Abstract
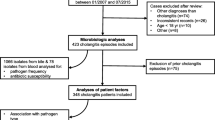
With rising antibiotic resistance, alternatives to carbapenems are needed for acute cholangitis (AC). Temocillin reaches high biliary concentrations with limited impact on microbiota. We retrospectively included 140 AC episodes and assessed the efficacy of temocillin using microbiology susceptibility testing from blood cultures. Considering all bacteria collected by episode, resistance to temocillin, PIP/TAZ and 3GC occurred in 27/140 (26%), 32 (22.8%) and 31 (22%) episodes, respectively (p = 0.7). After documentation, temocillin could have spared PIP/TAZ or carbapenems in 14/26 and 4/11 episodes. Temocillin may constitute an alternative treatment after microbiological documentation by sparing carbapenems and/or PIP/TAZ, but not as an empirical therapeutic option.

Similar content being viewed by others
Data availability
All data is available upon request.
References
Yokoe M, Hata J, Takada T, Strasberg SM, Asbun HJ, Wakabayashi G et al (2018) Tokyo Guidelines 2018: diagnostic criteria and severity grading of acute cholecystitis (with videos). J. Hepato-Biliary-Pancreat. Sci. 25:41–54. https://doi.org/10.1002/jhbp.515
Tagashira Y, Sakamoto N, Isogai T, Hikone M, Kosaka A, Chino R et al (2017) Impact of inadequate initial antimicrobial therapy on mortality in patients with bacteraemic cholangitis: a retrospective cohort study. Clin Microbiol Infect Off. Publ Eur Soc Clin Microbiol Infect Dis 23:740–747. https://doi.org/10.1016/j.cmi.2017.02.027
Sokal A, Sauvanet A, Fantin B, de Lastours V (2019) Acute cholangitis: diagnosis and management. J Visc Surg 156:515–525. https://doi.org/10.1016/j.jviscsurg.2019.05.007
Gomi H, Takada T, Hwang T-L, Akazawa K, Mori R, Endo I et al (2017) Updated comprehensive epidemiology, microbiology, and outcomes among patients with acute cholangitis. J Hepato-Biliary-Pancreat Sci 24:310–318. https://doi.org/10.1002/jhbp.452
Gomi H, Solomkin JS, Takada T, Strasberg SM, Pitt HA, Yoshida M et al (2013) TG13 antimicrobial therapy for acute cholangitis and cholecystitis. J. Hepato-Biliary-Pancreat. Sci. 20:60–70. https://doi.org/10.1007/s00534-012-0572-0
Solomkin JS, Mazuski JE, Bradley JS, Rodvold KA, Goldstein EJC, Baron EJ et al (2010) Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Clin Infect Dis Off Publ Infect Dis Soc Am 15(50):133–164. https://doi.org/10.1086/649554
WHO. WHO. Global action plan on antimicrobial resistance [Internet]. 2015;doi: http://apps.who.int/medicinedocs/documents/s21889en/s21889en.pdf
Rodríguez-Baño J, Gutiérrez-Gutiérrez B, Machuca I, Pascual A (2018) Treatment of infections caused by extended-spectrum-beta-lactamase-, AmpC-, and carbapenemase-producing Enterobacteriaceae. Clin Microbiol Rev 31. https://doi.org/10.1128/CMR.00079-17
Cassini A, Högberg LD, Plachouras D, Quattrocchi A, Hoxha A, Simonsen GS et al (2019) Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: a population-level modelling analysis. Lancet Infect Dis 19:56–66. https://doi.org/10.1016/S1473-3099(18)30605-4
Maudgal DP, Lanzini A, Northfield TC, Bridges C, Joseph AE (1985) Quantification of temocillin biliary excretion and gallbladder bile concentration in healthy subjects. Drugs 29(Suppl 5):146–150. https://doi.org/10.2165/00003495-198500295-00030
Poston GJ, Greengrass A, Moryson CJ (1985) Biliary concentrations of temocillin. Drugs 29(Suppl 5):140–145. https://doi.org/10.2165/00003495-198500295-00029
Livermore DM, Tulkens PM (2009) Temocillin revived. J Antimicrob Chemother 63:243–245. https://doi.org/10.1093/jac/dkn511
Alexandre K, Fantin B (2018) Pharmacokinetics and pharmacodynamics of temocillin. Clin Pharmacokinet 57:287–296. https://doi.org/10.1007/s40262-017-0584-7
