Abstract
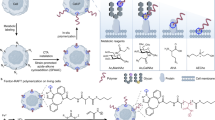
Dynamic control of mesenchymal stem cell (MSC) behaviors on biomaterial surface is critically involved in regulating the cell fate and tissue regeneration. Herein, a stimuli-responsive surface based on host-guest interaction with cell selectivity was developed to regulate migration of MSCs in situ by dynamic display of cell-specific peptides. Azobenzene-grafted MSC-affinitive peptides (EPLQLKM, Azo-E7) were grafted to β-cyclodextran (β-CD)-modified poly(2-hydroxyethyl methacrylate)-b-poly(2-hydroxyethyl methacrylate-co-glycidyl methacrylate) (PHG) brushes, which were prepared by using surface-initiated atom transfer radical polymerization (SI-ATRP). X-ray photoelectron spectroscopy (XPS), quartz crystal microbalance (QCM), and water contact angle were used to characterize their structure and property. Cell adhesion assay showed that the combination effect of resisting property of PHG and MSC-affinity of E7 could promote the selective adhesion of MSCs over other types of cells such as RAW264.7 macrophages and NIH3T3 fibroblasts to some extent. UV-Vis spectroscopy proved that the competing guest molecules, amantadine hydrochloride (Ama), could release Azo-E7 peptides from the CD surface to different extents, and the effect was enhanced when UV irradiation was employed simultaneously. As a result, the decrease of cell adhesion density and migration rate could be achieved in situ. The cell density and migration rate could be reduced by over 40% by adding 20 μmol/L Ama, suggesting that this type of surface is a new platform for dynamic regulation of stem cell behaviors in situ.
Similar content being viewed by others
Change history
25 July 2020
The authors regret that Fig. 4(c) was misplaced by the first author originally. The corrected version of Fig. 4 is provided below. The quantitative analysis shown in Fig. 4(b) is not changed, and thus the conclusions related to Fig. 4 and reported in this article are not altered. Additional text corrections are not needed. The authors apologize for any inconvenience caused.
References
Ermis, M.; Antmen, E.; Hasirci, V. Micro and nanofabrication methods to control cell-substrate interactions and cell behavior: A review from the tissue engineering perspective. Bioact. Mater.2018, 3, 355–369.
Byron, A.; Morgan, M. R.; Humphries, M. J. Adhesion signalling complexes. Curr. Biol.2010, 20, R1063–R1067.
Geiger, B.; Spatz, J. P.; Bershadsky, A. D. Environmental sensing through focal adhesions. Nat. Rev. Microbiol.2009, 10, 21–33.
Zamani, M.; Prabhakaran, M. P.; Thian, E. S.; Ramakrishna, S. Controlled delivery of stromal derived factor-1α from poly(lactic-co-glycolic acid) core-shell particles to recruit mesenchymal stem cells for cardiac regeneration. J. Colloid Interface Sci.2015, 451, 144–152.
Yan, C.; Sun, J.; Ding, J. D. Critical areas of cell adhesion on micropatterned surfaces. Biomaterials2011, 32, 3931–3938.
Okuyama, H.; Krishnamachary, B.; Zhou, Y. F.; Nagasawa, H.; Bosch-Marce, M.; Semenza, G. L. Expression of vascular endothelial growth factor receptor 1 in bone marrow-derived mesenchymal cells is dependent on hypoxia-inducible factor 1. J. Biol. Chem.2006, 281, 15554–15563.
Wu, C. T.; Chang, J.; Zhai, W. Y.; Ni, S. Y.; Wang, J. Y. Porous akermanite scaffolds for bone tissue engineering: Preparation, characterization, and in vitro studies. J. Biomed. Mater. Res. B2006, 78B, 47–55.
Charras, G.; Sahai, E. Physical influences of the extracellular environment on cell migration. Nat. Rev. Microbiol.2014, 15, 813–824.
Chan, C. E.; Odde, D. J. Traction dynamics of filopodia on compliant substrates. Science2008, 322, 1687–1691.
