanides. J. Am. Chem. Soc. 2021, 143, 3944–3950.
Li, M.; Duan, X.; Jiang, Y.; Sun, X.; Xu, X.; He, J.; Zheng, Y.; Song, W.; Zheng, N. Three-component asymmetric polymerization toward chiral polymer. CCS Chem. 2022, 4, 3402–3415.
Li, M.; Fu, X.; Wang, J.; Qin, A.; Tang, B. Z. Progress in isocyanide-based step-growth polymerization. Macromol. Chem. Phys. 2022, 224, 2200352.
Ren, Y.; Dai, W.; Guo, S.; Dong, L.; Huang, S.; Shi, J.; Tong, B.; Hao, N.; Li, L.; Cai, Z.; Dong, Y. Clusterization-triggered color-tunable room-temperature phosphorescence from 1,4-dihydropyridine-based polymers. J. Am. Chem. Soc. 2022, 144, 1361–1369.
Yan, H.; He, Y.; Wang, D.; Han, T.; Tang, B. Z. Aggregation-induced emission polymer systems with circularly polarized luminescence. Aggregate 2022, 4, e331.
Fu, X.; Qin, A.; Tang, B. Z. X-yne click polymerization. Aggregate 2022, 4, e350.
Nair, V.; Vinod, A. U.; Nair, J. S.; Sreekanth, A. R.; Rath, N. P. The reaction of cyclohexyl isocyanide and dimethyl acetylenedicarboxylate with o- and p-quinones: a novel synthesis of iminolactones. Tetrahedron Lett. 2000, 41, 6675–6679.
Nair, V.; Vinod, A. U.; Abhilash, N.; Menon, R. S.; Santhi, V.; Varma, R. L.; Viji, S.; Mathew, S.; Srinivas, R. Multicomponent reactions involving zwitterionic intermediates for the construction of heterocyclic systems: one pot synthesis of aminofurans and iminolactones. Tetrahedron 2002, 59, 10279–10286.
Yavari, I.; Djahaniani, H. One-step synthesis of substituted 4,7-bis[alkyl(aryl)imino]-3-oxa-6-thia-1-azaspiro[4.4]nona-1,8-dienes. Tetrahedron Lett. 2005, 46, 7491–7493.
Nair, V.; Menon, R. S. Nucleophile-initiated catalytic and multicomponent reactions. Chem. Rec. 2019, 19, 347–361.
Zhu, G.; Lin, N.; Wu, X.; Shi, J.; Tong, B.; Cai, Z.; Zhi, J.; Dong, Y. Multicomponent spiropolymerization of diisocyanides, activated alkynes, and bis-anhydrides. Macromolecules 2022, 55, 6150–6159.
Zhang, J.; Jin, J.; Cooney, R.; Fu, Q.; Qiao, G. G.; Thomas, S.; Merkel, T. C. Synthesis of perfectly alternating copolymers for polymers of intrinsic microporosity. Polym. Chem. 2015, 6, 5003–5008.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 21875019, 22175023, 21975020 and 21975021), the National Key Research and Development Program of China (No. 2018YFA0901800) College Students’ Innovative Entrepreneurial Training Plan Program (No. BIT2022LH180) and Beijing Institute of Technology Research Fund Program for Young Scholars.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no interest conflict.
Electronic Supplementary Information
10118_2023_3007_MOESM1_ESM.pdf
Catalyst-Free Four-Component Spiropolymerization for the Construction of Spirocopolymers with Tunable Photophysical Properties
Rights and permissions
About this article
Cite this article
Zhu, LJ., Zhu, GN., Yan, WY. et al. Catalyst-Free Four-Component Spiropolymerization for the Construction of Spirocopolymers with Tunable Photophysical Properties. Chin J Polym Sci 41, 1525–1532 (2023). https://doi.org/10.1007/s10118-023-3007-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-023-3007-2