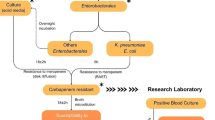
Abstract
The urgent need for rapid antimicrobial susceptibility is broadly apparent from government reports to the lay press. Accordingly, we developed a flow-cytometry assay (FCM) for evaluating ceftolozane-tazobactam (C/T) susceptibility directly on blood cultures (BC) requiring < 2 h from flag positivity to report. The protocol was optimized with C/T-susceptible and C/T-resistant gram-negative bacilli inoculated in BC aerobic bottles (Becton-Dickinson, USA), and afterward optimized for different C/T concentrations (1/4, 2/4, 4/4, and 8/4 mg/L) for 1 h incubation (37 °C), followed by FCM and software analysis. Fluorescent membrane permeability and membrane potential dyes were comparatively used to detect early cell lesions using the CytoFLEX cytometer (Beckman-Coulter, USA). Repeatability, reproducibility, and stability of the assay up to 48 h after BC positivity were determined. Internal validation was performed in spiked BC bottles with 130 Enterobacterales and 32 Pseudomonas aeruginosa isolates from Porto University (Portugal), including 13 ATCC isolates. Additionally, 64 gram-negative bacilli recovered from positive BC at Ramon y Cajal Hospital (Madrid, Spain) were tested. Categorical agreement (CA) and analytical errors were calculated comparing FCM with broth microdilution results. Only the membrane potential dyes clearly distinguished CT-susceptible and CT-resistant isolates. Excellent repeatability, reproducibility, and inter-method concordance was observed. Overall, CA was 99.1% using EUCAST criteria with 2 major errors and 98.7% with CLSI criteria with 2 major and 1 minor errors. A new, accurate, and ultra-rapid FCM (< 2 h) for testing C/T susceptibility gave accurate results and would expand current FCM antimicrobial susceptibility assay.



Similar content being viewed by others
References
Sorbera M, Chung E, Ho CW, Marzella N (2014) Ceftolozane/tazobactam: a new option in the treatment of complicated gram-negative infections. P & T : a peer-reviewed journal for formulary management 39(12):825–832
Organization WH (2016) World antibiotic awareness week 14-20 November WHO, WHO
Zhanel GG, Chung P, Adam H, Zelenitsky S, Denisuik A, Schweizer F, Lagace-Wiens PR, Rubinstein E, Gin AS, Walkty A, Hoban DJ, Lynch JP 3rd, Karlowsky JA (2014) Ceftolozane/tazobactam: a novel cephalosporin/beta-lactamase inhibitor combination with activity against multidrug-resistant gram-negative bacilli. Drugs 74(1):31–51. https://doi.org/10.1007/s40265-013-0168-2
Shortridge D, Pfaller MA, Streit JM, Flamm RK (2019) Antimicrobial activity of ceftolozane/tazobactam tested against contemporary (2015-2017) Pseudomonas aeruginosa isolates from a global surveillance programme. Journal of global antimicrobial resistance 21:60–64. https://doi.org/10.1016/j.jgar.2019.10.009
Garcia-Fernandez S, Garcia-Castillo M, Melo-Cristino J, Pinto MF, Goncalves E, Alves V, Vieira AR, Ramalheira E, Sancho L, Diogo J, Ferreira R, Silva D, Chaves C, Passaro L, Paixao L, Canton R (2020) In vitro activity of ceftolozane-tazobactam against Enterobacterales and Pseudomonas aeruginosa causing urinary, intra-abdominal and lower respiratory tract infections in intensive care units in Portugal: the STEP multicenter study. International journal of antimicrobial agents 55(3):105887. https://doi.org/10.1016/j.ijantimicag.2020.105887
Garcia-Fernandez S, Garcia-Castillo M, Bou G, Calvo J, Cercenado E, Delgado M, Pitart C, Mulet X, Tormo N, Mendoza DL, Diaz-Reganon J, Canton R (2019) Activity of ceftolozane/tazobactam against Pseudomonas aeruginosa and Enterobacterales isolates recovered from intensive care unit patients in Spain: the SUPERIOR multicentre study. International journal of antimicrobial agents 53(5):682–688. https://doi.org/10.1016/j.ijantimicag.2019.02.004
Siddiqui S, Razzak J (2010) Early versus late pre-intensive care unit admission broad spectrum antibiotics for severe sepsis in adults. The Cochrane database of systematic reviews (10):Cd007081. https://doi.org/10.1002/14651858.CD007081.pub2
Dubourg G, Lamy B, Ruimy R (2018) Rapid phenotypic methods to improve the diagnosis of bacterial bloodstream infections: meeting the challenge to reduce the time to result. Clinical Microbiology and Infection 24(9):935–943. https://doi.org/10.1016/j.cmi.2018.03.031
van Belkum A, Dunne WM Jr (2013) Next-generation antimicrobial susceptibility testing. Journal of clinical microbiology 51(7):2018–2024. https://doi.org/10.1128/jcm.00313-13
Costa-de-Oliveira S, Teixeira-Santos R, Silva AP, Pinho E, Mergulhao P, Silva-Dias A, Marques N, Martins-Oliveira I, Rodrigues AG, Paiva JA, Canton R, Pina-Vaz C (2017) Potential impact of flow cytometry antimicrobial susceptibility testing on the clinical management of gram-negative bacteremia using the FASTinov((R)) kit. Frontiers in microbiology 8:2455. https://doi.org/10.3389/fmicb.2017.02455
