Abstract
The nature of interactions of a toxic dye, thioflavin T (TFT), with pristine clay (bentonite (Bet)), clay minerals (montmorillonite (Mnt) and vermiculite (Vt)) and their organically modified forms (OMnt, OBet and OVt) has been investigated in aqueous media, and a methodology was developed for the utilization of TFT-treated colored residues as nanopigments, without producing the second-generation waste materials. The uptake of TFT was found to be more in case of pristine clay and clay minerals than their organically modified forms. In case of Mnt, TFT was present in the interlayer region, as supported by XRD data, and on the surface as the surface charge of TFT-treated pristine Mnt became almost zero, as indicated by the change in zeta potential value (− 17.6 to − 0.03 mV). In case of Bet and Vt, TFT was found to be only on clay mineral’s surface, as suggested by the change in zeta potential value (− 31 to − 0.6 mV in case of Bet and − 47 to − 0.04 mV in case of Vt). The corresponding increase in the particle size (158–443 nm in case of Bet and 145–460 nm in case of Vt) was also evocative of surface adsorption. In case of organically modified forms (OMnt, OBet and OVt), TFT was predominantly on the surface in all cases examined. The experimental data obtained during extractive removal of TFT using pristine and organically modified clays were best described with Langmuir adsorption isotherm model, whereas kinetic data are equipped with pseudo-second-order kinetic model. The intense yellow-colored solid material obtained after the extraction was used as colorant for the formation of polymer films with enhanced physico-chemical characteristics. These materials not only provide beautiful bright color but also prevent the accumulation of second-generation waste material.
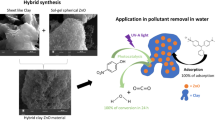
Graphical abstract












Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aragaw TA, Angerasa FT (2020) Synthesis and characterization of Ethiopian kaolin for the removal of basic yellow (BY 28) dye from aqueous solution as a potential adsorbent. Heliyon 19:e04975. https://doi.org/10.1016/j.heliyon.2020.e04975
Asgari G, Roshani G, Ghanizadeh G (2012) The investigation of kinetic and isotherm of fluoride adsorption onto functionalize pumic stone. J Hazrad Mater 17:123–132. https://doi.org/10.1016/j.jhazmat.2012.03.003
Collins JT, Gordon-Wiley SW, Horwitz C P (2000) Long-lived homogenous amide containing macrocyclic compounds. US Patent 6054580 A. https://patents.google.com/patent/US5847120A/en
Ehsani I, Turianicová E, Baláž M, Obut A (2014) Leaching of a commercial vermiculite in H2SO4 solutions. In: Proceedings of 14th international mineral processing symposium, Kusadasi Turkey, pp.757–763
Freundlic H (1906) Adsorption in solutions. Z Phys Chem (germany) 57:385–470
Gedam VV, Raut P, Chahande A, Pathak P (2019) Kinetic, thermodynamics and equilibrium studies on the removal of Congo red dye using activated teak leaf powder. Appl Water Sci 9:55. https://doi.org/10.1007/s13201-019-0933-9
Guo R, Wang R, Yin J, Jiao T, Huang H, Zhao X, Zhang L, Li Q, Zhou J, Peng Q (2019) Fabrication and Highly Efficient Dye Removal Characterization of Beta-Cyclodextrin-Based Composite Polymer Fibers by Electrospinning. Nanomaterials 9:127. https://doi.org/10.3390/nano9010127
Hackl EV, Darkwah J, Smith G, Ermolina I (2015) Effect on acidic and basic pH on Thioflavin T absorbance and fluorescence. Euro Biophys J 44:249–261. https://doi.org/10.1007/s00249-015-1019-8
Hameed BH, Ahmad AA, Aziz N (2007) Isotherm, kinetic, and thermodyanamics of acid dye adsorption on activated palm ash. Chem Eng J 133:195–203. https://doi.org/10.1016/j.cej.2007.01.032