Shabbir, S. H.; Eisenberg, J. L.; Mrksich, M. An inhibitor of a cell adhesion receptor stimulates cell Migration. Angew. Chem. Int. Ed.2010, 49, 7706–7709.
Wu, J. D.; Mao, Z. W.; Gao, C. Y. Controlling the migration behaviors of vascular smooth muscle cells by methoxy poly(ethylene glycol) brushes of different molecular weight and density. Biomaterials2012, 33, 810–820.
Biazar, E.; Khorasani, M. T.; Joupari, M. D. Cell adhesion and surface properties of polystyrene surfaces grafted with poly(N-isopropylacrylamide). Chinese J. Polym Sci.2013, 31, 1509–1518.
Cao, Z. Q.; Bian, Q.; Chen, Y.; Liang, F. X.; Wang, G. J. Light-responsive Janus-particle-based coatings for cell capture and release. ACS Macro Lett.2017, 6, 1124–1128.
Chen, Y. H.; Chang, S. H.; Wang, T. J.; Wang, I.; Young, T. H. Cell fractionation on pH-responsive chitosan surface. Biomaterials2013, 34, 854–863.
Xiao, Y.; Zhou, H. Y.; Xuan, N. X.; Cheng, M.; Rao, Y. F.; Luo, Y.; Wang, B.; Tang, R. Effective and selective cell retention and recovery from whole blood by electroactive thin films. ACS Appl. Mater. Interfaces2014, 6, 20804–20811.
Liu, H. Y.; Korc, M.; Lin, C. C. Biomimetic and enzyme-responsive dynamic hydrogels for studying cell-matrix interactions in pancreatic ductal adenocarcinoma. Biomaterials2018, 160, 24–36.
Costa, P.; Gautrot, J. E.; Connelly, J. T. Directing cell migration using micropatterned and dynamically adhesive polymer brushes. Acta Biomater.2014, 10, 2415–2422.
Boekhoven, J.; Perez, C. M. R.; Sur, S.; Worthy, A.; Stupp, S. I. Dynamic display of bioactivity through host-guest chemistry. Angew. Chem. Int. Ed.2013, 52, 12077–12080.
Voskuhl, J.; Sankaran, S.; Jonkheijm, P. Optical control over bioactive ligands at supramolecular surfaces. Chem. Commun.2014, 50, 15144–15147.
Gong, Y. H.; Li, C.; Yang, J.; Wang, H. Y.; Zhuo, R. X.; Zhang, X. Z. Photoresponsive “smart template” via host-guest interaction for reversible cell adhesion. Macromolecules2011, 44, 7499–7502.
Deng, J.; Liu, X. Y.; Shi, W. B.; Cheng, C.; He, C.; Zhao, C. S. Light-triggered switching of reversible and alterable biofunctionality via β-cyclodextrin/azobenzene-based host-guest interaction. ACS Macro Lett.2014, 3, 1130–1133.
Bian, Q.; Wang, W.; Wang, S.; Wang, G. Light-triggered specific cancer cell release from cyclodextrin/azobenzene and aptamer-modified substrate. ACS Appl. Mater. Interfaces2016, 8, 27360–27367.
Gu, L. Q.; Braha, O.; Conlan, S.; Cheley, S.; Bayley, H. Stochastic sensing of organic analytes by a pore-forming protein containing a molecular adapter. Nature1999, 398, 686–690.
Cheng, M. J.; Shi, F.; Li, J. S.; Lin, Z. F.; Jiang, C.; Xiao, M.; Zhang, L. Q.; Yang, W. T.; Nishi, T. Macroscopic supramolecular assembly of rigid building blocks through a flexible spacing coating. Adv. Mater.2014, 26, 3009–3013.
Khan, M.; Yang, J.; Shi, C. C.; Lv, J.; Feng, Y. K.; Zhang, W. C. Surface tailoring for selective endothelialization and platelet inhibition via a combination of SI-ATRP and click chemistry using Cys-Ala-Gly-peptide. Acta Biomater.2015, 20, 69–81.
Ren, T. C.; Yu, S.; Mao, Z. W.; Gao, C. Y. A complementary density gradient of zwitterionic polymer brushes and NCAM peptides for selectively controlling directional migration of schwann cells. Biomaterials2015, 56, 58–67.
