Abstract
In this study, the steam reforming of naphthalene and pyrene as heavy tar model compounds was investigated experimentally in a horizontal tube reactor and theoretically using the CHEMKIN simulation. The experimental results revealed that the reactivity of naphthalene was higher than that of pyrene in the presence of steam. The carbon content converted to soot is a little more than that converted to light gas during tar steam reforming. The kinetic parameters of the overall reaction were determined, and the pre-exponential factor and activation energy were calculated using the experimental data. The comparison of the numerical simulation with the experimental findings exhibited an excellent agreement for the prediction of the light gas products and soot after eliminating the influence of the water–gas shift. Further, the reaction schemes including the reaction pathway and associated kinetics were determined for the steam reforming of these model compounds. Both naphthalene and pyrene exhibited a similar performance during the reaction in the presence of steam. Benzene and naphthalene, representing the precursors of the light gas product, were confirmed to be the dominant intermediate components of naphthalene and pyrene, respectively. The consecutive reactions of these intermediates subsequently resulted in the generation of the light gaseous products.
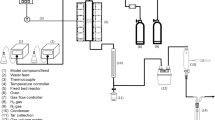
Graphic abstract











Similar content being viewed by others
References
Abu El-Rub Z, Bramer EA, Brem G (2008) Experimental comparison of biomass chars with other catalysts for tar reduction. Fuel 87:2243–2252
Anis S, Zainal Z (2011) Tar reduction in biomass producer gas via mechanical, catalytic and thermal methods: A review. Renew Sustain Energy Rev 15:2355–2377
Apicella B, Tregrossi A, Stazione F, Ciajolo A, Russo C (2017) Analysis of petroleum and coal tar pitches as large PAH Chemical. Eng Trans 57:775–780
Bridgwater A (1995) The technical and economic feasibility of biomass gasification for power generation. Fuel 74:631–653
Coll R, Salvado J, Farriol X, Montane D (2001) Steam reforming model compounds of biomass gasification tars: conversion at different operating conditions and tendency towards coke formation. Fuel Process Technol 74:19–31
Corella J, Sanz A (2005) Modeling circulating fluidized bed biomass gasifiers. A Pseudo-rigorous Model for Stationary State Fuel Process Technol 86:1021–1053
Devi L, Ptasinski KJ, Janssen FJ (2005) Decomposition of naphthalene as a biomass tar over pretreated olivine: effect of gas composition, kinetic approach, and reaction scheme. Ind Eng Chem Res 44:9096–9104
Fagbemi L, Khezami L, Capart R (2001) Pyrolysis products from different biomasses: application to the thermal cracking of tar. Appl Energy 69:293–306
Garcia XA, Hüttinger KJ (1989) Steam gasification of naphthalene as a model reaction of homogeneous gas/gas reactions during coal gasification. Fuel 68:1300–1310
Jess A (1996) Mechanisms and kinetics of thermal reactions of aromatic hydrocarbons from pyrolysis of solid fuels. Fuel 75:1441–1448
Li C, Suzuki K (2009) Tar property, analysis, reforming mechanism and model for biomass gasification—an overview. Renew Sustai Energy Rev 13:594–604
Li Q, Wang Q, Kayamori A, Zhang J (2018) Experimental study and modeling of heavy tar steam reforming. Fuel Process Technol 178:180–188
Milne TA, Evans RJ, Abatzaglou N (1998) Biomass Gasifier"Tars": Their Nature, Formation, and Conversion. National Renewable Energy Laboratory, Golden, CO (US)
Min Z, Yimsiri P, Zhang S, Wang Y, Asadullah M, Li C-Z (2013) Catalytic reforming of tar during gasification. Part III. Effects Feedstock on Tar Reforming Using Ilmenite as a Catalyst Fuel 103:950–955
Molino A, Chianese S, Musmarra D (2016) Biomass gasification technology: The state of the art overview. J Energy Chem 25:10–25
Norinaga K, Yatabe H, Matsuoka M, Hayashi J-i (2010) Application of an existing detailed chemical kinetic model to a practical system of hot coke oven gas reforming by noncatalytic partial oxidation. Ind Eng Chem Res 49:10565–10571
Norinaga K, Sakurai Y, Sato R, Hayashi J-i (2011) Numerical simulation of thermal conversion of aromatic hydrocarbons in the presence of hydrogen and steam using a detailed chemical kinetic model. Chem Eng J 178:282–290
Ren J, Liu Y-L, Zhao X-Y, Cao J-P (2020) Biomass thermochemical conversion: A review on tar elimination from biomass catalytic gasification. J Energy Inst 93:1083–1098
Richter H, Howard JB (2002) Formation and consumption of single-ring aromatic hydrocarbons and their precursors in premixed acetylene, ethylene and benzene flames. Phys Chem Chem Phys 4:2038–2055
Rios MLV, González AM, Lora EES, del Olmo OAA (2018) Reduction of tar generated during biomass gasification: A review. Biomass Bioenergy 108:345–370
Vivanpatarakij S, Rulerk D, Assabumrungrat S (2014) Removal of tar from biomass gasification process by steam reforming over nickel catalysts. Chem Eng Trans 37
Shahbaz M, Inayat A, Patrick DO, Ammar M (2017) The influence of catalysts in biomass steam gasification and catalytic potential of coal bottom ash in biomass steam gasification: a review. Renew Sustain Energy Rev 73:468–476
Siwal SS, Zhang Q, Sun C, Thakur S, Gupta VK, Thakur VK (2020) Energy production from steam gasification processes and parameters that contemplate in biomass gasifier–A review. Bioresource Technol 297:122481
Sutton D, Kelleher B, Ross JR (2001) Review of literature on catalysts for biomass gasification. Fuel Process Technol 73:155–173
Swierczynski D, Courson C, Kiennemann A (2008) Study of steam reforming of toluene used as model compound of tar produced by biomass gasification. Chem Eng Process 47:508–513
Wang H, Frenklach M (1994) Calculations of rate coefficients for the chemically activated reactions of acetylene with vinylic and aromatic radicals. J Phys Chem 98:11465–11489
Acknowledgements
The authors would like to express their gratitude to EditSprings (https://www.editsprings.com/) for the expert linguistic services provided.
Funding
This work is financially supported by the Key R&D Program of CHD (CHDKJ19-01-88) and IHI Corporation, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, W., Li, Q., Wang, Q. et al. Tar steam reforming during biomass gasification: kinetic model and reaction pathway. Clean Techn Environ Policy 24, 39–50 (2022). https://doi.org/10.1007/s10098-021-02062-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-021-02062-7