Abstract
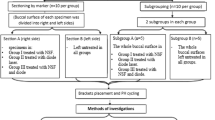
Altering the structure of the enamel surface around the orthodontic bracket by reducing its content of carbonate and phosphate resulting from application of CO2 laser may represent a more effective strategy in preventing caries in this region. This study aimed at determining whether irradiation with a CO2 laser combined with fluoride-releasing bonding material could reduce enamel demineralization around orthodontic brackets subjected to cariogenic challenge. Ninety bovine enamel slabs were divided into five groups (n = 18): non-inoculated brain–heart infusion broth group, non-fluoride-releasing composite resin (NFRCR—control group), resin-modified glass ionomer cement (RMGIC), CO2 laser + Transbond (L+NFRCR) and CO2 laser + Fuji (L+RMGIC). Slabs were submitted to a 5-day microbiological caries model. The Streptococcus mutans biofilm formed on the slabs was biochemically and microbiologically analysed, and the enamel Knoop hardness number (KHN) around the brackets was determined. The data were analysed by ANOVA and Tukey tests (α = 0.05). Biochemical and microbiological analyses of the biofilm revealed no statistically significant differences among the groups. Lased groups presented the highest KHN means, which statistically differed from NFRCR; however, no difference was found between these lased groups. RMGIC did not differ from NFRCR which presented the lowest KHN mean. The CO2 laser (λ = 10.6 μm; 10.0 J/cm2 per pulse) use with or without F-bonding materials was effective in inhibiting demineralization around orthodontic brackets. However, no additional effect was found when the enamel was treated with the combination of CO2 laser and an F-releasing material.

Similar content being viewed by others
References
Øgaard B (1989) Prevalence of white spot lesions in 19-year-olds: a study on untreated and orthodontically treated persons 5 years after treatment. Am J Orthod Dentofacial Orthop 96:423–427. doi:10.1016/0889-5406(89)90327-2
Pascotto RC, Navarro MF, Capelozza Filho L, Cury JA (2004) In vivo effect of a resin-modified glass ionomer cement on enamel demineralization around orthodontic brackets. Am J Orthod Dentofacial Orthop 125:36–41. doi:10.1016/S0889-5406(03)00571-7
Darling AI (1956) Studies of the early lesion of enamel caries with transmitted light, polarized light and radiography. Part II. Br Dent J 101:324–341. doi:10.1016/0002-9416(82)90548-6
Geiger AM, Gorelick L, Gwinnett AJ, Griswold PG (1988) The effect of a fluoride program on white spot formation during orthodontic treatment. Am J Orthod Dentofacial Orthop 93:29–37. doi:10.1016/0889-5406(88)90190-4
Marinelli CB, Donly KJ, Wefel JS, Jakobsen JR, Denehy GE (1997) An in vitro comparison of three fluoride regimens on enamel remineralization. Caries Res 31:418–422. doi:10.1159/000262432
Geiger AM, Gorelick L, Gwinnett AJ, Benson BJ (1992) Reducing white spot lesions in orthodontic populations with fluoride rinsing. Am J Orthod Dentofacial Orthop 101:403–407. doi:10.1016/0889-5406(92)70112-N
Gorton J, Featherstone JD (2003) In vivo inhibition of demineralization around orthodontic brackets. Am J Orthod Dentofacial Orthop 123:10–14. doi:10.1067/mod.2003.47
Fajen VB, Duncanson MG Jr, Nanda RS, Currier GF, Angolkar PV (1990) An in vitro evaluation of strength of three glass ionomer cements. Am J Orthod Dentofacial Orthop 97:316–322. doi:10.1016/0889-5406(90)70104-K
Hsu CY, Jordan TH, Dederich DN, Wefel JS (2000) Effects of low-energy CO2 laser irradiation and the organic matrix on inhibition of enamel demineralization. J Dent Res 79:1725–1730. doi:10.1177/00220345000790091401
Nelson DG, Shariati M, Glena R, Shields CP, Featherstone JD (1986) Effect of pulsed low energy infrared laser irradiation on artificial caries-like lesion formation. Caries Res 20:289–299. doi:10.1159/000260948