He H, Frost RL, Bostrom T, Yuan P, Duong L, Yang D, Xi Y, Kloprogge JT (2007). Changes in the morphology of organoclays with HDTMA+ surfactant loading. Appl Clay Sci 31:262–271. https://doi.org/10.1016/j.clay.2005.10.011
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465. https://doi.org/10.1016/S0032-9592(98)(001)12-5
Joseph NT, Chinonye OE, Philomena IK, Christian AC, Elijah OC (2016) Isotherm and kinetic modeling of adsorption of dyestuff onto kola nut (Cola Acuminata) shell activated carbon. J Chem Tech Metall 51:188–201
Karpinska J, Kotowaska U (2019) Removal of organic pollution in the water environment. Water 11:1–8. https://doi.org/10.3390/w11102017
Kumari N, Datta M (2016) An integrated approch for the removal of indigo carmine dye from its aqueous solution and the synthesis of nano pigment, Internat J Envir Eco Family Urban Stud 6:7–16
Kumari N, Mohan C, Yadav S, Uniyal V, Taskaeva N (2022) Interaction of Indigo carmine with naturally occurring clay minerals: An approach for the synthesis of nanopigments. Mater Today: Proc 69:82-86.https://doi.org/10.1016/j.matpr.2022.08.081
Kurhunzenkau SA, Maskevich AA (2012) Computation of vibrational spectra and structure modeling for Thioflavin T molecule. J App Spectrosc 79:547–552. https://doi.org/10.1007/s10812-012-9638-7
Lagergren S (1898) About theory of so-called adsorption of soluble substances. K Sven Vetensk Akad Handl 24:1–39
Langmuir I (1916) The constitution and fundamental properties of solids and liquids. J Am Chem Soc 38:2221–2295. https://doi.org/10.1021/ja02268a002
Marchante VR, Gomis AM, Rico MIB (2013) Nanoclay-based pigments: synthesis, characterization and applications. Nano Prod Rev J 6 201304
Maruthapandi M, Kumar CB, Luong JHT, Gedanken A (2018) Kinetics, isotherm, and thermodynamic studies of methylene blue adsorption on polyaniline and polypyrrole macro–nanoparticles synthesized by c-dot-initiated polymerization. ACS Omega 3:7196–7203. https://doi.org/10.1021/acsomega.8b00478
Maskevich AA, Stsiapura VI, Kuzmitsky VA, Kuznetsova IM, Povarova OI, Uversky VN, Turoverov KK (2007) Spectral properties of Thioflavin T in solvent with different dielectricproperties and in fibril incorporated form. J Proteome Res 6:1392–1401. https://doi.org/10.1021/pr0605567
Mouzdahir EI, Elmchaouri A, Mahboub R, Gil A, Korili SA (2009) Synthesis of nano-layered vermiculite of low density by thermal treatment. Powder Tech 189:2–5. https://doi.org/10.1016/j.powtec.2008.06.013
Naushad M, Alqadami AA, Alothman ZA, Alsohaimi IH, Algamdi MS, Aldawsari AM (2019) Adsorption kinetics, isotherm and reusability studies for the removal of cationic dye from aqueous medium using arginine modified activated carbon. J Mol Liquid 293:111442. https://doi.org/10.1016/j.molliq.2019.111442
Neumann R, Costa GE, Gaspar JC, Palmieri M, Silva SE (2011) The mineral phase quantification of vermiculite and interstratified clay minerals-containing ores by X-ray diffraction and rietveld method after K cation exchange. Miner Engin 24:1323–1334. https://doi.org/10.1016/j.mineng.2011.05.017
Oladipo AA, Gazi M (2014) Enhanced removal of crystal violet by low cost alginate/acid activated Betonite composite beads: optimization and modelling using non-linear regression technique. J Water Proc Eng 2:43–52. https://doi.org/10.1016/j.jwpe.2014.04.007
Patel RK, Prasad R, Shankar R , Khare P, Yadav M (2021) Adsorptive removal of methylene blue dye from soapnut shell & pineapple waste derived activated carbon. Internat J Engin Sci Tech 13:81-87. https://doi.org/10.4314/ijest.v13i1.12S
Prindle A, Liu J, Asally M, Ly S, Ojalavo JG, Suel GM (2015) Ion channels enable electrical communication in bacterial communities. Nature 527:59–63. https://doi.org/10.1038/nature15709
Ray HH, Zhou Q, Frost RL, Wood BJ, Duong LV, Kloprogge JT (2007) A X-ray photoelectron spectroscopy study of HDTMAB distribution within organoclays. Spectrochim Acta Mol Bimol Spectrosc 66:1180–1188. https://doi.org/10.1016/j.saa.2006.06.005
