Abstract
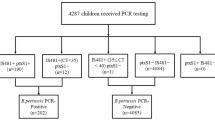
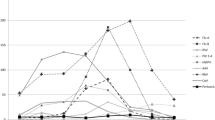
The purpose of this study is to identify predictive factors associated with missed diagnosis of B. pertussis–B. holmesii co-infection by assessing the analytical performance of a commercially available multiplexed PCR assay and by building a prediction model based on clinical signs and symptoms for detecting co-infections. This is a retrospective study on the electronic health records of all clinical samples that tested positive to either B. pertussis or B. holmesii from January 2015 to January 2018 at Geneva University Hospitals. Multivariate logistic regression was used to build a model for co-infection prediction based on the electronic health record chart review. Limit of detection was determined for all targets of the commercial multiplexed PCR assay used on respiratory samples. A regression model, developed from clinical symptoms and signs, predicted B. pertussis and B. holmesii co-infection with an accuracy of 82.9% (95% CI 67.9–92.8%, p value = .012), for respiratory samples positive with any of the two tested Bordetella species. We found that the LOD of the PCR reaction targeting ptxS1 is higher than that reported by the manufacturer by a factor 10. The current testing strategy misses B. pertussis and B. holmesii co-infections by reporting only B. holmesii infections. Thus, we advocate to perform serological testing for detecting a response against pertussis toxin whenever a sample is found positive for B. holmesii. These findings are important, both from a clinical and epidemiological point of view, as the former impacts the choice of antimicrobial drugs and the latter biases surveillance data, by underestimating B. pertussis infections during co-infections.


Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are not publicly available due to confidentiality issues but are available from the corresponding author on reasonable request.
References
Melvin JA, Scheller EV, Miller JF, Cotter PA (2014) Bordetella pertussis pathogenesis: current and future challenges. Nat Rev Microbiol 12(4):274–288
Guiso N (2009) Bordetella pertussis and pertussis vaccines. Clin Infect Dis 49(10):1565–1569
Bottero D, Griffith MM, Lara C, Flores D, Pianciola L, Gaillard ME et al (2013) Bordetella holmesii in children suspected of pertussis in Argentina. Epidemiol Infect 141(4):714–717
Yih WK, Silva EA, Ida J, Harrington N, Lett SM, George H (1999) Bordetella holmesii-like organisms isolated from Massachusetts patients with pertussis-like symptoms. Emerg Infect Dis 5(3):441–443
Mazengia E, Silva EA, Peppe JA, Timperi R, George H (2000) Recovery of Bordetella holmesii from patients with pertussis-like symptoms: use of pulsed-field gel electrophoresis to characterize circulating strains. J Clin Microbiol 38(6):2330–2333
Guthrie JL, Robertson AV, Tang P, Jamieson F, Drews SJ (2010) Novel duplex real-time PCR assay detects Bordetella holmesii in specimens from patients with pertussis-like symptoms in Ontario, Canada. J Clin Microbiol 48(4):1435–1437
Van den Bossche D, De Bel A, De Smet D, Heylen O, Vekens E, Vandoorslaer K et al (2013) Prevalence of Bordetella holmesii and Bordetella bronchiseptica in respiratory tract samples from Belgian patients with pertussis-like symptoms by sensitive culture method and mass spectrometry. Acta Clin Belg 68(5):341–348
Rodgers L, Martin SW, Cohn A, Budd J, Marcon M, Terranella A et al (2013) Epidemiologic and laboratory features of a large outbreak of pertussis-like illnesses associated with cocirculating Bordetella holmesii and Bordetella pertussis–Ohio, 2010–2011. Clin Infect Dis 56(3):322–331
