Abstract
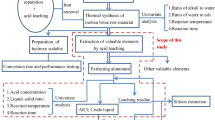
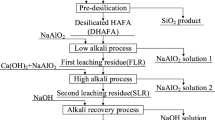
High-alumina coal fly ash (CFA) is considered as an important and cheap resource of aluminum. On account of high valence aluminum often trapped in aluminosilicate basic glassy phase and mullite (Al6Si2O13), efficiently separating aluminum and silicon is an essential precondition for maximizing the recycling of alumina from CFA, which is conducive to reducing the environment danger. In this study, a novel process combining alkali calcination and hydrochemical treatment was proposed to efficiently separate aluminum and silicon in CFA. The reactivity of CFA was greatly improved after the calcination treatment. The results show that alumina-rich phase, mullite, was transformed into nepheline (NaAlSiO4) by calcination with NaOH at a mass ratio 0.8:1 (wNaOH:wCFA) and calcination temperatures of 400 °C, which improved the dissolution of aluminum during the hydrochemical process and enriched the silicon in the residue. Then, the influence of the parameters of the hydrochemical process on alumina extraction was investigated. The results indicated that the two-stage method was more efficient than the direct hydrochemical process. The optimum conditions of the hydrochemical process were 40% NaOH solution and heating at 260 °C for 60 min. Approximately 94% of the alumina was easily recovered, whereas most of the silicon was converted to NaCaHSiO4. The combined treatment method highlights the potential for aluminum production from CFA.
Graphical abstract











Similar content being viewed by others
References
Bai GH, Qiao YH, Shen B (2011) Thermal decomposition of coal fly ash by concentrated sulfuric acid and alumina extraction process based on it. Fuel Process Technol 92(6):1213–1219. https://doi.org/10.1016/j.fuproc.2011.01.017
Blissett RS, Rowson NA (2012) A review of the multi-component utilisation of coal fly ash. Fuel 97:1–23. https://doi.org/10.1016/j.fuel.2012.03.024
Boycheva S, Zgureva D, Vassilev V (2013) Kinetic and thermodynamic studies on the thermal behaviour of fly ash from lignite coals. Fuel 108:639–646. https://doi.org/10.1016/j.fuel.2013.02.042
C, Sabolsky ME (2019) Microwave-assisted pretreatment of coal fly ash for enrichment and enhanced extraction of rare-earth elements. Energy Fuels, 33, 12083−12095. https://doi.org/10.1021/acs.energyfuels.9b02846
Ding J, Ma SH, Zheng SL (2016) Study of extracting alumina from high-alumina PC fly ash by a hydro-chemical process. Hydrometallurgy 161:58–64. https://doi.org/10.1016/j.hydromet.2016.01.025
Doucet FJ, Mohamed S, Neyt N (2016) Thermochemical processing of a South African ultrafine coal fly ash using ammonium sulphate as extracting agent for aluminium extraction. Hydrometallurgy 166:174–184. https://doi.org/10.1016/j.hydromet.2016.07.017
Fuller A, Omidiji Y, Viefhaus T (2019) The impact of an additive on fly ash formation/transformation from wood dust combustion in a lab-scale pulverized fuel reactor. Renew Energy 136:732–745 https://doi.org/10.1016/j.renene.2019.01.013
Gao XM, Dai Y, Zhang Y (2016) Effective Dye Removal from waste water using a Novel Low-Cost NaOH-Modified Fly Ash. Clays Clay Miner 64(6):695–705. https://doi.org/10.1346/CCMN.2016.064028
Gillian T, Canon RM, Egan BZ (1982) Economic metal recovery from fly ash. Resour Conserv 9:155–168. https://doi.org/10.1016/0166-3097(82)90071-2
Guo YX, Zhao ZS, Zhao Q, Cheng FQ (2017) Novel process of alumina extraction from coal fly ash by pre-desilicating—Na2CO3 activation—Acid leaching technique. Hydrometallurgy 169:418–425. https://doi.org/10.1016/j.hydromet.2017.02.021
Guo C, Zou J, Ma S, Yang J, Wang K (2019) Alumina extraction from coal fly ash via low-temperature potassium bisulfate calcination. Minerals 9:585. https://doi.org/10.3390/min9100585
Hamilton JP, Brantley SL (2001) Dissolution of nepheline, jadeite and albite glasses: toward better models for aluminosilicate dissolution. Geochim Cosmochim Acta 65(21):3683–3702. https://doi.org/10.1016/S0016-7037(01)00724-4
Han GH, Yang SZ, Peng WJ (2018) Enhanced recycling and utilization of mullite from coal fly ash with a flotation and metallurgy process. J Clean Prod 178:804–813. https://doi.org/10.1016/j.jclepro.2018.01.073
Hemalatha T, Ramaswamy A (2017) A review on fly ash characteristics e Towards promoting high volume utilization in developing sustainable concrete. J Clean Prod 2017(147):546–559. https://doi.org/10.1016/j.jclepro.2017.01.114
Hui KS, Chao CY (2006). Effects of step-change of synthesis temperature on synthesis of zeolite 4A from coal fly ash. Microporous and Mesoporous Materials, 88(1–3), 145–151. https://doi.org/10.1016/j.micromeso.2005.09.005.
