Abstract
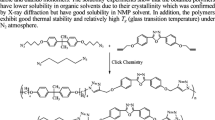
Thermally stable polymers containing 1,3,4-oxadiazole units have been synthesized through Huisgen reaction of the aromatic/aliphatic bis-tetrazole compounds with the aromatic/aliphatic bis-acid chlorides in pyridine as solvent. The obtained polymers are insoluble or slightly soluble even in polar aprotic solvents such as DMSO and DMF. Relatively high inherent viscosity values (0.61–1.33 dL/g, in 0.125% H2SO4 at 25°C) were observed for these compounds. Thermal analyses of the polymers using DSC and TGA techniques showed that the polymers have improved thermal stabilities. The glass transition temperature has not been observed in the fully aromatic polymers, but the polymers obtained from 5-[6-(1H-tetrazol-5-yl)hexyl]-1H-tetrazole (IV) showed very clear Tg. A model reaction was also investigated and the resulting bis-1,3,4-oxadiazole compound was characterized by conventional spectroscopy methods.
Similar content being viewed by others
References
Hsiao, S.H. and Chiou, J.H., J. Polym. Sci. Part A: Polym. Chem., 2001, 39: 2271
Souza, Jr.F.G., Sena, M.E. and Soares, B.G., J. Appl. Polym. Sci., 2004, 93: 1631
Kizhnyaev, V.N., Pokatilov, A.F., Vereshchagin, L.I., Adamova, L.V., Safronov, A.P. and Smirnov, A.I., Russ. J. Org. Chem., 2006, 79: 1167
Burroughes, J.H., Bradly, D.D.C. and Friend, R.H., Nature, 1990, 347: 539
Lee, D.W., Kwon, K.Y., Jin, J.I., Park, Y., Kim, Y.R. and Hwang, I.W., Chem. Mater., 2001, 13: 565
Janietz, S., Anlauf, S. and Wedel, A., Synth. Met., 2001, 122: 11
Kim, J.J., Kim, K.S., Baek, S., Kim, H.C. and Ree, M., J. Polym. Sci. Part A: Polym. Chem., 2002, 40: 1173
Zhang, Z., Li, G. and Shen, Y.M., J. Mater. Chem. Phys., 2003, 82: 613
Hsieh, B.Y., Yeh, K.M. and Chen, Y., J. Polym. Sci. Part A: Polym. Chem., 2005, 43: 5009
Morgado, J., Moons, E., Friend, R. and Gacialli, F., Adv. Mater., 2001, 13: 810
Deng, L., Furuta P.T., Garon, S., Li, J., Kavulak, D., Thompson, M.E. and Frechet, J.M., J. Chem. Mater., 2006, 18: 386
Dailey, S., Feast, W.J., Peace, R.J., Sage, I.C., Till, S. and Wood, E.L., J. Mater. Chem., 2001, 11: 2238
Wang, C., Kilitziraki, M., Palsson, L.O., Bryce, M. R., Monkman, A.P. and Samuel, I.D.W., Adv. Funct. Mater., 2001, 11: 47
Detert, H. and Sugiono, E., Synth. Met., 2001, 122: 19
Kim, J.H. and Lee, H., Synth. Met., 2004, 143: 13
Kim, J.H. and Lee, H., Synth. Met., 2004, 144: 169
Chen, Y., Huang, Y.Y. and Wu, T.Y., J. Polym. Sci. Part A: Polym. Chem., 2002, 40: 2927
Yang, N.C., Lee, C.I., Kim, J.K. and Suh, D.H., J. Appl. Polym. Sci., 2004, 92: 3112
Ding, J., Day, M., Robertson, G. and Roovers, J., Macromolecules, 2002, 35: 3474
Hamciuc, C., Hamciuc, E. and Bruma, M., Polymer, 2005, 46: 5851
Ma, W., Iyer, P.K., Gong, X., Liu, B., Moses, D., Bazan, G.C. and Heeger, A., J. Adv. Mater., 2005, 17: 274
Sava, I., Iosip, M.D., Bruma, M., Hamciuc, C., Robison, J., Okrasa, L. and Pakula, T., Eur. Polym. J., 39, 2003: 725
Jiang, X., Register, R.A., Pschenitzka, F., Sturm, J.C., Killeen, K.A. and Thompson, M.E., Mat. Res. Soc. Symp. Proc., 2000, 558: 433
Aldea, G., Vyprachticky, D. and Cimrova, V. Macromol. Symp., 2004, 212: 523
Mansoori, Y., Sarvari, R., Zamanloo, M.R. and Imanzadeh, G.H., Chinese J. Polym. Sci., 2010, 28(1): 21
Mansoori, Y., Sarvari, R., Zamanloo, M.R. and Imanzadeh, G.H., Russ. J. Org. Chem., 2009, 49: 154
Dabbagh, H.A. and Mansoori, Y., J. Chem. Res., 2000, (S): 442
Dabbagh, H.A. and Mansoori, Y., Russ. J. Org. Chem., 2001, 37: 1771
Dabbagh, H.A. and Mansoori, Y., Dyes Pigm., 2002, 54: 37
Gallardo, H., Magnago, R. and Bortoluzzi, A., J. Liq. Cryst., 2001, 28: 1343
Abshire, C.J. and Marvel, C.S., Die Makromol. Chem., 2003, 44: 388
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by the Research Council of the University of Mohaghegh Ardabili (Iran).
Rights and permissions
About this article
Cite this article
Mansoori, Y., Barghian, G., Koohi-Zargar, B. et al. Thermally stable polymers containing 1,3,4-oxadiazole units obtained from Huisgen reaction. Chin J Polym Sci 30, 36–44 (2012). https://doi.org/10.1007/s10118-012-1102-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-012-1102-x