Pittet LF, Emonet S, Schrenzel J, Siegrist CA, Posfay-Barbe KM (2014) Bordetella holmesii: an under-recognised Bordetella species. Lancet Infect Dis 14(6):510–519
Fong W, Timms V, Holmes N, Sintchenko V (2018) Detection and incidence of Bordetella holmesii in respiratory specimens from patients with pertussis-like symptoms in New South Wales, Australia. Pathology 50(3):322–326
Weyant RS, Hollis DG, Weaver RE, Amin MF, Steigerwalt AG, O’Connor SP et al (1995) Bordetella holmesii sp. nov., a new gram-negative species associated with septicemia. J Clin Microbiol 33(1):1–7
Fishbain JT, Riederer K, Sawaf H, Mody R (2015) Invasive Bordetella holmesii infections. Infect Dis (Lond) 47(2):65–68
Pittet LF, Posfay-Barbe KM (2016) Bordetella holmesii: still emerging and elusive 20 years on. Microbiol Spectr 4(2). https://doi.org/10.1128/microbiolspec.EI10-0003-2015
Humphrey JM, Lacaille SN, Patel K, Thompson E, Tulumba S, Healey JH et al (2015) Prosthetic-joint-associated Bordetella holmesii infection. Open Forum Infect Dis 2(4):ofv169
Tang YW, Hopkins MK, Kolbert CP, Hartley PA, Severance PJ, Persing DH (1998) Bordetella holmesii-like organisms associated with septicemia, endocarditis, and respiratory failure. Clin Infect Dis 26(2):389–392
Russell FM, Davis JM, Whipp MJ, Janssen PH, Ward PB, Vyas JR et al (2001) Severe Bordetella holmesii infection in a previously healthy adolescent confirmed by gene sequence analysis. Clin Infect Dis 33(1):129–130
Dorbecker C, Licht C, Korber F, Plum G, Haefs C, Hoppe B et al (2007) Community-acquired pneumonia due to Bordetella holmesii in a patient with frequently relapsing nephrotic syndrome. J Infect 54(4):e203–e205
Nei T, Hyodo H, Sonobe K, Dan K, Saito R (2012) First report of infectious pericarditis due to Bordetella holmesii in an adult patient with malignant lymphoma. J Clin Microbiol 50(5):1815–1817
Pittet LF, Emonet S, Francois P, Bonetti EJ, Schrenzel J, Hug M et al (2014) Diagnosis of whooping cough in Switzerland: differentiating Bordetella pertussis from Bordetella holmesii by polymerase chain reaction. PLoS ONE 9(2):e88936
Njamkepo E, Bonacorsi S, Debruyne M, Gibaud SA, Guillot S, Guiso N (2011) Significant finding of Bordetella holmesii DNA in nasopharyngeal samples from French patients with suspected pertussis. J Clin Microbiol 49(12):4347–4348
Antila M, He Q, de Jong C, Aarts I, Verbakel H, Bruisten S et al (2006) Bordetella holmesii DNA is not detected in nasopharyngeal swabs from Finnish and Dutch patients with suspected pertussis. J Med Microbiol 55(Pt 8):1043–1051
Tatti KM, Sparks KN, Boney KO, Tondella ML (2011) Novel multitarget real-time PCR assay for rapid detection of Bordetella species in clinical specimens. J Clin Microbiol 49(12):4059–4066
Glare EM, Paton JC, Premier RR, Lawrence AJ, Nisbet IT (1990) Analysis of a repetitive DNA sequence from Bordetella pertussis and its application to the diagnosis of pertussis using the polymerase chain reaction. J Clin Microbiol 28(9):1982–1987
Harvill ET, Goodfield LL, Ivanov Y, Smallridge WE, Meyer JA, Cassiday PK et al (2014) Genome sequences of nine Bordetella holmesii strains isolated in the United States. Genome Announc 2(3):e00438-14. https://doi.org/10.1128/genomeA.00438-14
Templeton KE, Scheltinga SA, van der Zee A, Diederen BM, van Kruijssen A, Goossens H et al (2003) Evaluation of real-time PCR for detection of and discrimination between Bordetella pertussis, Bordetella parapertussis, and Bordetella holmesii for clinical diagnosis. J Clin Microbiol 41(9):4121–4126
Valero-Rello A, Henares D, Acosta L, Jane M, Jordan I, Godoy P et al (2019) Validation and implementation of a diagnostic algorithm for DNA detection of Bordetella pertussis, B. parapertussis, and B. holmesii in a pediatric referral hospital in Barcelona, Spain. J Clin Microbiol 57(1):e01231-18. https://doi.org/10.1128/JCM.01231-18