Ren L, Zhao G, Pan L, Chen B, Chen Y, Zhang Q, Xiao X, Xu W (2021) Efficient removal of dye from wastewater without selectivity using activated carbon-juncus effusus porous fibril composites. ACS Appl Mater Interfaces 13:19176–19186. https://doi.org/10.1021/acsami.0c22104
Rung international (2008) Global dyestuff industry. Retrieved 12 July 2021, from http://www.foodcolorworld.com/global-dyestuff-industry.html
Shin SA (2007) In-situ polymerization and characterization of polyethylene–clay nanocomposite. University of Waterloo, Canada, Thesis
Tempkin MI, Pyzhev V (1940) Kinetic of ammonia synthesis on promoted iron catalyst. Acta Phys Chim USSR 12:327–356
Textile dyes industry is projected to achieve a global market size of US$ 8 Bn by 2031, currently US is the second largest exporter in the world. Retrieved on 20 March 2022. https://www.globenewswire.com/news-release/2022/02/16/2386373/0/en/Textile-Dyes-Industry-is-Projected-to-Achieve-a-Global-Market-Size-of-US-8-Bn-by-2031-Currently-US-is-the-Second-Largest-Exporter-in-the-World.html
Titchou FE, Zazou H, Afanga H, Gaayda EJ, Akbour RA, Nidheesh PV, Hamdani M (2021) Removal of organic pollutants from wastewater by advanced oxidation processes and its combination with membrane processes. Chem Eng Proc Proc 169:108631. https://doi.org/10.1016/j.cep.2021.108631
Ullah H, Nafees M, Iqbal F, Awan S, Shah A, Waseem A (2020) Adsorption kinetics of malachite green and methylene blue from aqueous solutions using surfactant-modified organoclays. Acta Chim Slov, 64. https://doi.org/10.17344/acsi.2017.3285
Weber WJ, Morris JC (1963) Kinetic of adsorption on carbon from solutions. J Sanit Eng Div Am Soc Civ Eng 89:31–60
Yahiaoui C, Kameche M, Innocent C, Khenifi A (2021) Conception of yeast microbial desalination cell: applications to dye wastewater treatment and lead removal. Chem Engin Comm 208:364-375, https://doi.org/10.1080/00986445.2020.1721479
Yakout SM, Elsherif E (2010) Batch kinetics, isotherm and thermodynamic studies of adsorption of strontium from aqueous solutions onto low cost rice-straw based carbons. Carbon Sci Tech 1:144–153
Zhao X, Baharinikoo L, Farahani MD, Farizhandi AAK (2021) Experimental modelling studies on the removal of dyes and heavy metal ions using ZnFe2O4 nanoparticles. Sci Rep 12:5987https://doi.org/10.1038/s41598-022-10036-y
Acknowledgement
We are thankful to the financial support provided for these studies by CSIR, New Delhi, and technical support from K. R. Mangalam University, Gurugram.
Funding
This work was supported by CSIR. Dr Neeraj Kumari has received research support from Council of Scientific and Industrial Research, India, as fellowship.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by NK and CM. Resourcing, investigation and data curation were done by RSV. The first draft of the manuscript was written by NK, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
Non-financial interests: Authors express their sincere thank to the Director, University Science Instrumentation Centre, University of Delhi, Delhi 110007, India, for providing instrumentation facilities. Authors are grateful to SMITA research laboratory, IIT Delhi, for providing zeta sizer facility. Authors also wish to express their sincere thanks to the Head, Sceince department, Delhi Technology University, Delhi, for providing the tensile strength tester facility.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohan, C., Varma, R.S. & Kumari, N. Novel approach for the synthesis of hybrid clay-based nanopigments and their application as coloring agents. Clean Techn Environ Policy 25, 1721–1735 (2023). https://doi.org/10.1007/s10098-023-02468-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-023-02468-5