Jiang ZQ, Yang J, Ma HW, Wang L, Ma X (2015) Reaction behaviour of Al2O3 and SiO2 in high alumina coal fly ash during alkali hydrothermal process. Trans Nonferrous Metal Soc China 25:2065–2072. https://doi.org/10.1016/S1003-6326(15)63816-X
Kanchan S, Kumar V, Yadav K (2015) Effect of fly ash disposal on ground water quality near Parichha thermal power plant, Jhansi: a case study. Current World Environ 2:572–580. https://doi.org/10.12944/cwe.10.2.21
Khan I, Umar R (2019) Environmental risk assessment of coal fly ash on soil and groundwater quality, Aligarh, India. Ground Water Sustain Dev 8:346–357. https://doi.org/10.1016/j.gsd.2018.12.002
Li S, Qin S, Kang L, Liu J, Wang J, Li Y (2017) An efficient approach for lithium and aluminum recovery from coal fly ash by pre-desilication and intensified acid leaching processes. Metals 7:272. https://doi.org/10.3390/met7070272
Li HQ, Hui JB, Wang CY (2014) Extraction of alumina from coal fly ash by mixed-alkaline hydrothermal method. Hydrometallurgy 147–148:183–187. https://doi.org/10.1016/j.hydromet.2014.05.012
Li Z, Zhang YF, Zhang Y (2009) Extraction of alumina and sodium oxide from red mud by a mild hydro-chemical process. J Hazardous Mater 172:1629–1634. https://doi.org/10.1016/j.jhazmat.2009.08.036
Liu W, Yang JK, Xiao B (2009) Review on treatment and utilization of bauxite residues in China. Int J Miner Process 93(3–4):220–231. https://doi.org/10.1016/j.minpro.2009.08.005
Liu CL, Ma SH, Zheng SL, Luo Y, Ding J, Wang XH, Zhang Y (2018) Combined treatment of red mud and coal fly ash by a hydro-chemical process. Hydrometallurgy 175:224–231. https://doi.org/10.1016/j.hydromet.2017.11.005
Liu CL, Ma SH, Ding J (2019) Kinetics of decomposition of mullite and corundum in coal fly ash under highly alkaline condition. Trans Nonferrous Metals Soc China 29(4):868–875. https://doi.org/10.1016/S1003-6326(19)64997-6
Matjie RH, Bunt JR, Heerden V (2005) Extraction of alumina from coal fly ash generated from a selected low rank bituminous South African coal. Miner Eng 18(3):299–310. https://doi.org/10.1016/j.mineng.2004.06.013
Nayak AK, Raja R, Rao KS (2015) Effect of fly ash application on soil microbial response and heavy metal accumulation in soil and rice plant. Ecotoxicol Environ Saf 114:257–262. https://doi.org/10.1016/j.ecoenv.2014.03.033
Sangita S, Nayak N, Panda CR (2017) Extraction of aluminium as aluminium sulphate from thermal power plant fly ashes. Trans Nonferrous Metals Soc China 27(9):2082–2089. https://doi.org/10.1016/S1003-6326(17)60231-0
Sen SK, Das MM, Bandyopadhyay P, Dash R (2016) Green process using hot spring bacterium to concentrate alumina in coal fly ash. Ecol Eng 88:10–19. https://doi.org/10.1016/j.ecoleng.2015.12.012
Shafiee S, Topal E (2009) When will fossil fuel reserves be diminished? Energy Policy 37:181–189. https://doi.org/10.1016/j.enpol.2008.08.016