BioGX. Bordetella Speciation Plus Toxin OSR for BD MAXTM. BioGX, The Netherlands. 27 August 2021. https://www.biogx.com/download/76/400-002-c-max/12065/400-002-c-max-bordetella-speciation-rev09-27aug2021.pdf
Bouchez V, Brun D, Cantinelli T, Dore G, Njamkepo E, Guiso N (2009) First report and detailed characterization of B. pertussis isolates not expressing pertussis toxin or pertactin. Vaccine 27(43):6034–41
Scanlon K, Skerry C, Carbonetti N (2019) Association of pertussis toxin with severe pertussis disease. Toxins (Basel) 11(7):373. https://doi.org/10.3390/toxins11070373
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP et al (2007) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ 335(7624):806–808
Eberhardt CS, Blanchard-Rohner G, Lemaitre B, Combescure C, Othenin-Girard V, Chilin A et al (2017) Pertussis antibody transfer to preterm neonates after second- versus third-trimester maternal immunization. Clin Infect Dis 64(8):1129–1132
Miranda C, Porte L, Garcia P (2012) Bordetella holmesii in nasopharyngeal samples from Chilean patients with suspected Bordetella pertussis infection. J Clin Microbiol 50(4):1505 (author reply 6)
Mir-Cros A, Codina G, Martin-Gomez MT, Fabrega A, Martinez X, Jane M et al (2017) Emergence of Bordetella holmesii as a causative agent of whooping cough, Barcelona. Spain Emerg Infect Dis 23(11):1856–1859
Lotfi MN, Nikbin VS, Nasiri O, Badmasti F, Shahcheraghi F (2017) Molecular detection of Bordetella holmesii in two infants with pertussis-like syndrome: the first report from Iran. Iran J Microbiol 9(4):219–223
Shepard CW, Daneshvar MI, Kaiser RM, Ashford DA, Lonsway D, Patel JB et al (2004) Bordetella holmesii bacteremia: a newly recognized clinical entity among asplenic patients. Clin Infect Dis 38(6):799–804
Gordon KA, Fusco J, Biedenbach DJ, Pfaller MA, Jones RN (2001) Antimicrobial susceptibility testing of clinical isolates of Bordetella pertussis from northern California: report from the SENTRY Antimicrobial Surveillance Program. Antimicrob Agents Chemother 45(12):3599–3600
Ohtsuka M, Kikuchi K, Shimizu K, Takahashi N, Ono Y, Sasaki T et al (2009) Emergence of quinolone-resistant Bordetella pertussis in Japan. Antimicrob Agents Chemother 53(7):3147–3149
Office fédéral de la santé publique (2021) OFSP-Bulletin 47/2021. OFSP, Berne. 22 November 2021. https://www.bag.admin.ch/dam/bag/fr/dokumente/cc/Kampagnen/Bulletin/2021/bu-47-21.pdf.download.pdf/BU_47_21_FR.pdf (French)
Acknowledgements
The authors would like to acknowledge Pre Claire-Anne Siegrist and the Laboratory of Vaccinology of the Geneva University Hospitals for their help in diagnosing the case of Bordetella co-infection that led to our study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by Yannick Charretier, Catherine Hafner, Sarah Rosset-Zufferey, Barbara Lemaitre, Gesuele Renzi, Mikaël de Lorenzi-Tognon, and Jacques Schrenzel. Data analyses were performed by Mikaël de Lorenzi-Tognon. The first draft of the manuscript was written by Mikaël de Lorenzi-Tognon and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The project was approved by the Geneva Ethical Committee and registered as study n°2020–02072 and a consent was waived due to the retrospective aspect of the study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Lorenzi-Tognon, M., Charretier, Y., Iten, A. et al. Missed pertussis diagnosis during co-infection with Bordetella holmesii. Eur J Clin Microbiol Infect Dis 41, 1227–1235 (2022). https://doi.org/10.1007/s10096-022-04488-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-022-04488-3