Balakrishnan I, Koumaki V, Tsakris A (2019) Temocillin: is this the right momentum for its global use? Future Microbiol 14:81–83. https://doi.org/10.2217/fmb-2018-0316
European Committee on Antimicrobial Susceptibility Testing (EUCAST). Breakpoint tables for interpretation of MICs and zone diameters. www.eucast.org 2013;
EUCAST_General_Consultation_on_temocillin_breakpoints_20191011.pdf [Internet]. [cited 2020 7];doi: http://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Consultation/2019/EUCAST_General_Consultation_on_temocillin_breakpoints_20191011.pdf
Woerther P-L, Lepeule R, Burdet C, Decousser J-W, Ruppé É, Barbier F (2018) Carbapenems and alternative β-lactams for the treatment of infections due to extended-spectrum β-lactamase-producing Enterobacteriaceae: what impact on intestinal colonisation resistance? Int. J Antimicrob Agents 52:762–770. https://doi.org/10.1016/j.ijantimicag.2018.08.026
Alexandre K, Chau F, Guérin F, Massias L, Lefort A, Cattoir V et al (2016) Activity of temocillin in a lethal murine model of infection of intra-abdominal origin due to KPC-producing Escherichia coli. J Antimicrob Chemother 71:1899–1904. https://doi.org/10.1093/jac/dkw066
Alexandre K, Soares A, Chau F, Fantin B, Caron F, Etienne M. Temocillin breakpoints in pyelonephritis: evaluation in a murine model due to ESBL-producing Escherichia coli clinical isolates. J Antimicrob Chemother. 2019 1;74:1323–6. https://doi.org/10.1093/jac/dky569
Laterre P-F, Wittebole X, Van de Velde S, Muller AE, Mouton JW, Carryn S et al (2015) Temocillin (6 g daily) in critically ill patients: continuous infusion versus three times daily administration. J Antimicrob Chemother 70:891–898. https://doi.org/10.1093/jac/dku465
Balakrishnan I, Awad-El-Kariem FM, Aali A, Kumari P, Mulla R, Tan B et al (2011) Temocillin use in England: clinical and microbiological efficacies in infections caused by extended-spectrum and/or derepressed AmpC β-lactamase-producing Enterobacteriaceae. J Antimicrob Chemother 66:2628–2631. https://doi.org/10.1093/jac/dkr317
Acknowledgements
This work was presented in part as a poster at the European Congress of Clinical Microbiology and Infectious Diseases in Amsterdam in April 2019 (Abstract #P0840).
Author information
Authors and Affiliations
Contributions
MHNC, BF and VDL designed the study. SC and AS collected clinical data. MD, AH and VLG performed the microbiology studies. SC, BF and VDL performed the analysis and wrote the paper.
Corresponding author
Ethics declarations
Ethical approval
Data collection for the cohort was declared to the French “Commission Nationale Informatique et des Libertés” (CNIL), registration no. 2080377 on 4/4/2017. Under French law, approval by an Institutional Review Board is not mandatory for retrospective monocentric studies.
Consent to participate
All patients were informed of the study. No specific consent was necessary due to the retrospective nature of the work, according to French law.
Consent to publish
All authors have read the paper and gave their consent for publication.
Competing interests
Victoire de Lastours, Véronique Leflon and Bruno Fantin report non-financial support from EUMEDICA (travel expenses). Bruno Fantin reports financial support for participating on a board organised by EUMEDICA on clinical development of temocillin. All other authors declare no conflict of interest concerning this publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chawki, S., Sokal, A., Duprilot, M. et al. Temocillin as an alternative treatment for acute bacterial cholangitis: a retrospective microbiology susceptibility-based study of 140 episodes. Eur J Clin Microbiol Infect Dis 40, 1773–1777 (2021). https://doi.org/10.1007/s10096-021-04158-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-021-04158-w