Nelson DG, Wefel JS, Jongebloed WL, Featherstone JD (1987) Morphology, histology and crystallography of human dental enamel treated with pulsed low-energy infrared laser radiation. Caries Res 21:411–426. doi:10.1159/000261047
Featherstone JD, Barrett-Vespone NA, Fried D, Kantorowitz Z, Seka W (1998) CO2 laser inhibitor of artificial caries-like progression in dental enamel. J Dent Res 77:1397–1403. doi:10.1177/00220345980770060401
Kantorowitz Z, Featherstone JD, Fried D (1998) Caries prevention by CO2 laser treatment: dependency on the number of pulses used. J Am Dent Assoc 129:585–591
Hsu CY, Jordan TH, Dederich DN, Wefel JS (2001) Laser-matrix-fluoride effects on enamel demineralization. J Dent Res 80:1797–1801. doi:10.1177/00220345010800090501
Klein ALL, Rodrigues LK, Eduardo CP, Nobre dos Santos M, Cury JA (2005) Caries inhibition around composite restorations by pulsed carbon dioxide laser application. Eur J Oral Sci 113:239–244. doi:10.1111/j.1600-0722.2005.00212.x
Rodrigues LK, Nobre Dos Santos M, Featherstone JD (2006) In situ mineral loss inhibition by CO2 laser and fluoride. J Dent Res 85:617–621. doi:10.1177/154405910608500707
Steiner-Oliveira C, Rodrigues LK, Soares LE, Martin AA, Zezell DM, Nobre-dos-Santos M (2006) Chemical, morphological and thermal effects of 10.6-microm CO2 laser on the inhibition of enamel demineralization. Dent Mater J 25:455–462
Gilmour AS, Edmunds DH, Newcombe RG (1997) Prevalence and depth of artificial caries-like lesions adjacent to cavities prepared in roots and restored with a glass ionomer or a dentin-bonded composite material. J Dent Res 76:1854–1861. doi:10.1177/00220345970760120801
Bolliger CT, Zellweger JP, Danielsson T, van Biljon X, Robidou A, Westin A, Perruchoud AP, Säwe U (2000) Smoking reduction with oral nicotine inhalers: double blind, randomised clinical trial of efficacy and safety. BMJ 321:329–333. doi:10.1136/bmj.321.7257.329
Rodrigues LK, Pinto TA, Ferrarini M, Rosalen PL, Cury JA, Nobre dos Santos M (2005) Validation of a dental sterilization dose using gamma radiation and its effect on enamel microhardness. Caries Res 39:331–332. doi:10.1159/000084836
Zanin IC, Lobo MM, Rodrigues LK, Pimenta LA, Höfling JF, Gonçalves RB (2006) Photosensitization of in vitro biofilms by toluidine blue O combined with a light-emitting diode. Eur J Oral Sci 114:64–69. doi:10.1111/j.1600-0722.2006.00263.x
Hayacibara MF, Rosa OP, Koo H, Torres SA, Costa B, Cury JA (2003) Effects of fluoride and aluminum from ionomeric materials on S. mutans biofilm. J Dent Res 82:267–271. doi:10.1177/154405910308200405
Rosalen PL, Pearson SK, Bowen WH (1996) Effect of copper co-crystallized with sugar on caries development in desalivated rats. Caries Res 30:367–372. doi:10.1159/000262344
Herigstad B, Hamilton M, Heersink J (2001) How to optimize the drop plate method for enumerating bacteria. J Microbiol Methods 44:121–129. doi:10.1016/S0167-7012(00)00241-4
Dubois M, Gilles K, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars of related substances. Anal Chem 28:350–356. doi:10.1021/ac60111a017
Vogel GL, Chow LC, Brown WE (1983) A microanalytical procedure for the determination of calcium, phosphate and fluoride in enamel biopsy samples. Caries Res 17:23–31. doi:10.1159/000260645
Fiske CH, Subbarow Y (1925) The colorimetric determination of phosphorus. J Biol Chem 66:375–400
Featherstone JD, Ten Cate JM, Shariati M, Arends J (1983) Comparison of artificial caries-like lesions by quantitative microradiography and microhardness profiles. Caries Res 17:385–391. doi:10.1159/000260692
Kielbassa AM, Wrbas KT, Schulte-Mönting J, Hellwing E (1999) Correlation of transversal microradiography and microhardness on in situ-induced demineralization in irradiated and nonirradiated human dental enamel. Arch Oral Biol 44:234–251. doi:10.1016/S0003-9969(98)00123-X
Sudjalim TR, Woods MG, Manton DJ (2006) Prevention of white spot lesions in orthodontic practice: a contemporary review. Aust Dent J 51:284–289