Gucker FT Jr, O'Konski CT et al (1947) A photoelectronic counter for colloidal particles. Journal of the American Chemical Society 69(10):2422–2431. https://doi.org/10.1021/ja01202a053
Alvarez-Barrientos A, Arroyo J, Canton R, Nombela C, Sanchez-Perez M (2000) Applications of flow cytometry to clinical microbiology. Clinical microbiology reviews 13(2):167–195. https://doi.org/10.1128/cmr.13.2.167-195.2000
Cidália Pina-Vaz, Sofia Costa-de-Oliveira, Ana Silva Dias, Ana Pinto Silva, Rita Teixeira Santos (2017) Flow cytometry in microbiology: the reason and the need. In: Cossarizza JPRaA (ed) Single cell analysis – contemporary research and clinical applications. 1st edn edn. Springer Nature Singapore Pte Ltd, Singapore, pp 153-169
Espinar MJ, Rocha R, Ribeiro M, Goncalves Rodrigues A, Pina-Vaz C (2011) Extended-spectrum beta-lactamases of Escherichia coli and Klebsiella pneumoniae screened by the VITEK 2 system. Journal of medical microbiology 60(Pt 6):756–760. https://doi.org/10.1099/jmm.0.024075-0
Fonseca ESD, Silva-Dias A, Gomes R, Martins-Oliveira I, Ramos MH, Rodrigues AG, Canton R, Pina-Vaz C (2019) Evaluation of rapid colistin susceptibility directly from positive blood cultures using a flow cytometry assay. International journal of antimicrobial agents. https://doi.org/10.1016/j.ijantimicag.2019.08.016
Pina Vaz C, Rodrigues A, Faria Ramos IC, Costa De Oliveira AS, Pinto E Silva AT, Silva Dias A, Teixeira Dos Santos R (2016) Fluorescent method of detecting microorganisms resistant to a therapeutic agent United States Patent US 9,290,790 B2
Pina Vaz C, Rodrigues A, Faria Ramos IC, Rocha Do Rosário R, Costa De Oliveira AS, Pinto E Silva AT (2017) Method of detecting the resistant microorganisms to a therapeutic agent. European Patent EP 2 714 922 B1
Puttaswamy S, Doo Lee B, Sengupta S (2011) Novel electrical method for early detection of viable bacteria in blood cultures, vol 49. https://doi.org/10.1128/JCM.00369-11
Testing ECoAS (2019) European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters Version 9.0, valid from 2019-01-01 edn
Institute CaLS (2019) M100 performance standards for antimicrobial susceptibility testing 29th edn
Systems CLTaIVDT (2019) Susceptibility testing of infection agents and evaluation of performance of antimicrobial susceptibility test devices part 1: reference method for testing the in vitro activity of antimicrobial agents against rapidly growing aerobic bacteria involved in infectious diseases vol ISO/FDIS 20776-1:2019
Lopez-Pintor JM, Navarro-San Francisco C, Sanchez-Lopez J, Garcia-Caballero A, Loza Fernandez de Bobadilla E, Morosini MI, Canton R (2019) Direct antimicrobial susceptibility testing from the blood culture pellet obtained for MALDI-TOF identification of Enterobacterales and Pseudomonas aeruginosa. European journal of clinical microbiology & infectious diseases : official publication of the European Society of Clinical Microbiology 38(6):1095–1104. https://doi.org/10.1007/s10096-019-03498-y
Marschal M, Bachmaier J, Autenrieth I, Oberhettinger P, Willmann M, Peter S (2017) Evaluation of the Accelerate Pheno system for fast identification and antimicrobial susceptibility testing from positive blood cultures in bloodstream infections caused by gram-negative pathogens. Journal of clinical microbiology 55(7):2116–2126. https://doi.org/10.1128/jcm.00181-17
Oviaño M, Gómara M, Barba MJ, Revillo MJ, Barbeyto LP, Bou G (2017) Towards the early detection of β-lactamase-producing Enterobacteriaceae by MALDI-TOF MS analysis. Journal of Antimicrobial Chemotherapy 72 (8):2259-2262. 10.1093/jac/dkx127 %J Journal of Antimicrobial Chemotherapy
Systems CLTaIVDT (2007) Susceptibility testing of infectious agents and evaluation of performance of antimicrobial susceptibility test devices part 2: evaluation of performance of antimicrobial susceptibility test devices, vol ISO 20776-2:2007
Acknowledgments
We thank Merck Sharp & Dohme, LDA, who kindly provided ceftolozane-tazobactam for these studies.
Funding
This work received funding from H2020 FTIPilot 2016 project no. 730713 “FAST-bact A novel fast and automated test for antibiotic susceptibility testing for Gram positive and negative bacteria.”
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Rafael Cantón has participated in educational programs sponsored by MSD. FASTinov is a spin-off of Porto University founded by some of the authors.
Ethics statement
The study was approved by the ethical committee of this center (Ref. 161-17).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Martins-Oliveira, I., Pérez-Viso, B., Quintas, S. et al. Evaluation of ultra-rapid susceptibility testing of ceftolozane-tazobactam by a flow cytometry assay directly from positive blood cultures. Eur J Clin Microbiol Infect Dis 39, 1907–1914 (2020). https://doi.org/10.1007/s10096-020-03926-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-020-03926-4