Ji, Y.; Wei, Y.; Liu, X. S.; Wang, J. L.; Ren, K. F.; Ji, J. Zwitterionic polycarboxybetaine coating functionalized with REDV peptide to improve selectivity for endothelial cells. J. Biomed. Mater. Res. A2012, 100A, 1387–1397.
Zheng, X. W.; Pan, X.; Pang, Q.; Shuai, C. A.; Ma, L.; Gao, C. Y. Selective capture of mesenchymal stem cells over fibroblasts and immune cells on E7-modified collagen substrates under flow circumstances. J. Mater. Chem. B2018, 6, 165–173.
Man, Z. T.; Yin, L.; Shao, Z. X.; Zhang, X.; Hu, X. Q.; Zhu, J. X.; Dai, L. H.; Huang, H. J.; Yuan, L.; Zhou, C. Y. The effects of co-delivery of BMSC-affinity peptide and rhTGF-β1 from coaxial electrospun scaffolds on chondrogenic differentiation. Biomaterials2014, 35, 5250–5260.
Shao, Z. X.; Zhang, X.; Pi, Y. B.; Wang, X. K.; Jia, Z. Q.; Zhu, J. X.; Dai, L. H.; Chen, W. Q.; Yin, L.; Chen, H. F. Polycaprolactone electrospun mesh conjugated with an MSC affinity peptide for MSC homing in vivo. Biomaterials2012, 33, 3375–3387.
Yang, M.; Chu, L. Y.; Wang, H. D.; Xie, R.; Song, H.; Niu, C. H. A thermoresponsive membrane for chiral resolution. Adv. Funct. Mater.2008, 18, 652–663.
Tugulu, S.; Arnold, A.; Sielaff, I.; Johnsson, K.; Klok, H. A. Protein-functionalized polymer brushes. Biomacromolecules2005, 6, 1602–1607.
Ren, T. C.; Mao, Z. W.; Guo, J.; Gao, C. Y. Directional migration of vascular smooth muscle cells guided by a molecule weight gradient of poly(2-hydroxyethyl methacrylate) brushes. Langmuir2013, 29, 6386–6395.
Ren, T. C.; Mao, Z. W.; Moya, S. E.; Gao, C. Y. Immobilization of enzymes on 2-hydroxyethyl methacrylate and glycidyl methacrylate copolymer brushes. Chem. Asian J.2014, 9, 2132–2139.
Huang, S.; Xu, L. L.; Sun, Y. X.; Wu, T. Y.; Wang, K. X.; Li, G. An improved protocol for isolation and culture of mesenchymal stem cells from mouse bone marrow. J. Orthop. Transl.2015, 3, 26–33.
Li, X.; Wang, M. M.; Wang, L.; Shi, X. J.; Xu, Y. J.; Song, B.; Chen, H. Block copolymer modified surfaces for conjugation of biomacromolecules with control of quantity and activity. Langmuir2013, 29, 1122–1128.
Yan, S.; Wang, Z. N.; Gao, X. L.; Gao, C. J. Antifouling PVDF ultrafiltration membranes incorporating PVDF-g-PHEMA additive via atom transfer radical graft polymerizations. J. Membr. Sci.2012, 413-414, 38–47.
Zhu, L. J.; Zhu, L. P.; Jiang, J. H.; Yi, Z.; Zhao, Y. F.; Zhu, B. K.; Xu, Y. Y. Hydrophilic and anti-fouling polyethersulfone ultrafiltration membranes with poly(2-hydroxyethyl methacrylate) grafted silica nanoparticles as additive. J. Membr. Sci.2014, 451, 157–168.
Barbey, R.; Klok, H. Room temperature, aqueous postpolymerization modification of glycidyl methacrylate-containing polymer brushes prepared via surface-initiated atom transfer radical polymerization. Langmuir2010, 26, 18219–18230.
Kim, M.; Lee, B.; Yang, K.; Park, J.; Jeon, S.; Um, S. H.; Kim, D.; Im, S. G.; Cho, S. BMP-2 peptide-functionalized nanopatterned substrates for enhanced osteogenic differentiation of human mesenchymal stem cells. Biomaterials2013, 34, 7236–7246.