Hsu DJ, Darling CL, Lachica MM, Fried D (2008) Nondestructive assessment of the inhibition of enamel demineralization by CO2 laser treatment using polarization sensitive optical coherence tomography. J Biomed Opt 13:054027. doi:10.1117/1.2976113
Mellberg JR (1992) Hard-tissue substrates for evaluation of cariogenic and anti-cariogenic activity in situ. J Dent Res 71(Spec Iss):913–919. doi:10.1177/00220345920710041201
Esteves-Oliveira M, Zezell DM, Meister J, Franzen R, Stanzel S, Lampert F, Eduardo CP, Apel C (2009) CO2 laser (10.6 microm) parameters for caries prevention in dental enamel. Caries Res 43:261–268. doi:10.1159/000217858
Rodrigues LK, Nobre dos Santos M, Pereira D, Assaf AV, Pardi V (2004) Carbon dioxide laser in dental caries prevention. J Dent 32:531–540. doi:10.1016/j.jdent.2004.04.004
Carvalho AS, Cury JA (1999) Fluoride release from some dental materials in different solutions. Oper Dent 24:14–19
McNeill CJ, Wiltshire WA, Dawes C, Lavelle CL (2001) Fluoride release from new light-cured orthodontic bonding agents. Am J Orthod Dentofacial Orthop 120:392–397. doi:10.1067/mod.2001.118103
Lin YC, Lai YL, Chen WT, Lee SY (2008) Kinetics of fluoride release from and reuptake by orthodontic cements. Am J Orthod Dentofacial Orthop 133:427–434. doi:10.1016/j.ajodo.2006.01.052
Derks A, Katsaros C, Frencken JE, van’t Hof MA, Kuijpers-Jagtman AM (2004) Caries-inhibiting effect of preventive measures during orthodontic treatment with fixed appliances. A systematic review. Caries Res 38:413–420. doi:10.1159/000079621
Cohen WJ, Wiltshire WA, Dawes C, Lavelle CL (2003) Long-term in vitro fluoride release and rerelease from orthodontic bonding materials containing fluoride. Am J Orthod Dentofacial Orthop 124:571–576. doi:10.1016/S0889-5406(03)00573-0
van Dijken JW, Kalfas S, Litra V, Oliveby A (1997) Fluoride and mutans streptococci levels in plaque on aged restorations of resin-modified glass ionomer cement, compomer and resin composite. Caries Res 31:379–383. doi:10.1159/000262422
Montanaro L, Campoccia D, Rizzi S, Donati ME, Breshi L, Prati C, Arciola CR (2004) Evaluation of bacterial adhesion of Streptococcus mutans on dental restorative materials. Biomaterials 25:4457–4463. doi:10.1016/j.biomaterials.2003.11.031
Chin MY, Busscher HJ, Evans R, Noar J, Pratten J (2006) Early biofilm formation and the effects of antimicrobial agents on orthodontic bonding materials in a parallel plate flow chamber. Eur J Orthod 28:1–7. doi:10.1093/ejo/cji094
Loesche WJ (1986) Role of Streptococcus mutans in human dental decay. Microbiol Rev 50:353–380
Dibdin GH, Shellis RP (1988) Physical and biochemical studies of Streptococcus mutans sediments suggest new factors linking the cariogenicity of plaque with its extracellular polysaccharide content. J Dent Res 67:890–895. doi:10.1177/00220345880670060101
Ccahuana-Vásquez RA, Tabchoury CP, Tenuta LM, Del Bel Cury AA, Vale GC, Cury JA (2007) Effect of frequency of sucrose exposure on dental biofilm composition and enamel demineralization in the presence of fluoride. Caries Res 41:9–15. doi:10.1159/000096100
Acknowledgments
This study was supported by FAPESP (2008/02813-0) grant. The funder had no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript. This paper was based on a thesis submitted by the first author to the Piracicaba Dental School, University of Campinas, in partial fulfillment of the requirements for a MS degree in Dentistry (Pediatric Dentistry area). The authors thank the Biochemistry Laboratory of FOBUSP and LELO-FOUSP Laboratory for the use of their equipments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Souza-e-Silva, C.M., Parisotto, T.M., Steiner-Oliveira, C. et al. Carbon dioxide laser and bonding materials reduce enamel demineralization around orthodontic brackets. Lasers Med Sci 28, 111–118 (2013). https://doi.org/10.1007/s10103-012-1076-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-012-1076-5