Abstract
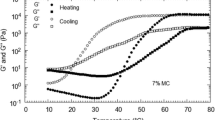
The sol-gel transition of methylcellulose (MC) solutions in the presence of ortho-methoxycinnamic acid (OMCA) or cetyltrimethylammonium bromide (CTAB) and in the coexistence of OMCA and CTAB was determined by the rheological measurement. It has been found that the sol-gel transition temperature of MC solutions increases linearly with the concentration of either OMCA or CTAB in solution, respectively. However, in the coexistence of OMCA and CTAB, the sol-gel transition temperature of MC solutions remains invariable, independent of the concentration of CTAB in solution. The experimental results show that OMCA has priority to adsorb on the methyl group of MC chains to form polymer-bound aggregates. In particular, these aggregates inhibit the hydrophobic interaction between CTAB and the methyl group of MC chains completely. Taking into account the fact that OMCA is almost insoluble in MC-free solutions but dissolves very well in aqueous MC solutions, we propose the formation of the core-shell architecture prompted by OMCA and the methyl group of MC chains, with the methyl group of MC chains serving as the core and the self-assembly of OMCA molecules serving as the shell. Obviously, the formation of the core-shell structure increases the solubility of OMCA, improves the stability of methyl groups of MC chains at high temperatures and inhibits the hydrophobic interaction between CTAB and the methyl group of MC chains in solution. The abnormal behavior relating to the sol-gel transition of MC solutions in the presence of OMCA or in the coexistence of OMCA and CTAB is therefore explained. Upon UV irradiation, the sol-gel transition temperature of MC solutions in the presence of OMCA, or in the coexistence of OMCA and CTAB, decreases notably. However, the dependence of the sol-gel transition temperature of MC solutions as a function of OMCA concentration, or CTAB concentration in the presence of OMCA, does not change after UV irradiation.
Similar content being viewed by others
References
Chakraborty, T., Chakraborty, I. and Ghosh, S., Langmuir, 2006, 22: 9905
Hoff, E., Nystrm, B. and Lindman, B., Langmuir, 2001, 17: 28
Panmai, S., Prud’homme, R.K., Peiffer, D.G., Jockusch, S. and Turro, N.J., Langmuir, 2002, 18: 3860
Lee Jr, C.T., Smith, K.A. and Hatton, T.A., Macromolecules, 2004, 37: 5397
Goddard, E., J. Colloid Interf. Sci., 2002, 256: 228
Dan, A., Ghosh, S. and Moulik, S.P., J. Phys. Chem. B, 2009, 113: 8505
Bu, H., Kjniksen, A.L., Knudsen, K.D. and Nystrm, B., Langmuir, 2005, 21: 10923
Dar, A.A., Garai, A., Das, A.R. and Ghosh, S., J. Phys. Chem. A, 2010, 114: 5083
Wang, Q.Q., Li, L., Liu, E., Xu, Y. and Liu, J., Polymer, 2006, 47: 1372
Li, L., Liu, E. and Lim, C.H., J. Phys. Chem. B, 2007, 111: 6410
Wang, Q.Q., Li, L., Chen, G. and Yang, Y., Carbohydr. Polym., 2007, 70: 345
Xu, Y., Wang, C., Tam, K. and Li, L., Langmuir, 2004, 20: 646
Kundu, P., Kundu, M., Sinha, M., Choe, S. and Chattopadhayay, D., Carbohydr. Polym., 2003, 51: 57
Xu, Y. and Li, L., Polymer, 2005, 46: 7410
Kundu, P. and Kundu, M., Polymer, 2001, 42: 2015
Xu, Y., Li, L., Zheng, P., Lam, Y.C. and Hu, X., Langmuir, 2004, 20: 6134
Li, L., Shan, H., Yue, C., Lam, Y., Tam, K. and Hu, X., Langmuir, 2002, 18: 7291
Mir, M.A., Dar, A.A., Amin, A. and Rather, G.M., J. Mol. Liq., 2009, 150: 86
Kjniksen, A.L., Nystrm, B. and Lindman, B., Langmuir, 1998, 14: 5039
Zhou, S., Yang, H.Y., Xie, Y.J., Li, H.Z. and Liu, G.M., Chin. J. Chem. Phys., 2011, 24: 489.
Davies, T.S., Ketner, A.M. and Raghavan, S.R., J. Am. Chem. Soc., 2006, 128: 6669
Ketner, A.M., Kumar, R., Davies, T.S., Elder, P.W. and Raghavan, S.R., J. Am. Chem. Soc., 2007, 129: 1553
Kumar, R. and Raghavan, S.R., Soft Matter, 2009, 5: 797
Shi, H.F., Wang, Y., Fang, B., Talmon, Y., Ge, W., Raghavan, S.R. and Zakin, J.L., Langmuir, 2011, 27: 5806
Kumar, R., Ketner, A.M. and Raghavan, S.R., Langmuir, 2010, 26: 5405
Sun, K., Kumar, R., Falvey, D.E., Raghavan, S.R., J. Am. Chem. Soc., 2009, 131: 7135
Lee, H.Y., Diehn, K., Sun, K., Chen, T. and Raghavan, S., J. Am. Chem. Soc., 2011, 133: 8461
Tomatsu, I., Peng, K. and Kros, A., Adv. Drug Deliver. Rev., 2011, 63:1257
Chambon, F. and Winter, H.H., Polym. Bull., 1985, 13: 499
Winter, H.H. and Chambon, F., J. Rheol., 1986, 30: 367
Villetti, M.A., Bica, C.I.D., Garcia, I.T.S., Pereira, F.V., Ziembowicz, F.I., Kloster, C. L. and Giacomelli, C., J. Phys. Chem. B, 2011, 115: 5868
Kodashima, H., Sakai, H., Abe, M., Ohkubo, T., Tsuchiya, K., Orihara, Y. and Matsumura, A., J. Am. Chem. Soc., 2005, 127: 13454
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tao, J., Yang, Hy., Hao, X. et al. Effect of ortho-methoxycinnamic acid on the sol-gel transition of methylcellulose solutions in the presence of cetyltrimethylammonium bromide. Chin J Polym Sci 31, 263–274 (2013). https://doi.org/10.1007/s10118-013-1197-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-013-1197-8