Zhou, W. F.; Yang, M.; Zhao, Z.; Li, S. P.; Cheng, Z. Q.; Zhu, J. S. Controlled hierarchical architecture in poly[oligo(ethylene glycol) methacrylate-b-glycidyl methacrylate] brushes for enhanced label-free biosensing. Appl. Surf. Sci.2018, 450, 236–243.
Hu, W. H.; Liu, Y. S.; Lu, Z. S.; Li, C. M. Poly[oligo(ethylene glycol) methacrylate-co-glycidyl methacrylate] brush substrate for sensitive surface plasmon resonance imaging protein arrays. Adv. Funct. Mater.2010, 20, 3497–3503.
Lei, Z.; Gao, J. X.; Liu, X.; Liu, D. J.; Wang, Z. X. Poly(glycidyl methacrylate-co-2-hydroxyethyl methacrylate) brushes as peptide/protein microarray substrate for improving protein binding and functionality. ACS Appl. Mater. Interfaces2016, 8, 10174–10182.
Wu, S.; Du, W.; Duan, Y. Y.; Zhang, D. T.; Liu, Y. X.; Wu, B. B.; Zou, X. H.; Ouyang, H. W.; Gao, C. Y. Regulating the migration of smooth muscle cells by a vertically distributed poly(2-hydroxyethyl methacrylate) gradient on polymer brushes covalently immobilized with RGD peptides. Acta Biomater.2018, 75, 75–92.
Dehghani, E. S.; Spencer, N. D.; Ramakrishna, S. N.; Benetti, E. M. Crosslinking polymer brushes with ethylene glycol-containing segments: Influence on physicochemical and antifouling properties. Langmuir2016, 32, 10317–10327.
Mandal, J.; Varunprasaath, R. S.; Yan, W.; Divandari, M.; Spencer, N. D.; Dubner, M. In situ monitoring of SI-ATRP throughout multiple reinitiations under flow by means of a quartz crystal microbalance. RSC Adv.2018, 8, 20048–20055.
Fuhrmann, I.; Karger-Kocsis, J. Photoinitiated grafting of glycidyl methacrylate and methacrylic acid on ground tire rubber. J. Appl. Polym. Sci.2003, 89, 1622–1630.
Tabata, Y. Biomaterial technology for tissue engineering applications. J. R. Soc. Interface2009, 63, S311–S324.
Ren, T. C.; Ni, Y. L.; Du, W.; Yu, S.; Mao, Z.; Gao, C. Y. Dual responsive surfaces based on host-guest interaction for dynamic mediation of cell-substrate interaction and cell migration. Adv. Mater. Interfaces2017, 4, 1500865.
Lauffenburger, D. A.; Horwitz, A. F. Cell migration: A physically integrated molecular process. Cell1996, 84, 359–369.
Dimilla, P. A.; Stone, J. A.; Quinn, J. A.; Albelda, S. M.; Lauffenburger, D. A. Maximal migration of human smooth muscle cells on fibronectin and type IV collagen occurs at an intermediate attachment strength. J. Cell Bio.1993, 122, 729–737.
Acknowledgments
This work was financially supported by the National Key Research and Development Program of China (No. 2016YFC1100403) and the National Natural Science Foundation of China (Nos. 21434006 and 51873188).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Information
10118_2019_2324_MOESM1_ESM.pdf
Mediating the Migration of Mesenchymal Stem Cells by Dynamically Changing the Density of Cell-selective Peptides Immobilized on β Cyclodextrin-modified Cell-resisting Polymer
Rights and permissions
About this article
Cite this article
Du, W., Zhang, DT., Wang, XM. et al. Mediating the Migration of Mesenchymal Stem Cells by Dynamically Changing the Density of Cell-selective Peptides Immobilized on β-Cyclodextrin-modified Cell-resisting Polymer Brushes. Chin J Polym Sci 38, 126–136 (2020). https://doi.org/10.1007/s10118-019-2324-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-019-2324-y