Sun ZH, Li HQ, Bao WJ, Wang CY (2016) Mineral phase transition of desilicated high alumina fly ash with alumina extraction in mixed alkali solution. Int J Miner Process 153:109–117. https://doi.org/10.1016/j.minpro.2016.05.004
Toniolo N, Boccaccini AR (2017) Fly ash-based geopolymers containing added silicate waste. Rev Ceramics Int 43(17):14545–14551. https://doi.org/10.1016/j.ceramint.2017.07.221
Valeev D, Kunilova I, Shoppert A, Salazar-Concha C, Kondratiev A (2020) High-pressure HCl leaching of coal ash to extract Al into a chloride solution with further use as a coagulant for water treatment. J Clean Prod 276:123206. https://doi.org/10.1016/j.jclepro.2020.123206
Wang RC, Zhai YC, Wu XW (2014) Extraction of alumina from fly ash by ammonium hydrogen sulfate roasting technology. Trans Nonferrous Metals Soc China 24(5):1596–1603. https://doi.org/10.1016/S1003-6326(14)63230-1
Wei CD, Cheng S, Zhu FJ (2018) Digesting high-aluminum coal fly ash with concentrated sulfuric acid at high temperatures. Hydrometallurgy 180:41–48. https://doi.org/10.1016/j.hydromet.2018.07.004
Wu YS, Xu P, Chen J (2014) Effect of temperature on phase and Alumina extraction efficiency of the product from sintering coal fly ash with Ammonium Sulfate. Chin J Chem Eng 22(11–12):1363–1367. https://doi.org/10.1016/j.cjche.2014.09.008
Xing YW, Guo FY, Xu MD, Gui XH, Li HS, Li GH, Xia YC, Han HS (2019) Separation of unburned carbon from coal fly ash: a review. Powder Technol 353:372–384. https://doi.org/10.1016/j.powtec.2019.05.037
Yang QC, Ma SH, Zhang SL (2014) Recovery of alumina from circulating fluidized bed combustion Al-rich fly ash using mild hydrochemical process. Trans of Nonferrous Metals Soc China 24(4):1187–1195. https://doi.org/10.1016/S1003-6326(14)63178-2
Yao ZT, Xia MS, Sarker PK (2014) A review of the alumina recovery from coal fly ash, with a focus in China. Fuel 120:74–85. https://doi.org/10.1016/j.fuel.2013.12.003
You SW, Zhang YF, Chen FF (2014) Transformation of NaCaHSiO4 to sodalite and katoite in sodium aluminate solution. Hydrometallurgy 141:43–48. https://doi.org/10.1016/j.hydromet.2013.10.004
Yuan S, Jiang KX, Zhang TA (2020) Cleaner alumina production from coal fly ash: Membrane electrolysis designed for sulfuric acid leachate. J Clean Prod 234:118–470. https://doi.org/10.1016/j.jclepro.2019.11
Acknowledgements
This work was financially supported by the Longshan academic talent research and Innovation Team Project of SWUST (17LZXT11). We thank all the interviewees for sharing their ideas and experiences with us during our field studies and all the anonymous reviewers who help us to improve the paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, J., Sun, H., Peng, T. et al. Separation of alumina from aluminum-rich coal fly ash using NaOH molten salt calcination and hydrochemical process. Clean Techn Environ Policy 24, 1507–1519 (2022). https://doi.org/10.1007/s10098-021-02262-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-021-02262-1