Abstract
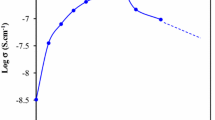
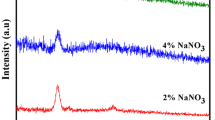
Polyethylene oxide (PEO)-polyvinylpyrrolidone (PVP) blended Na+ ion conducting solid polymeric membranes: (1−x) [75PEO:25NaPO3] + x PVP, where 0 < x < 12 wt%, are reported. The polymeric blending was done using a solventfree hot-press method. Two orders of conductivity enhancement (σ ca. 1.07 × 10−5 S·cm−1) have been achieved with 3 wt% of PVP (i.e. the composition: [97(75PEO:25NaPO3) + 3PVP]), from that of the pure host: (75PEO:25NaPO3). The conductivity enhancement in PEO-PVP blended solid polymeric membranes have been explained by the ionic conductivity, ionic mobility and mobile ion concentration measurements. Materials characterization and polymer-salt complexation were done with the help of X-ray diffraction (XRD), scanning electron microscopy (SEM), differential scanning calorimetry (DSC) and thermo gravimetric analysis (TGA) studies. The temperature dependent conductivity studies have also been done to compute the activation energy (E a) values from lg σ1/T Arrhenius plots. A solid state polymeric battery was fabricated by using optimum conducting composition of solid polymer electrolyte (SPE OCC), and some important cell parameters were also calculated from the discharge profile of the cell.
Similar content being viewed by others
References
Van Gool, W., “Fast ionic transport in solids”, North Holland Pub. Co., Amsterdam, New York, 1973
Mahan, G.D. and Roth, W.L., “Superionic conductors”, Plenum Press, New York, 1976
Chandra, S., “Superionic solids-principles and applications”, North Holland Pub. Co., Amsterdam, New York, 1981
Laskar, A.L. and Chandra, S., “Superionic solids and solid electrolytes-recent trends”, Academic Press, New York, 1989
Berthier, C., Gorecki, W., Miner, M., Armand, M.B., Chabagno, J.M. and Rigaud, P., Solid State Ionics, 1983, 11: 91
Armand, M.B., Adv. Mat., 1990, 2: 278
Gray, F.M., “Polymer electrolytes”, Royal Society of Chemistry, Letchworth HN, 1997
Subba Rao, C.V., Ravi, M., Raja, V., Bhargav, P.B., Sharma, A.K. and Rao, V.V.R.N., Iran. Polym. J., 2012, 21: 531
Appetecchi, G.B., Scaccia, S. and Passerini, S., J. Electrochem. Soc., 2000, 147: 4448
Appetecchi, G.B., Croce, F., Hasson, J., Scrosati, B., Salomon, M. and Cassel, F., J. Power Sources, 2003, 114: 105
Chandra, A., Agrawal, R.C. and Mahipal, Y.K., J. Phys. D: Appl. Phys., 2009, 42: 135107
Chandra, A., Eur. Phys. J. Appl. Phys., 2010, 50: 21103
Chandra, A. and Chandra, A., “Hot-pressed solid polymer electrolytes: synthesis and characterization”, Lambert Academic Pub., Saarbrücken, Germany, 2010
Chandra, A., Chandra, A. and Thakur, K., Chinese J. Polym. Sci., 2013, 31(2): 302
Chandra, A., Chandra, A., Bhatt, A. and Thakur, K., Int. J. Chem., 2012, 1(2): 209
Wagner, J.B.Jr. and Wagner, C., J. Chem. Phys., 1957, 26: 1597
Reddy, M.J. and Chu, P.P., Electrochim. Acta, 2002, 47: 1189
Chandra, A., Chandra, A. and Thakur, S.S., Int. J. Chem. Res., 2012, 3: 81
Chu, P.P., Reddy, J.M. and Kao, H.M., Solid State Ionics, 2003, 156: 141
Miyamoto, T. and Shibayana, K., J. Appl. Phys., 1973, 44: 5372
Chetia, R., Maullick, M., Dutta, A. and Wass, N.N., Mat. Sci. Eng. B, 2004, 107: 134
Owens, B., Argue, G.R., Groce, J.J. and Hermo, L.D., J. Electrochem. Soc., 1969, 116: 312
Chandra, A., Chandra, A. and Thakur, K., Int. J. Appl. Phys., 2012, 2: 69
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chandra, A. PEO-PVP blended Na+ ion conducting solid polymeric membranes. Chin J Polym Sci 31, 1538–1545 (2013). https://doi.org/10.1007/s10118-013-1347-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-013-1347-z